How Many Orbitals Are Occupied In A Silicon Atom
Ever wondered what's going on inside a tiny, tiny atom? It's like a miniature solar system, but with a whole lot more rules and a lot less sunshine. Today, we're diving into the exciting world of silicon, that amazing element that makes our computers tick and the sand between our toes shimmer.
You see, atoms have these special "rooms" where their tiny particles, called electrons, like to hang out. These rooms are called orbitals. Think of them as cozy little apartments for electrons, and they come in different shapes and sizes, just like real apartments!
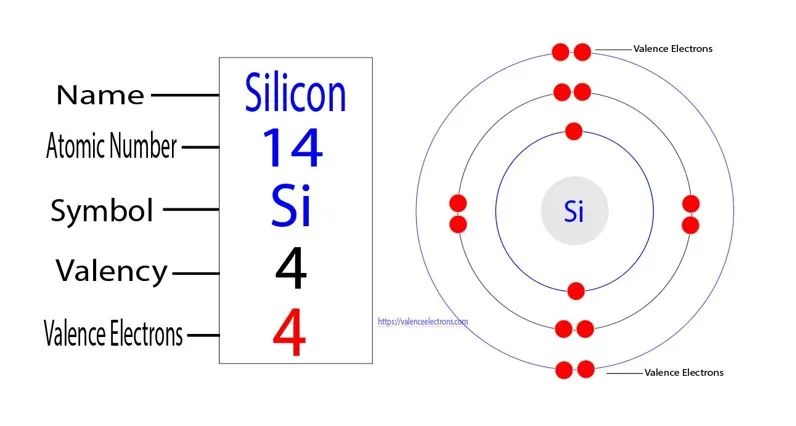
Now, for our star of the show: silicon. This element is super important. It's number 14 on the periodic table, which means it has 14 protons in its core. And in a neutral atom, it also has 14 electrons buzzing around.
Must Read
These 14 electrons don't just float around randomly. Oh no, they have specific places to go, and these places are the orbitals. It’s like assigning seats at a very important, very tiny concert.
So, how many of these electron apartments are occupied in a silicon atom? This is where things get really interesting! It’s not just one or two. We have to fill up these orbital "rooms" following some pretty neat rules.
First, we have the innermost "rooms." These are the 1s orbitals. They are like the super cozy studio apartments, very close to the atom's center. Each orbital can hold a maximum of two electrons. So, for silicon, we fill up one 1s orbital with two electrons. That’s 2 electrons down, 12 to go!
Next up, we move to the "second floor" of our atomic building. Here, we have the 2s orbitals. These are also small apartments, a bit further out than the 1s. Again, each can hold two electrons. So, we fill up the 2s orbital with another two electrons. Now we've used 4 electrons. We’re getting there!
But wait, there’s more on the second floor! We also have the 2p orbitals. These are like slightly larger apartments, and there are three of them. Imagine three identical, but slightly bigger, studio apartments side-by-side. Each of these 2p orbitals can also hold two electrons.
Since there are three 2p orbitals, and each can hold two electrons, these 2p orbitals together can accommodate a grand total of six electrons. So, we fill all three of these 2p orbitals with our next six electrons. That brings our total electrons housed to 2 (from 1s) + 2 (from 2s) + 6 (from 2p) = 10 electrons.

We’re almost done filling the "second floor" apartments! We have 10 electrons placed. But silicon has 14 electrons in total. So, we have 4 more electrons that need a place to stay.
Now we move to the "third floor." Here, we find the 3s orbitals. Just like the 1s and 2s, it's a single, small apartment. It can hold two electrons. So, we place two of our remaining electrons into the 3s orbital. Now we have 10 + 2 = 12 electrons accounted for.
We’ve got 2 electrons left! Where do they go? They head to the "third floor" as well, but into a different type of apartment: the 3p orbitals. These are like the 2p orbitals – there are three of them, and each can hold two electrons.
Silicon has 4 electrons left to place, and the 3p orbitals can hold up to 6. So, we fill two of the 3p orbitals with one electron each. That's 2 electrons. And then, we add the final two electrons, one into each of the remaining 3p orbitals. So, two of the 3p orbitals will have one electron, and one of the 3p orbitals will have two electrons. This uses up our final 4 electrons. Total electrons = 12 + 4 = 14. Perfect!
So, let's count how many orbitals are actually occupied with electrons in a silicon atom. We have:
The 1s orbital: 1 occupied orbital (with 2 electrons).

The 2s orbital: 1 occupied orbital (with 2 electrons).
The 2p orbitals: 3 occupied orbitals (all filled with 2 electrons each, totaling 6 electrons).
The 3s orbital: 1 occupied orbital (with 2 electrons).
The 3p orbitals: 3 occupied orbitals (partially filled, with a total of 4 electrons spread across them).
Let's tally them up! 1 (from 1s) + 1 (from 2s) + 3 (from 2p) + 1 (from 3s) + 3 (from 3p) = 9 occupied orbitals!
That's right, 9 orbitals are occupied by electrons in a single silicon atom. Isn't that neat? It’s like a busy little atomic apartment building, with most of the "rooms" taken.

What makes this so entertaining is imagining these electrons zipping around in their specific shapes. The 's' orbitals are like little spheres, while the 'p' orbitals are shaped like dumbbells, all clustered around the atom's nucleus. It's a dance of probability and energy levels.
The fact that silicon has these partially filled 3p orbitals is what makes it so special and useful in our technology. These "outer" electrons are the ones that get to interact with other atoms. They are the ones that form the bonds that create all the amazing materials we use, from computer chips to glass.
Think of those outer electrons in the 3p orbitals as the outgoing, social electrons of silicon. They are the ones ready to make new friends (form chemical bonds) and help build incredible structures.
It's the unique arrangement and the number of occupied orbitals, especially the ones on the outer shell, that give silicon its superpowers. It's a semi-conductor, meaning it can conduct electricity under certain conditions, which is the foundation of all modern electronics. All because of how its electrons are arranged in these orbitals!
So, the next time you use a smartphone or a computer, you can think about the humble silicon atom and its 9 occupied orbitals. It's a tiny world with big implications. It’s a little bit like understanding a secret code that unlocks our modern world.
The beauty of it is that this isn't just a dry fact; it's the basis of chemistry and physics. It's the reason why different elements behave differently and why they can combine to form everything around us. Every element has its own unique story of occupied orbitals.
And silicon's story is particularly exciting because of its role in our technological revolution. It's a testament to how understanding the very small can lead to some truly colossal innovations.
It's like uncovering the blueprint of an incredibly complex and functional machine. Each occupied orbital is a vital piece of that blueprint. And for silicon, this specific arrangement allows it to be the building block of our digital age.
So, while the number 9 might seem like a simple count, it represents a whole universe of atomic activity. It's a universe that's been harnessed and understood to create the amazing devices we rely on every day. It’s a fantastic example of how fundamental science can lead to incredible, tangible results.
The journey of those 14 electrons, filling up their designated orbitals, is a fundamental process. And for silicon, this process results in an element with properties that are nothing short of extraordinary. It's a fundamental aspect of its identity and its utility.
Isn't it amazing to think that the complex workings of a computer are built upon the simple arrangement of electrons in atomic orbitals? It's a story that starts small and becomes incredibly significant.
The next time you hold a piece of technology, remember the 9 occupied orbitals of a silicon atom, silently powering the world around you. It’s a fascinating piece of the puzzle that makes up our reality, and a truly entertaining one at that!
So, go on, be curious! Dive a little deeper. The world of atoms is full of surprises, and silicon's story is just one of many captivating tales waiting to be discovered.
