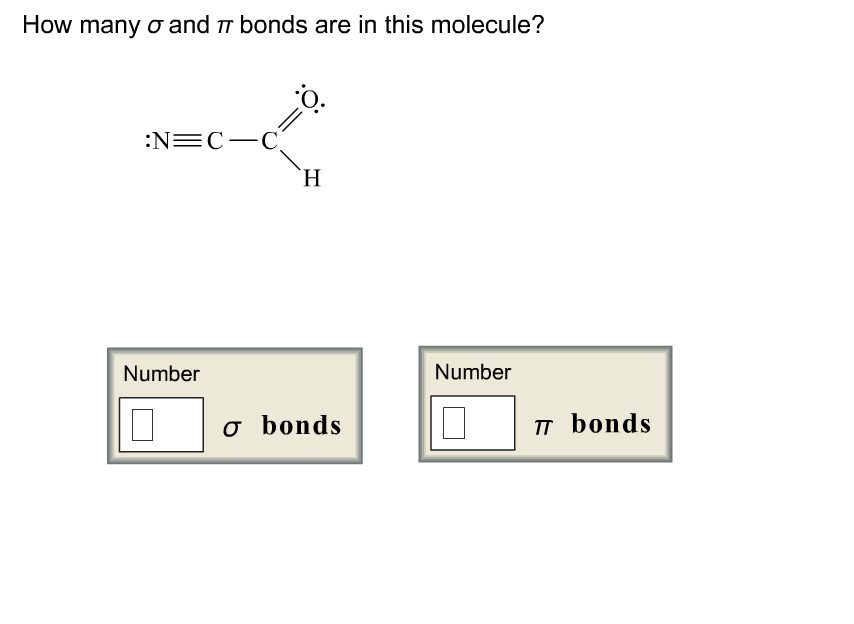
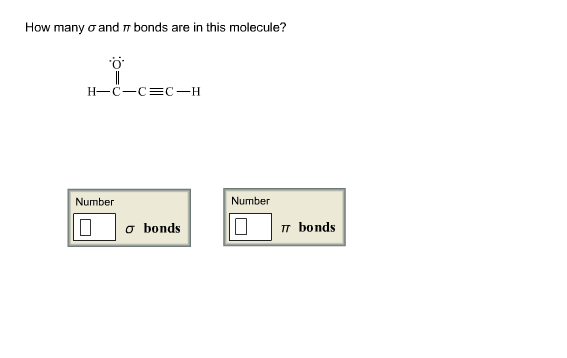
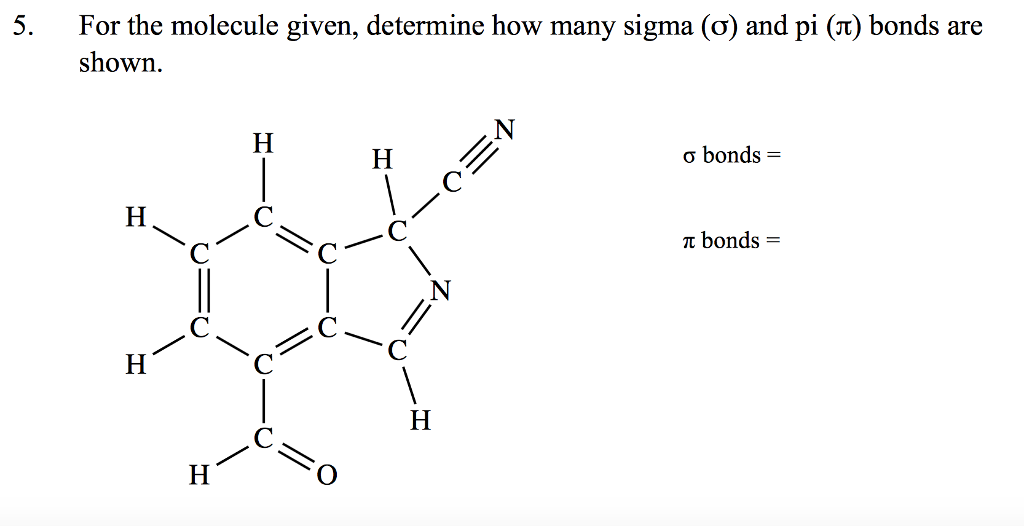
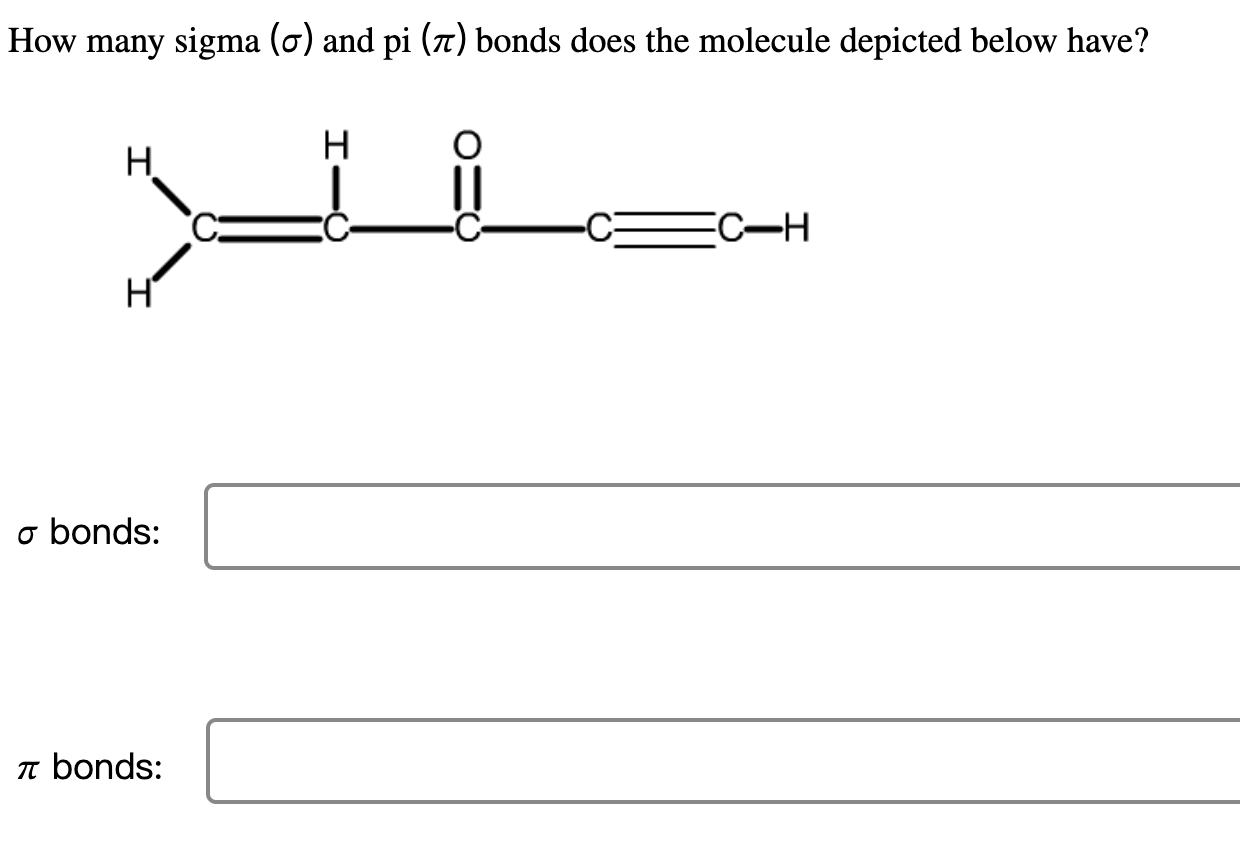
How Many σ And π Bonds Are In Each Compound
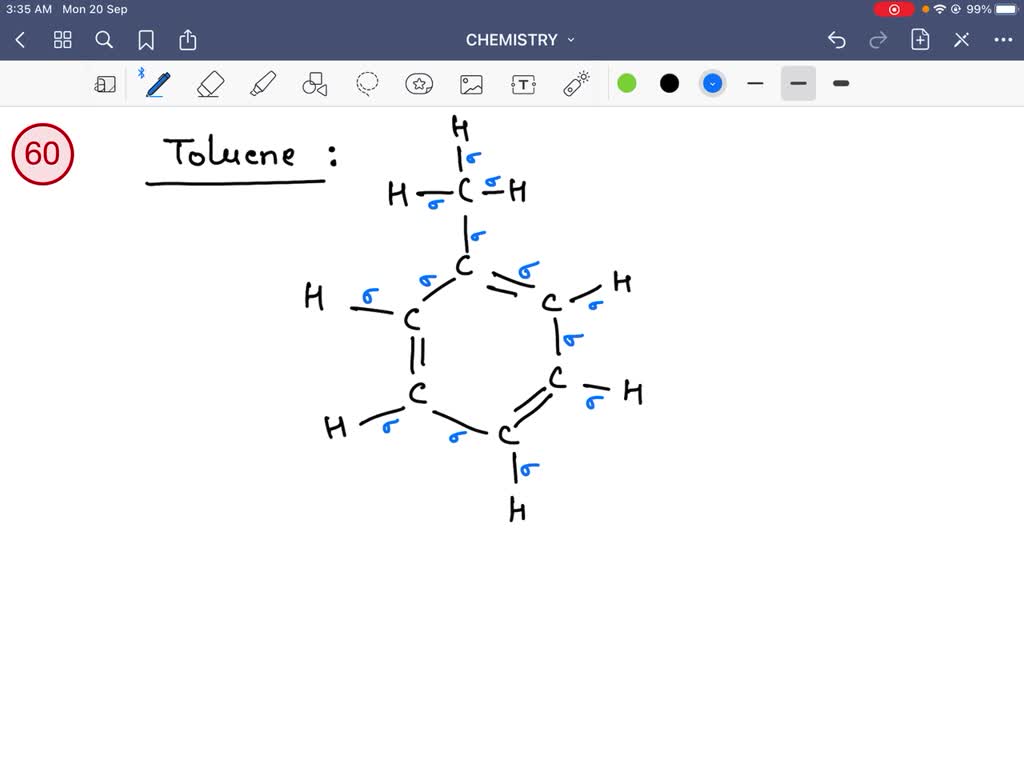
Have you ever marveled at the intricate beauty of a perfectly crafted molecule? The way atoms dance together to form everything from the air we breathe to the vibrant pigments in a painter's palette? While the concept of chemical bonding might sound intimidating, understanding the humble sigma (σ) and pi (π) bonds can unlock a fascinating new perspective, even for the most casual observer. It's not just for seasoned chemists; exploring these fundamental building blocks can spark creativity and offer a surprisingly delightful intellectual challenge, much like the satisfying click of solving a complex puzzle or the joy of discovering a new artistic technique.
For artists, a newfound appreciation for σ and π bonds can breathe fresh life into their work. Imagine creating abstract sculptures inspired by molecular structures, or developing a color scheme based on the electron distribution within a molecule. Hobbyists, whether they're into model building, intricate LEGO creations, or even culinary arts (think about the molecular gastronomy!), can find a deeper understanding of structure and stability. And for casual learners, it’s an accessible gateway to the wonders of chemistry, demystystifying a subject often perceived as abstract and difficult. It’s about seeing the world, quite literally, at an atomic level!
The beauty lies in their simplicity and universality. Sigma bonds, formed by the direct overlap of atomic orbitals, are the sturdy single bonds, the backbone of every molecule. Think of them as the reliable, essential foundation. Pi bonds, on the other hand, are formed by the sideways overlap of p orbitals, creating double and triple bonds. They are the extra layers of interest and reactivity, adding flavor and dynamic possibilities. For example, consider the simple molecule of methane (CH₄). It boasts four σ bonds, all single and incredibly strong, giving it a stable tetrahedral shape. Now, let's look at ethene (C₂H₄), the building block of many plastics. It has five σ bonds and one π bond within its double bond, giving it a planar structure and the ability to undergo exciting chemical reactions. Even something as ubiquitous as water (H₂O) has two σ bonds between oxygen and hydrogen, showcasing the foundational nature of these bonds in everyday substances.
Must Read
Ready to dip your toes into this molecular world? It’s easier than you think! Start with readily available online resources. Many websites offer interactive molecular viewers where you can manipulate and inspect molecules, often highlighting the σ and π bonds. You can also find simple diagrams explaining how these bonds form. If you’re feeling adventurous, consider picking up a molecular model kit. The tactile experience of building molecules can solidify your understanding of how atoms connect and the types of bonds they form. Even sketching out simple Lewis structures for common molecules like carbon dioxide (CO₂) or nitrogen (N₂) can help you identify and count the σ and π bonds.
Ultimately, exploring σ and π bonds is a journey of discovery. It’s about appreciating the elegant architecture of the universe, one atom at a time. It’s about finding a new lens through which to view the world, one that is both intellectually stimulating and surprisingly beautiful. The satisfaction of understanding how these fundamental bonds contribute to the stability, shape, and reactivity of countless compounds is a rewarding experience, proving that even the smallest components of matter hold immense wonder and inspire endless possibilities.