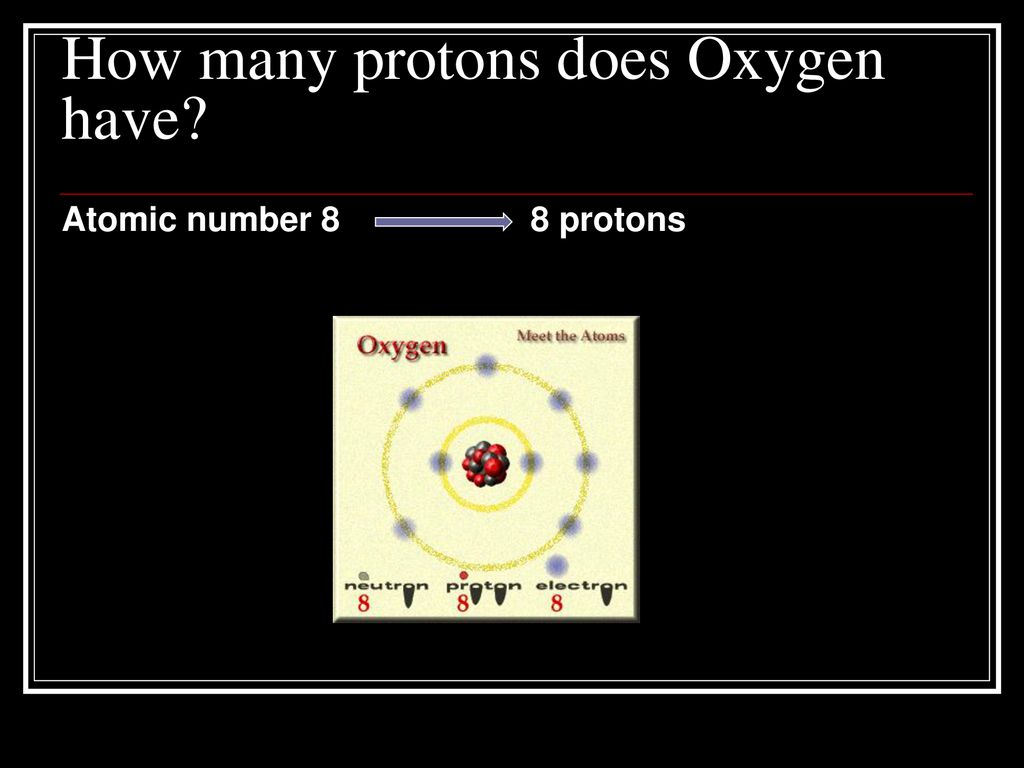
How Many Protons And Electrons Does Oxygen Have
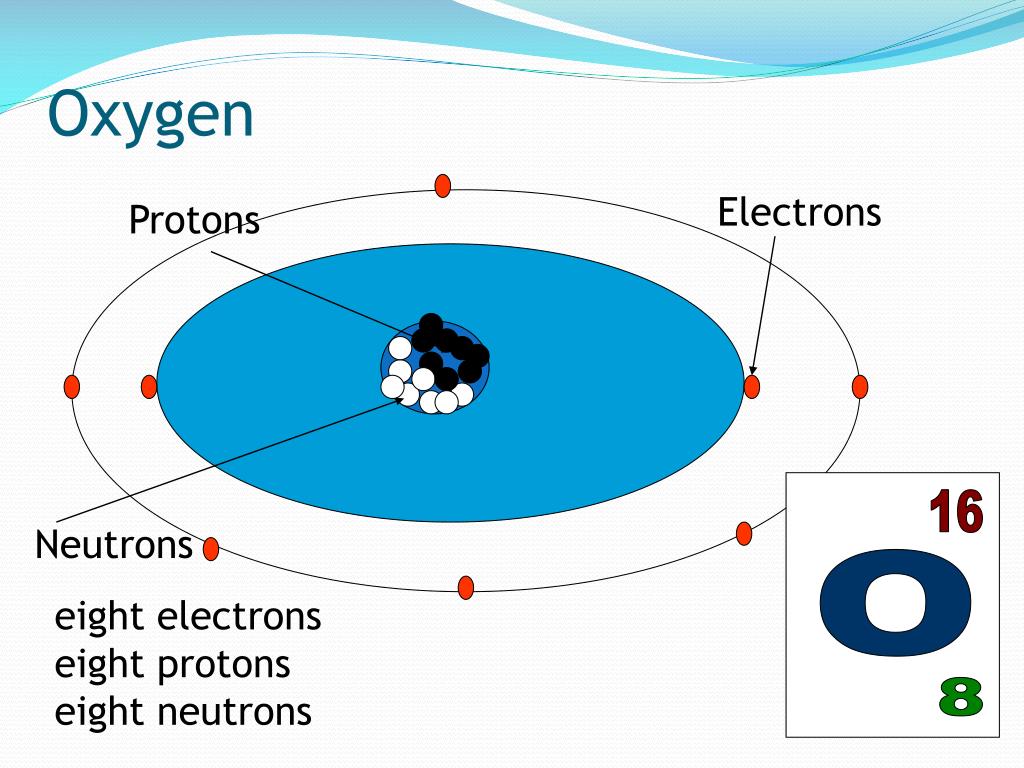
Ever wondered about the tiny building blocks that make up everything around us? It’s like a secret code of nature, and one of the most common characters in that code is Oxygen. You breathe it in every second, it's in your water, and it's even in that yummy cookie you might be dreaming about right now!
But what makes Oxygen so special? It all comes down to its super-tiny parts. Imagine a miniature solar system, but instead of planets and stars, we have even smaller things called protons and electrons. These little guys are like the ingredients in a recipe. Change the number of ingredients, and you get something totally different!
So, how many of these special ingredients does Oxygen have? Get ready for a little bit of atomic magic! It turns out that every single atom of Oxygen has a whopping 8 protons. That’s right, a solid 8! These protons are like the identity card for Oxygen. No matter where you find Oxygen in the universe, if it’s Oxygen, it’s got those 8 protons. It’s like having a secret handshake that only Oxygen atoms know.
Must Read
Now, what about those electrons? These are like the energetic little dancers that zoom around the protons. For a neutral, happy Oxygen atom, it also has 8 electrons. So, we’ve got 8 protons and 8 electrons doing their thing. It's a perfect balance, like a perfectly stacked tower of blocks. This balance is what keeps the Oxygen atom stable and chill.
Isn't that neat? Just knowing this little secret about Oxygen makes you feel like you're in on something big. It's like knowing a secret password to a hidden club. The world is full of these amazing details, and Oxygen is just one of the stars of the show.

Think about it: that breath you just took? It's a bunch of Oxygen atoms, each with its 8 protons and 8 electrons, working together to keep you alive. It’s a cosmic dance happening inside you all the time. Pretty cool, huh?
What’s really fun is how these numbers, 8 and 8, are so fundamental. They dictate how Oxygen behaves. Because it has these specific numbers, Oxygen loves to grab onto other atoms. It’s like it’s always looking for friends to share its electrons with. This is why Oxygen is so important in so many chemical reactions. It's the ultimate team player!
Consider how many things Oxygen is a part of. Water, for instance, is made of Oxygen and Hydrogen. You know, H2O? That O is our star, Oxygen, with its 8 protons and 8 electrons, holding hands with two Hydrogen atoms. And without water, life as we know it just wouldn't be a thing. So, a big shout-out to those 8 protons and 8 electrons for helping create something so essential!
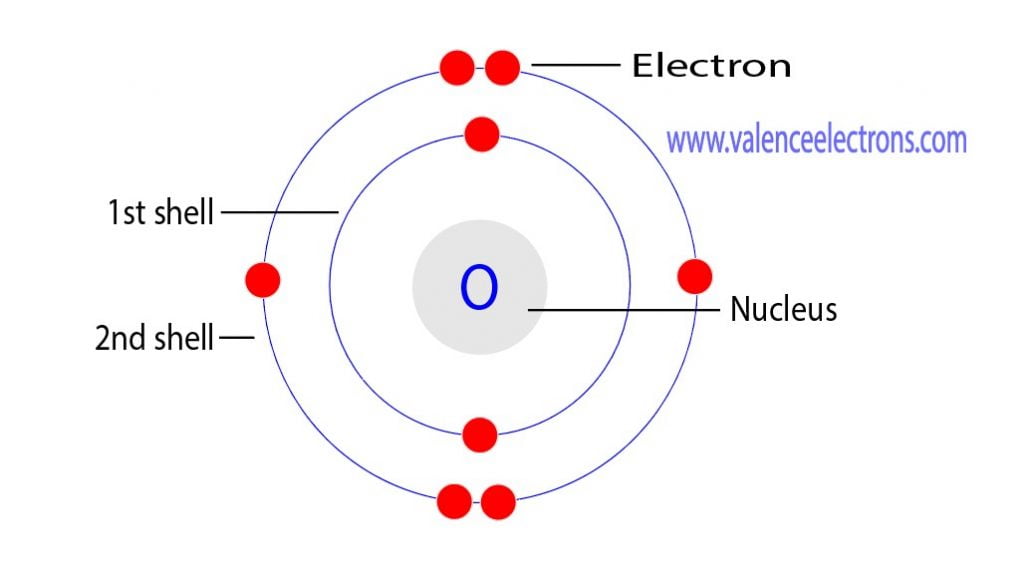
Then there's rust. Ever seen a bike or a fence get all reddish-brown and flaky? That's Oxygen at work, reacting with iron. Again, it’s those 8 protons and 8 electrons playing their part. Oxygen is like a busybody in the best way possible, always joining in and making things happen. It's this eagerness to interact that makes it such a common and important element.
It’s almost like these numbers are a destiny for Oxygen. If it had, say, 9 protons, it wouldn't be Oxygen anymore! It would be Fluorine, a whole different character with its own set of amazing properties. But Oxygen is Oxygen because of those 8 protons. It’s like each element has its own unique fingerprint, and for Oxygen, that fingerprint is the number 8 for protons.

And the 8 electrons? They're not just sitting there. They're buzzing around, looking for ways to connect. This is where the excitement really is! The way these electrons are arranged is what makes Oxygen so reactive. It has these "outer shell" electrons that are just itching to bond with other atoms. It’s like a party where some guests are eager to mingle and make new friends.
So, next time you see a flame, or a plant growing, or even just take a deep breath, remember the humble Oxygen atom. Remember its 8 protons, its guiding number, and its 8 electrons, its energetic dancers. They are the silent architects of so much of our world. It’s a tiny detail, but it’s a detail that unlocks a universe of wonder. It's these fundamental truths, presented in simple numbers, that make the study of atoms so endlessly fascinating. Who knew that counting could be so exciting?
You can find Oxygen everywhere. It’s in the air we breathe, the water we drink, and even in the rocks beneath our feet. And no matter where it is, it’s always carrying its signature of 8 protons and, in its neutral form, 8 electrons. It’s a constant, a reliable player in the grand cosmic game. Isn’t it fun to think that something so common is also so profoundly special, just because of a few numbers?
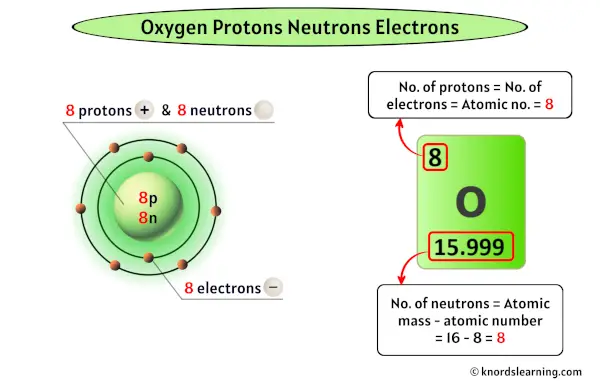
It’s this kind of curiosity that can lead you down the path of discovering even more amazing things about the universe. Every element has its own story, its own special count of protons that defines it. And for Oxygen, that count is a beautiful, life-giving 8. So go on, explore! The world of atoms is waiting with open arms, and its secrets are just waiting to be uncovered, one proton at a time.
Think of it like this: Oxygen is the number 8 in the alphabet of the universe. Simple, yet powerful enough to form endless words and stories!
So, the next time you hear about atoms, remember Oxygen. Remember its 8 protons and its 8 electrons. It's a simple fact, but it's a gateway to understanding so much about how everything works. It’s a little peek behind the curtain of reality, and it’s absolutely captivating.