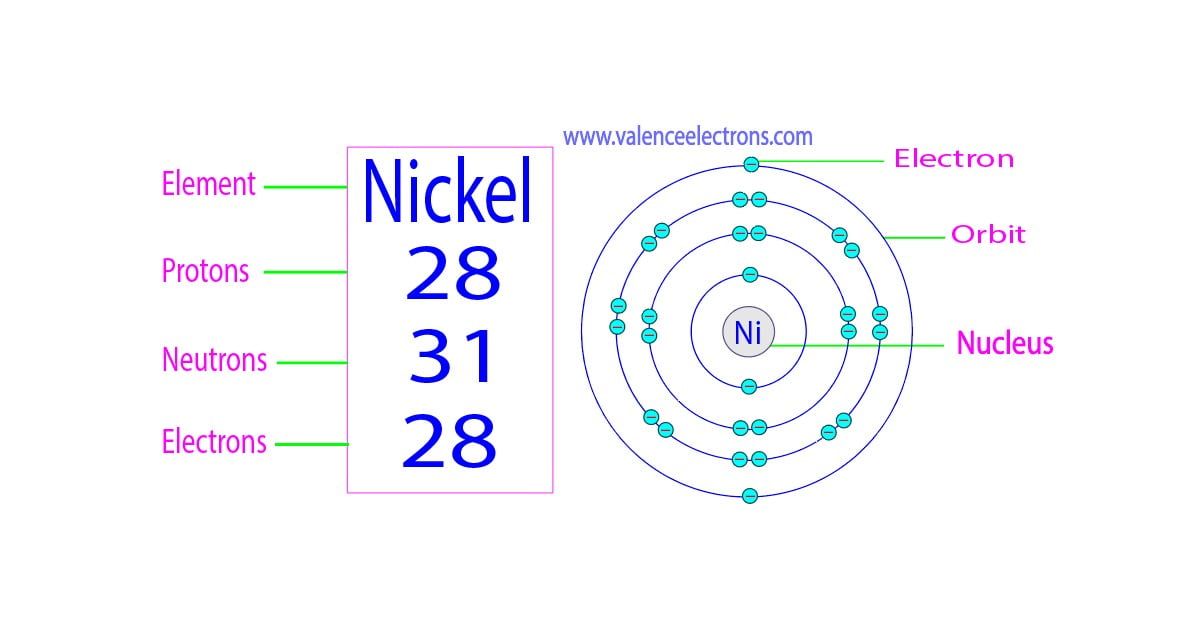
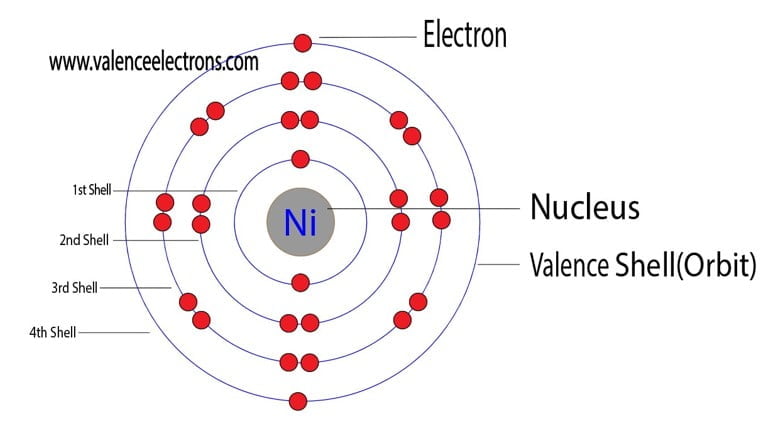
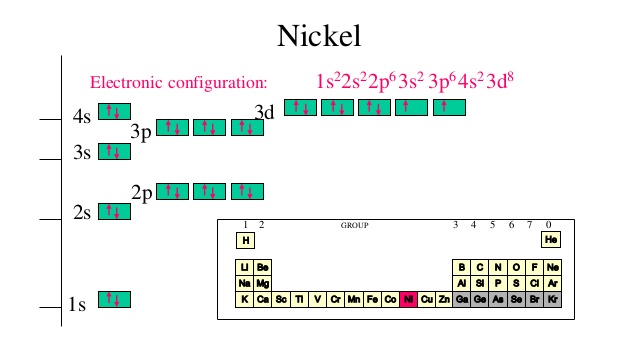
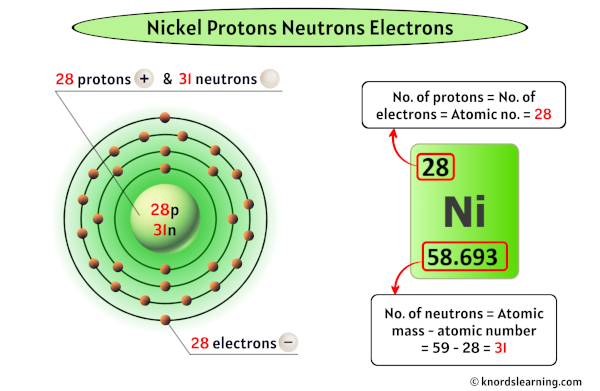
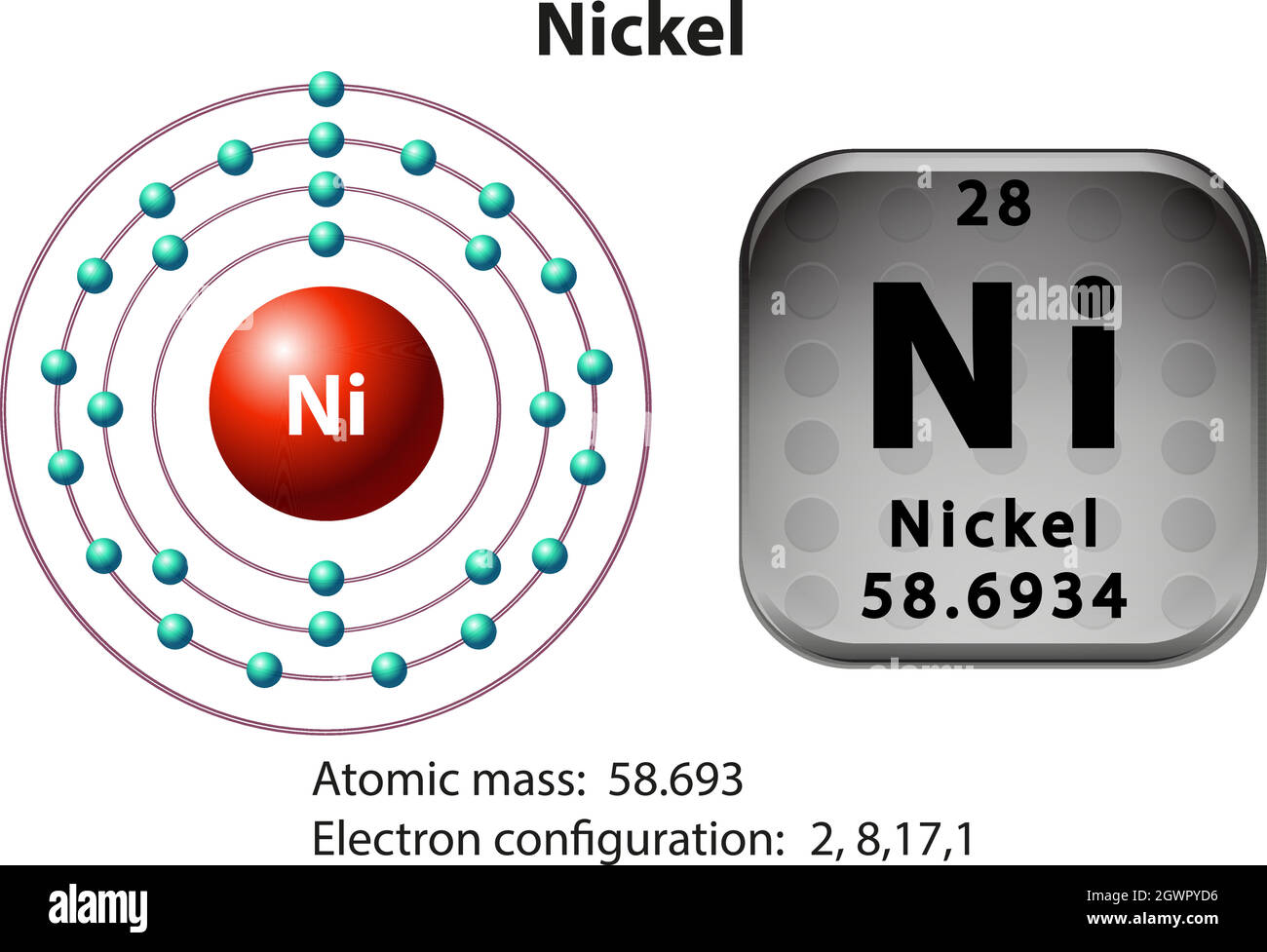
How Many Unpaired Electrons Does Nickel Have

Ever find yourself staring at a particularly shiny coin, or perhaps a sturdy piece of metal and wonder about its inner workings? It’s a common curiosity, a little spark of intrigue that ignites when we encounter the elemental building blocks of our world. And when it comes to those intriguing, often metallic, elements, nickel is a star player. We use it every day, from the change in our pockets to the protective coatings on our electronics, and understanding its properties, like how many unpaired electrons it has, offers a surprisingly satisfying glimpse into why it behaves the way it does. It’s like unlocking a tiny secret about the universe!
Now, you might be thinking, “What do unpaired electrons have to do with my everyday life?” Well, it's more than you might imagine! These little dynamos of energy are the key to how atoms bond together, forming the molecules that make up everything around us. For nickel, the number of unpaired electrons dictates its magnetic properties, its ability to form alloys (those super-strong metal mixtures!), and even its role in chemical reactions. Think about the powerful magnets you use on your fridge or in your stereo speakers – their strength is directly related to the electron configurations of the elements involved, and nickel often plays a part in their magnetic might. It also contributes to the corrosion resistance of many materials, meaning your trusty stainless steel cutlery stays shiny and your car’s body doesn't rust away in a matter of years. That’s a pretty big deal for durability and longevity!
So, how many of these crucial unpaired electrons does nickel actually sport? Drumroll, please... Nickel has two unpaired electrons. This might seem like a small number, but in the quantum world, it’s a significant detail. This configuration is what gives nickel its ferromagnetic properties, meaning it's strongly attracted to magnets and can itself become magnetized. You’ll see this in action in everything from the motors in your appliances to the recording heads in old cassette tapes (remember those?). It's also why nickel is a vital component in many batteries, contributing to their ability to store and release energy efficiently. Even the vibrant green color of some glass can be attributed to nickel ions with their specific electron arrangements.
Must Read
If you’re feeling inspired to delve a little deeper into the fascinating world of elements like nickel, here are a few tips for enhancing your exploration. Firstly, don’t be afraid to get hands-on! While you can’t exactly dissect a nickel to count its electrons, you can observe its properties. Notice how certain objects stick to a nickel-plated surface. Secondly, consider looking at a periodic table. It’s a treasure trove of information, and understanding electron configurations is a fundamental part of deciphering it. Many online resources and apps can visually represent electron shells and unpaired electrons, making the concept much easier to grasp. Finally, connect it back to the real world. The next time you see something made of or containing nickel, take a moment to appreciate the underlying science. It’s these tiny, invisible details, like the two unpaired electrons in nickel, that make our tangible world so wonderfully functional and, well, interesting!