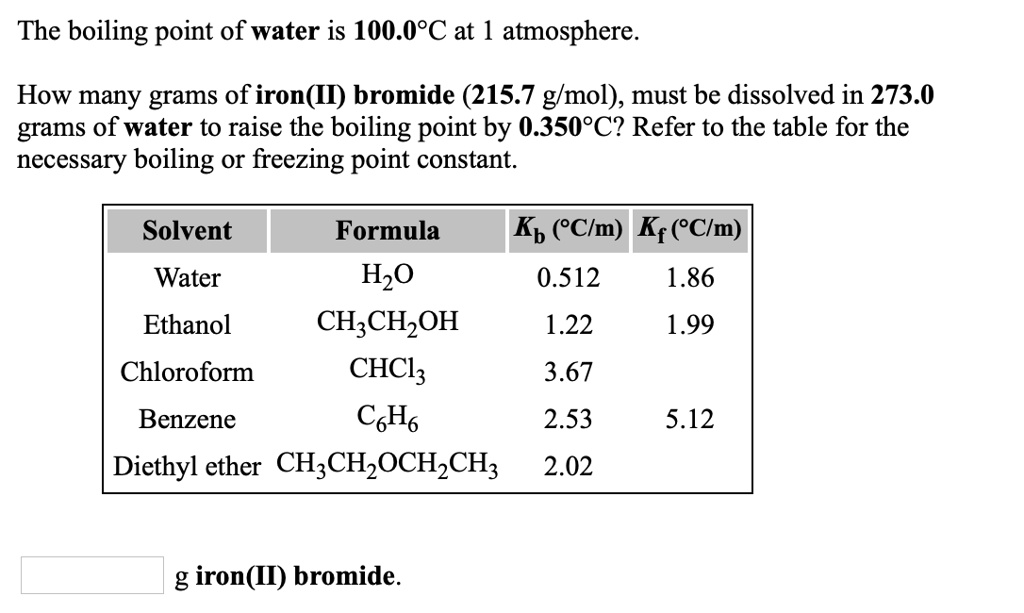
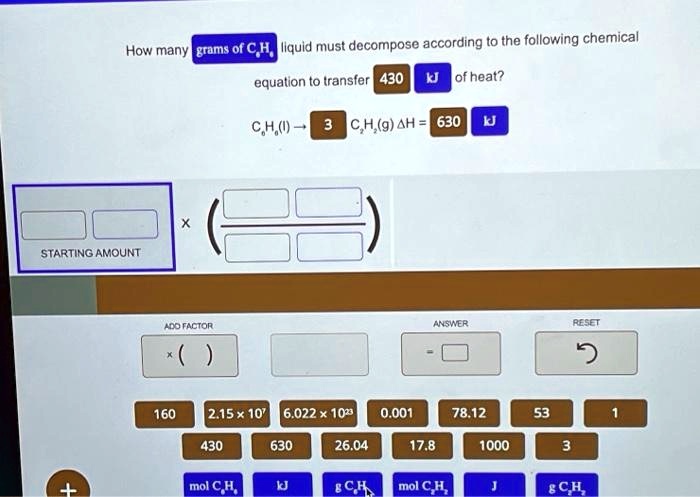
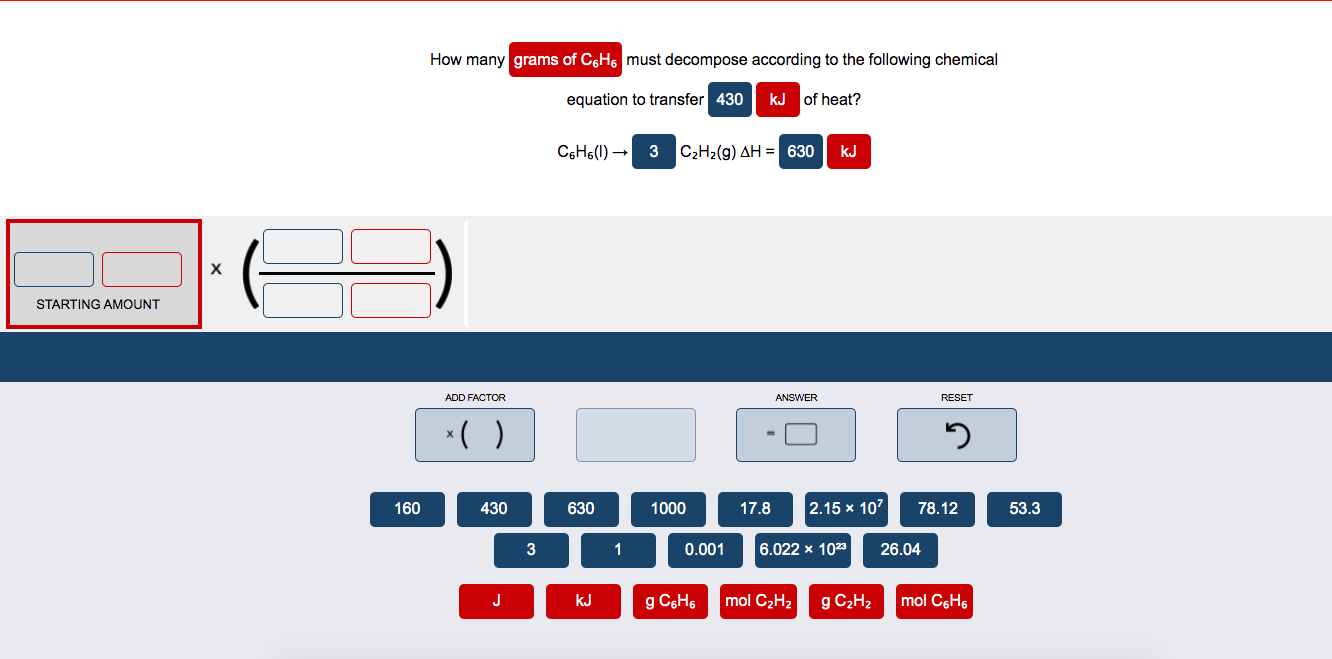
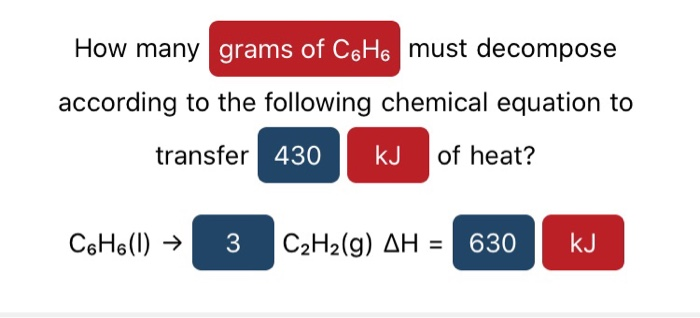
How Many Grams Of C6h6 Liquid Must Decompose

Ever found yourself staring at a mysterious little bottle of liquid, wondering what its secrets are? Maybe it’s a science experiment gone rogue, or perhaps you’re just that kind of curious person. Today, we're diving into the wonderfully weird world of chemical decomposition. Specifically, we're going to ponder a rather peculiar question: how many grams of C6H6 liquid must decompose?
Now, before you picture exploding beakers and mad scientists cackling, let’s dial it back. This isn't about causing a chemical catastrophe. It’s more about a gentle nudge, a little chemical nudge, to see what happens. Think of it like asking a grumpy cat how much tuna it needs to purr. It’s a question that tickles the brain.
Our star player today is C6H6. You might know it by a much cooler, more down-to-earth name: benzene. Yes, that very same benzene. It’s a ubiquitous little molecule, found in all sorts of places, some more pleasant than others. But when it decides to decompose, oh boy, does it get interesting.
Must Read
So, the big question looms: how many grams? It’s not like there’s a pre-set button for “Decompose This Much Benzene.” Life, and chemistry, rarely works like that. We’re not dealing with a recipe here. This is more like a philosophical inquiry, with a dash of chemical jargon.
Let’s be honest, asking “how many grams of C6H6 liquid must decompose” feels a bit like asking “how much chocolate is too much?” The answer, of course, is subjective and depends entirely on the circumstances. For some, one gram is a catastrophe. For others, it’s barely a warm-up.
The beauty of this question lies in its ambiguity. It invites us to consider the forces at play. What makes a molecule decide, “You know what? I’m done with this.” Is it heat? Light? A particularly aggressive catalyst? It’s a chemical existential crisis.
Imagine a tiny little molecule of C6H6. It’s happily buzzing along, part of the liquid collective. Then, something happens. A tiny jolt of energy, perhaps. And suddenly, it’s breaking apart. Into what, you ask? Ah, that’s another delightful layer of complexity.
Benzene, with its six carbon atoms and six hydrogen atoms (hence the C6H6), is quite stable. It’s like a well-built house of molecular LEGOs. To get it to break down, you need to apply some serious persuasion. This persuasion usually comes in the form of significant energy input.
So, if we’re talking about a truly spontaneous decomposition, the kind where the benzene just shrugs and falls apart on its own with no outside help? Well, that's a rare event. It would take an astronomical amount of time, far longer than any of us will be around to witness.
This is where our "unpopular opinion" might start to creep in. Perhaps the answer to "how many grams must decompose" is simply: "the ones that feel like it." It's a bit cheeky, I know. But honestly, who are we to dictate the chemical destiny of a molecule?
Think of it this way. You have a pile of perfectly good cookies. How many cookies must you eat before you’re “full”? It’s a question with no single right answer, is it? Some people are content after two. Others see a whole box as a personal challenge.
Similarly, the decomposition of C6H6 is a process governed by kinetics and thermodynamics. It's not a simple "on/off" switch. It's more like a dimmer switch, with varying degrees of “breaking down.”
If we’re talking about controlled decomposition, like in a laboratory setting to produce other chemicals, then the amount is dictated by the desired outcome. You need a certain amount to yield a certain amount of product. It’s about stoichiometry, the science of how much of what you need.
But the question, as phrased, has a lovely open-endedness to it. “How many grams… must decompose?” The word “must” implies a necessity, an obligation. And who is obligating this poor benzene molecule?
Perhaps it’s the universe, in its infinite wisdom, that dictates when a molecule has lived its fullest benzene life and is ready for a change. Or perhaps it’s the relentless march of entropy, the universal tendency towards disorder, that eventually nudges even the most stable molecules to break free.
Let's imagine a single gram of C6H6 liquid. That's a tiny, almost imperceptible amount to us. But at the molecular level, it's a bustling metropolis of benzene rings. For one of those molecules to decompose, it needs a reason. A good reason.
Consider the energy barriers. It’s like trying to push a heavy boulder up a hill. You need a good push to get it over the top. For benzene, that push is a significant amount of energy. So, the grams that decompose are the ones that get that energy boost.
Maybe the real answer is that no grams must decompose. They decompose when conditions are right, when the energy is there, when the chemical forces align. It’s not a reluctant obligation; it’s a chemical opportunity.
And here’s where my personal, probably unpopular, opinion comes in. The number of grams of C6H6 liquid that must decompose is precisely the number that choose to. We can't force them. We can only provide the environment for their transformation.
It’s like asking how many stars must twinkle for the night sky to be beautiful. Is there a minimum number? Or is it the collective twinkle, the shared glow, that makes it so? I lean towards the latter.
So, next time you see a bottle of something that smells vaguely like your local gas station, don't fret about its potential decomposition. Just appreciate its molecular existence. And if any of it decides to break down, well, let it have its moment.
The question, “How many grams of C6H6 liquid must decompose?” ultimately leads us down a rabbit hole of chemical principles. But it also gives us a chance to ponder the nature of change, the role of energy, and the gentle, perhaps even willing, transformation of matter.
Ultimately, the answer isn't a number you'll find in a textbook. It's a feeling, a philosophical shrug, and a quiet understanding that some things just happen when they're ready. And that, my friends, is a perfectly valid way to look at it, even if it doesn't involve a calculator.
So, let's raise a metaphorical (and very safe!) beaker to the mysterious, the spontaneous, and the beautifully decomposing world of chemistry. The grams that decompose are simply the ones that embark on their next chemical adventure. And who are we to question their journey?
Perhaps the real answer is less about the quantity and more about the quality of the decomposition. Is it a clean break? A messy disintegration? The nuance is part of the fun. It’s the subtle poetry of molecular mechanics.
And in the grand scheme of things, the number of grams of C6H6 liquid that decompose is a tiny drop in the vast ocean of chemical reactions constantly occurring around us. It’s a reminder that the world is always in motion, always transforming, one molecule at a time.
So, to reiterate my bold (and likely unpopular) stance: the grams that must decompose are the ones that feel the call to change. No more, no less. It’s a chemical rebellion of sorts, a quiet declaration of independence from their molecular bonds.
And that, I think, is a wonderfully entertaining way to think about it. It injects a bit of personality into the inorganic world, doesn't it? Chemical entities with their own little desires and destinies.
The next time you ponder the decomposition of C6H6 liquid, just remember: it's not about a rigid requirement. It's about opportunity, energy, and the inherent drive of molecules to explore their potential. And that's something to smile about.
So, how many grams? The ones that are ready. It’s as simple, and as complex, as that. Let them decompose. Let them transform. It's their chemical journey, after all.