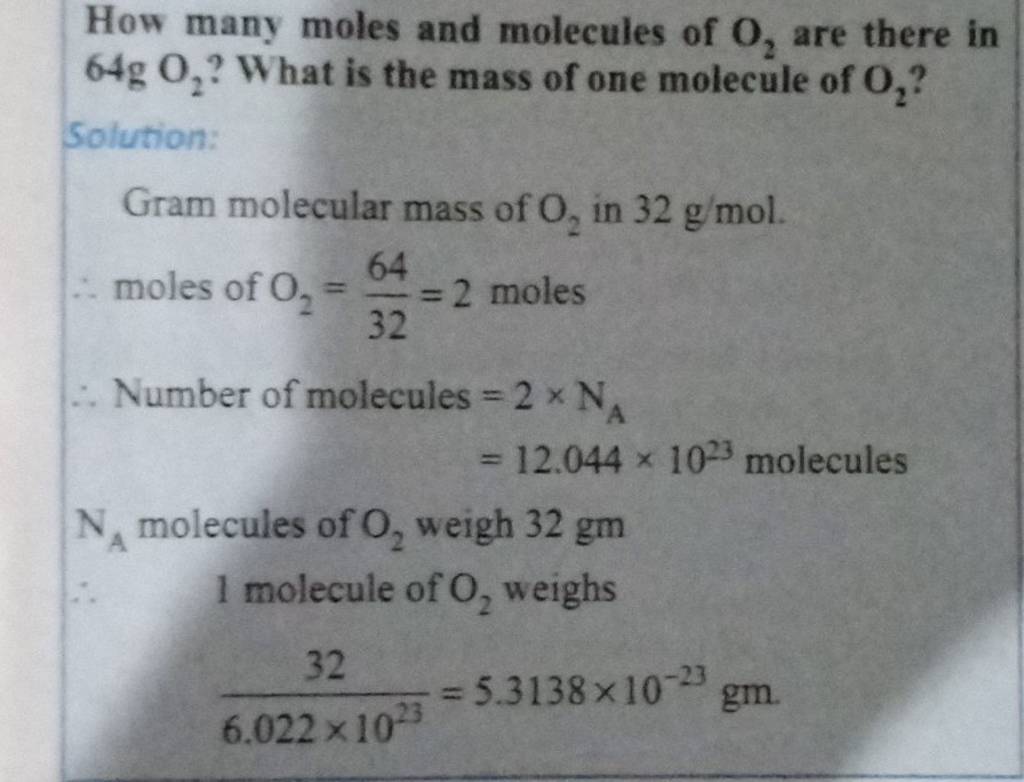How Many Molecules Are There In 2.1 Mol Co2
Ever find yourself staring at a fizzy drink, maybe a delicious latte, or even just taking a deep breath of that crisp evening air, and wonder… what’s really going on in there? It’s easy to think of the world as solid, tangible stuff, but beneath the surface, it’s a dazzling dance of tiny particles. Today, we’re going to dive into one of those fascinating, microscopic universes: the world of molecules. And we’re going to tackle a question that might sound a bit intimidating, but trust me, it’s as simple as counting sprinkles on a cupcake: how many molecules are there in 2.1 mol of CO2?
Now, before your eyes glaze over and you start picturing complex chemistry equations, let’s take a breath. We’re not in a sterile lab coat situation here. Think of this more like a peek behind the curtain of your favorite cafe. That bubbly CO2 in your soda? It’s everywhere! It's the stuff that makes plants grow, it's a byproduct of us breathing (that’s right, you’re exhaling CO2 right now!), and it plays a role in everything from climate to that satisfying sigh of relief after a long day. So, understanding its molecular makeup isn’t just for scientists; it’s a way to appreciate the intricate world around us.
Let’s break down the star of our show: CO2, or carbon dioxide. It’s made of one carbon atom and two oxygen atoms, all linked together. Imagine a tiny, three-part structure, like a Mickey Mouse head if the ears were oxygen and the face was carbon. Simple, right? But even this simple structure is made up of atoms, and atoms themselves are mostly empty space, with a nucleus and some electrons buzzing around. We could go down that rabbit hole, but for today, we’re focusing on the larger building blocks: molecules.
Must Read
So, what’s this “mol” thing? This is where things get really cool, and honestly, pretty ingenious. The “mol” is a unit of measurement, like a dozen eggs or a pound of sugar. But instead of counting discrete items, a mole counts a specific, enormous number of particles. This number is called Avogadro’s number, and it’s roughly 6.022 x 10^23. Let that sink in. That’s a 6 followed by 23 zeros! It’s a number so vast it’s almost incomprehensible. Think of it as the universe’s ultimate counting tool for the teeny-tiny things.
Why do scientists use such a massive number? Well, imagine trying to count individual grains of sand on a beach. It’s impossible, right? But if you knew the average weight of a grain of sand, you could figure out how many were there by weighing a bucketful. The mole concept works similarly. It’s a way to bridge the gap between the microscopic world of atoms and molecules and the macroscopic world we can actually measure and weigh in a lab. It allows chemists to work with practical amounts of substances while still knowing the exact number of particles they’re dealing with.
Our question is about 2.1 mol of CO2. We know that 1 mol of anything contains Avogadro’s number of particles. So, if we have 2.1 mol, we simply need to multiply Avogadro’s number by 2.1. It’s like saying, “If one dozen cookies is 12 cookies, then 2.1 dozen cookies is 2.1 times 12.” In our case, the “dozen” is this humongous 6.022 x 10^23.

Let’s do the math. We’re multiplying 2.1 by 6.022 x 10^23. On your calculator, this might look something like 2.1 * 6.022E23. The result will be approximately 1.265 x 10^24. This is the staggering number of CO2 molecules present in 2.1 moles of carbon dioxide. Isn’t that mind-blowing?
To put that number into perspective, imagine trying to count all the stars in the observable universe. Even that might not get you to 1.265 x 10^24. It’s a number that highlights the sheer abundance of matter, even in seemingly small amounts. Think about the air you breathe. That’s made up of countless molecules of nitrogen, oxygen, and yes, carbon dioxide.
Let’s throw in a fun little fact. The term “molecule” itself comes from Latin, meaning “little mass.” It’s a fitting name, considering how incredibly small these units are. And for CO2, the structure we mentioned earlier? It’s actually a linear molecule, meaning the atoms are in a straight line: O-C-O. It’s not bent like water, for example.
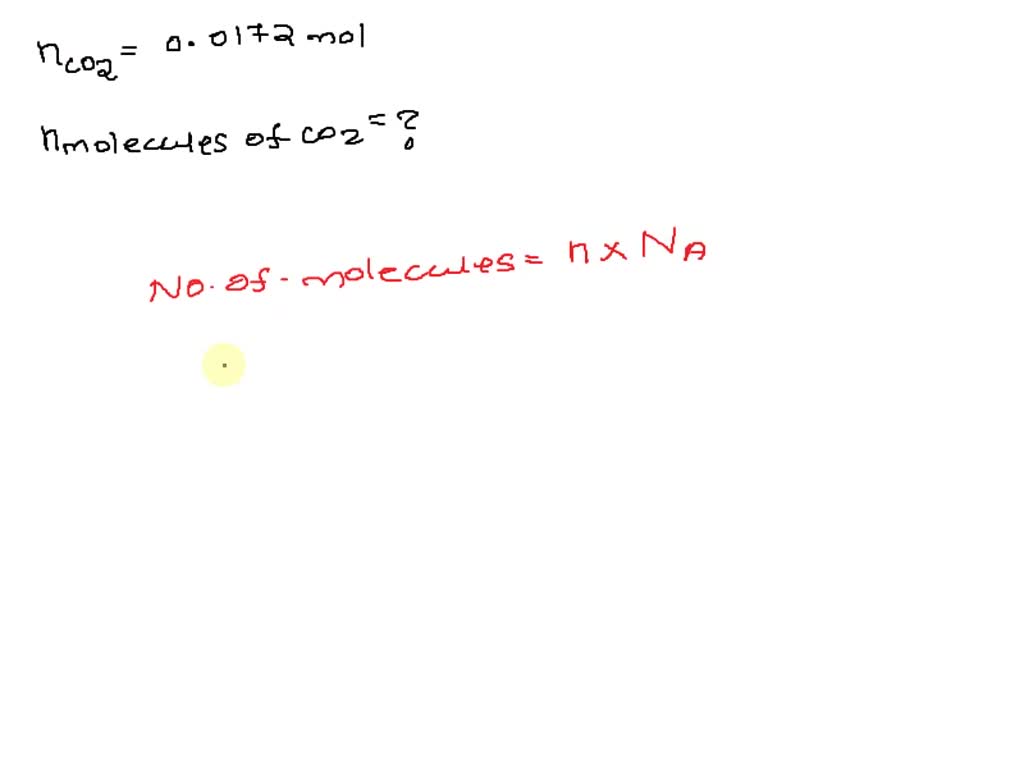
Another interesting tidbit is how we talk about moles in everyday language. When you hear someone say, “I’m feeling a bit moody today,” it’s a linguistic echo of a chemical concept! While “moody” refers to a state of mind, “mole” in chemistry is about a quantity. It’s a delightful quirk of language that sometimes scientific terms find their way into common parlance, albeit with a different meaning.
Consider the process of baking a cake. You use precise measurements of flour, sugar, and eggs, all measured in grams or cups. But underlying those measurements is the exact number of molecules of each ingredient. The chemical reactions that make your cake rise and become delicious happen at the molecular level. Understanding the mole helps chemists predict how much of a reactant they need to achieve a desired outcome, just like a baker follows a recipe to get a perfect cake.
Think about your daily coffee. The beans themselves are made of complex organic molecules. When you roast them, you’re changing their chemical structure. When you brew your coffee, hot water extracts soluble compounds, forming that delicious liquid. And the steam you see rising? That’s mostly water vapor, but it also carries tiny amounts of volatile aromatic molecules that give coffee its signature scent. Even in that comforting mug, there are trillions upon trillions of molecules at play.

Now, let’s think about some practical tips, even if they’re more about appreciating the concept than performing calculations in your kitchen. The next time you’re at a grocery store, look at the labels on packaged goods. You might see information about nutrition, but it’s all based on the molecular composition of the food. Vitamins, proteins, carbohydrates – they are all complex molecules that our bodies break down and use.
And when you’re enjoying a sparkling beverage, whether it’s champagne or a probiotic soda, remember that the fizz is a direct result of dissolved CO2 escaping. That effervescence is a visible manifestation of those 1.265 x 10^24 molecules (or a fraction thereof, depending on the volume!) doing their thing.
Perhaps the most significant cultural reference point for CO2 molecules, especially in recent times, is its role in our environment and climate change. The increase of CO2 in the atmosphere, largely due to human activities like burning fossil fuels, is a critical concern. Understanding that even a seemingly small increase in concentration means a vast number of additional molecules is crucial to grasping the scale of the issue.
So, we’ve established that 2.1 mol of CO2 contains approximately 1.265 x 10^24 molecules. That’s a number so big it makes your head spin, and yet, it’s a perfectly normal amount in the world of chemistry. It’s the foundation upon which our understanding of matter is built.
Let’s take a moment for a short reflection. We started with a seemingly technical question about molecules. But by exploring it, we’ve touched upon the fundamental building blocks of our universe, the cleverness of scientific measurement, and the pervasive presence of matter in our daily lives. From the air we breathe to the food we eat, to the very planet we inhabit, everything is a grand, molecular symphony.
The next time you see a plant unfurling a new leaf, or feel the warmth of the sun, or even just exhale a sigh, remember that behind these everyday phenomena lies an astonishing dance of molecules. It’s a reminder that even the smallest units have immense power and contribute to the grand tapestry of existence. And that, my friends, is a pretty cool thing to ponder. So go forth, and appreciate the tiny, but mighty, world of molecules!