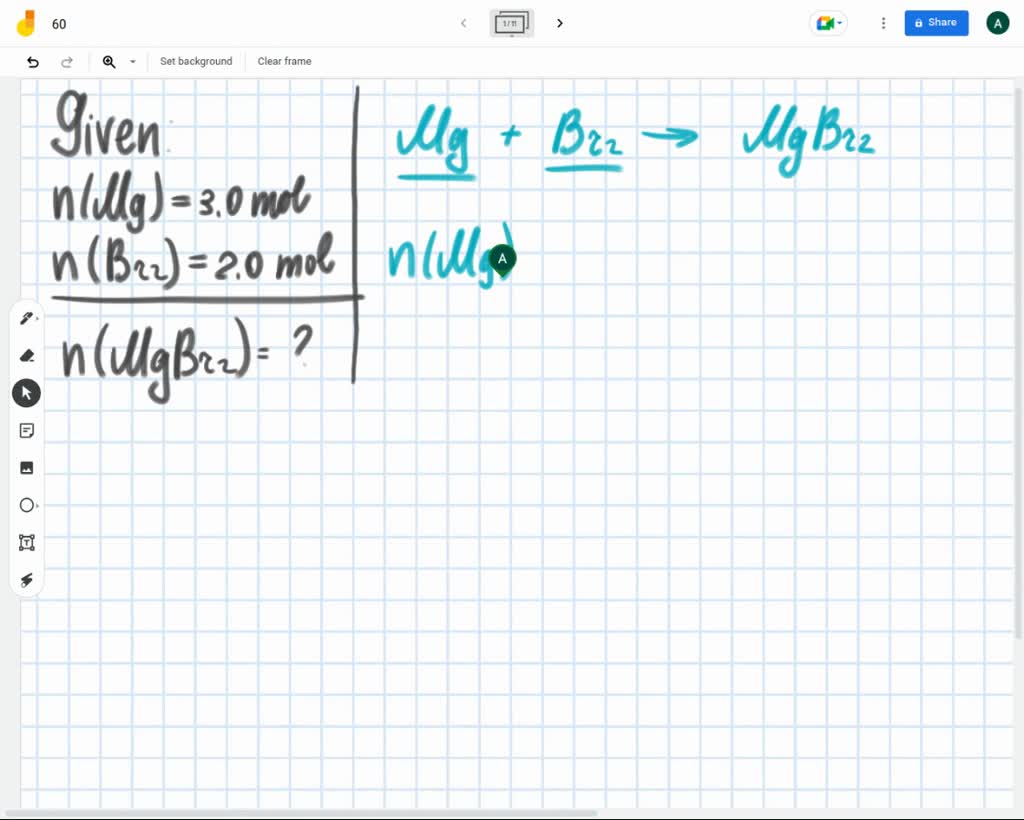
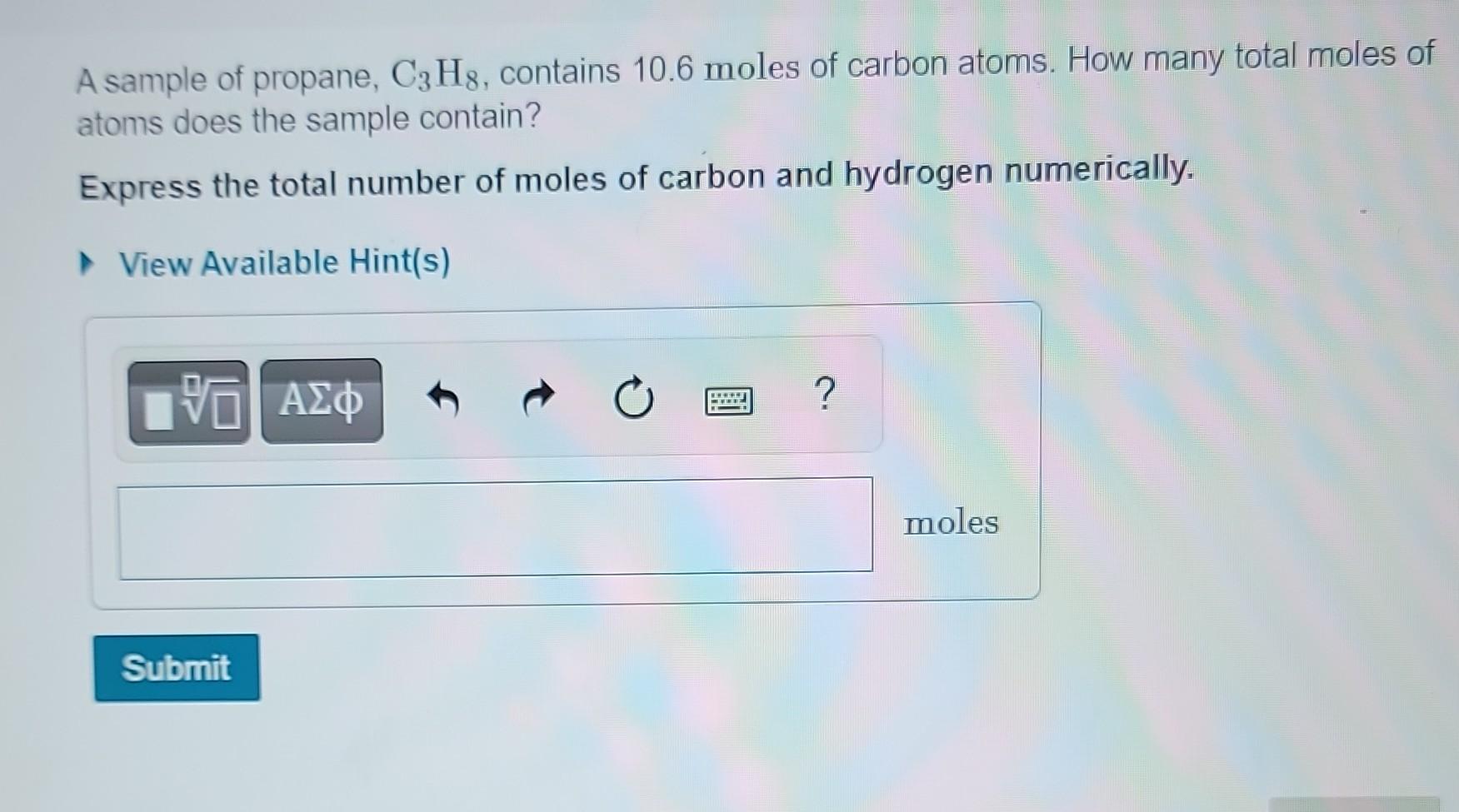
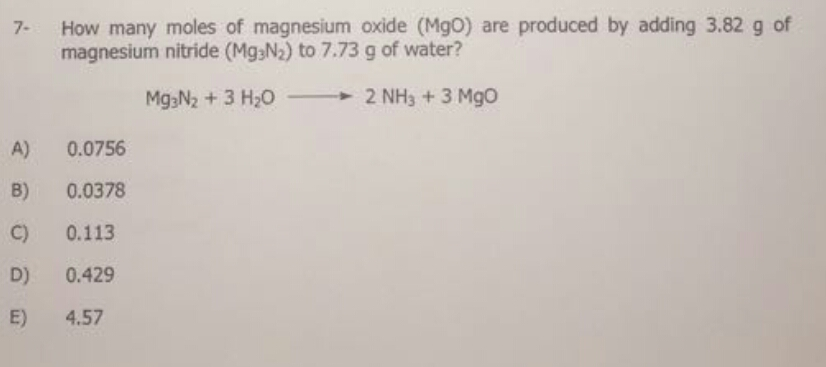How Many Moles Of Magnesium Is 3.01 X 10 22
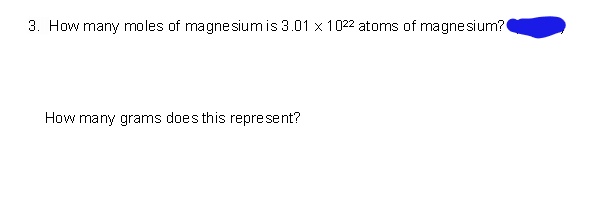
Hey there, science explorer! Ever looked at a number like 3.01 x 1022 and thought, "Whoa, what even is that?" Well, buckle up, buttercup, because we're about to dive into the wonderfully weird world of chemistry, and specifically, we're going to figure out how many moles of magnesium are hiding in that massive number. It sounds fancy, but trust me, it's more fun than it looks!
So, what's a mole, anyway? Think of it as a baker's dozen, but for atoms and molecules. A dozen is 12 things. A baker's dozen is 13. A mole is... well, it's a lot more. It's 6.022 x 1023 of something. We call this special number Avogadro's number. Yep, some dude named Avo-who-dro figured out this magic number, and now we use it for everything in chemistry.
Imagine you're counting grains of sand on a beach. Now imagine you're counting atoms. Suddenly, that mole thing starts to make a lot more sense, right? Atoms are TINY. So, so, so tiny. You'd need an absurdly large number to even have a sniff of a tangible amount. A mole is just our way of making those impossibly small numbers manageable.
Must Read
Now, let's talk magnesium. This stuff is pretty cool. Did you know magnesium is what makes fireworks go boom and sparkle? It burns with a super bright, white light. Pretty neat, huh? It's also essential for life! Your bones have magnesium. Your muscles need it. So, it's not just about fancy numbers; it's about the stuff that makes the world (and you!) tick.
Okay, back to our number: 3.01 x 1022. This is our "quantity of magnesium stuff." We want to know how many groups of 6.022 x 1023 (that's one mole!) fit into this quantity. It's like asking, "If I have 24 cookies, and a pack has 12 cookies, how many packs do I have?" Easy peasy, right?
In chemistry, we do a similar thing. We take our given number (3.01 x 1022) and divide it by Avogadro's number (6.022 x 1023). This is where the magic happens. It's like a cosmic calculation, but with much less glitter than fireworks.
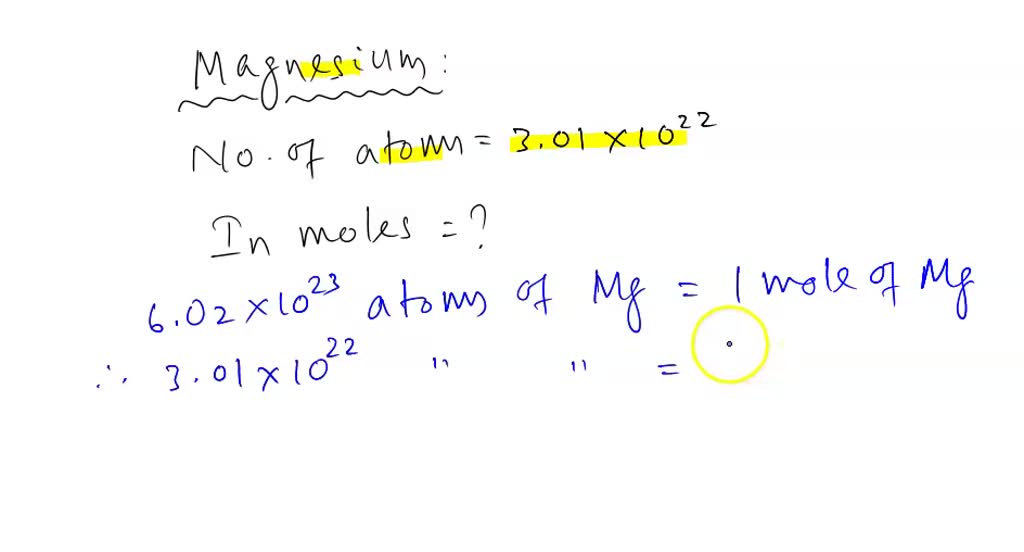
So, here's the math part, but don't sweat it! We're dividing 3.01 x 1022 by 6.022 x 1023. Let's break down those giant numbers. The "x 10 to the power of something" part is just a shorthand for moving a decimal point a gazillion times. It's a scientist's trick to avoid writing out all those zeroes!
When you divide these two numbers, something really fun happens. Let's look at the numbers without the "x 10" part first: 3.01 and 6.022. What's 3.01 divided by 6.022? It's almost exactly 0.5! Isn't that neat? It's like finding a perfect half.
Now, let's think about the "x 10" parts. We have 1022 divided by 1023. When you divide powers of 10, you subtract the exponents. So, 22 minus 23 equals -1. This means we have 10-1, which is the same as 0.1.

So, putting it all together, we have 0.5 multiplied by 0.1. That gives us... drumroll please... 0.05!
Ta-da! 3.01 x 1022 particles of magnesium is equal to 0.05 moles of magnesium. See? Not so scary after all!
What does 0.05 moles mean in real life? It means you have a very, very small fraction of a mole. If a mole is a whole pizza, 0.05 moles is like a couple of delicious slices. It's not a huge amount, but it's still a lot of magnesium atoms when you think about how small they are!

Think about it: if one mole is Avogadro's number of atoms, then 0.05 moles is 5% of Avogadro's number. That's still roughly 3 x 1022 atoms. It's mind-boggling! It’s a quantity that’s too big to count by hand, but too small to see easily with the naked eye. It lives in that fascinating in-between space.
Why is this fun? Because it connects the impossibly tiny world of atoms to the numbers we can actually write down and understand. It’s like having a secret code for the universe. When you see a chemical formula, or a problem like this, you're actually deciphering how much of a certain ingredient is there.
Magnesium has an atomic mass of about 24.3 grams per mole. So, 0.05 moles of magnesium would weigh about (0.05 moles) * (24.3 grams/mole) = 1.215 grams. That's about the weight of a paperclip! So, a tiny paperclip's worth of magnesium has about 3 x 1022 atoms in it. Mind. Blown.
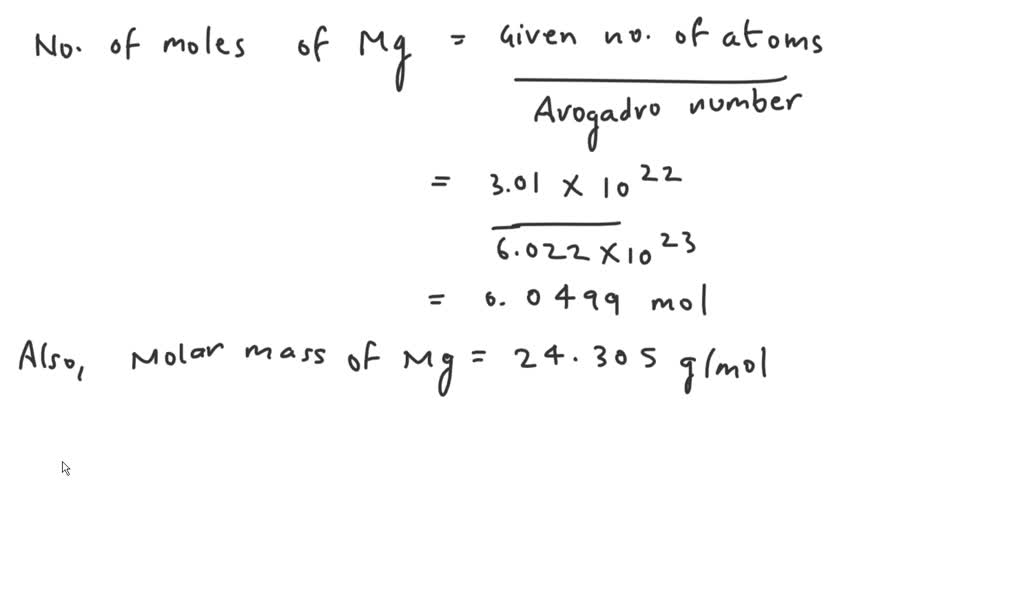
It's also fun because numbers like 3.01 x 1022 are everywhere in science. In biology, you might talk about the number of DNA molecules. In physics, you might talk about the number of electrons. Once you understand the mole, you unlock a whole new level of understanding these scientific concepts.
So, next time you see a giant number in a science book, don't run away! Think of it as an invitation. An invitation to explore. An invitation to calculate. An invitation to understand the building blocks of everything around you. And who knows, maybe you'll even start to see the fun in figuring out how many moles of, say, sparkly fireworks-making magnesium you've got!
It’s a little bit like being a detective, but your clues are numbers and your mystery is the universe itself. And the best part? You don't need a trench coat, just a little bit of curiosity and a willingness to play with big numbers. And remember, even a small fraction of a mole is still an enormous amount of "stuff" when you're talking about atoms.
So there you have it! 3.01 x 1022 magnesium things? That's 0.05 moles of magnesium. A small, but mighty, amount. Now go forth and impress your friends with your newfound mole-tastic knowledge!