How Many Grams Are In 6.50 Moles Of H2so4

Ever stared at a recipe and wondered, "Just how much is a 'mole' anyway?" Yeah, me too. It sounds fancy, like something a wizard would whisper, but turns out, even us regular folks dabbling in a bit of science (or just trying to understand what's going on in the world) might bump into these "moles." Today, we're going to tackle a specific one: 6.50 moles of H₂SO₄. Sounds like a mouthful, right? But stick with me, and we'll break it down so it's as easy as figuring out how many cookies are in a dozen.
So, what in the heck is H₂SO₄? Don't let the subscripts and the uppercase letters scare you! That's just the secret code for sulfuric acid. Think of it as a really important chemical. It’s a bit like the baker's yeast in a bread recipe – it’s crucial for so many things, even if you don’t see it listed as the star ingredient in your final loaf. Sulfuric acid is used in making fertilizers, cleaning metals, and even in the batteries that power your car. Pretty important stuff, wouldn't you say?
Now, about "moles." Imagine you're at a party, and someone asks you to bring a "bunch" of balloons. What's a bunch? Is it 10? 20? It's a bit vague, right? Well, in science, they needed a way to be super precise. A mole is just a special number, a gigantic number, that chemists use to count atoms and molecules. It's like a chemist's "dozen," but instead of 12, a mole is approximately 602,200,000,000,000,000,000,000. Yep, that's a 6 followed by 23 zeros. Mind-boggling, I know!
Must Read
Think of it this way: If you had a mole of tiny, tiny LEGO bricks, you could build a tower that reaches the moon and back countless times. Or, if you had a mole of grains of sand, you'd have more sand than all the beaches on Earth combined! It's an astronomical number, designed to make dealing with incredibly small things a bit more manageable.
So, we have 6.50 moles of sulfuric acid. This means we have 6.50 times that gigantic number of sulfuric acid molecules. That’s a whole lot of H₂SO₄. But the question isn't how many molecules there are; it's how much mass all those molecules weigh. We want to know how many grams are in our 6.50 moles.
This is where the periodic table comes in. You know, that chart with all the elements that used to make us sweat in chemistry class? It’s actually incredibly useful! Each element on that table has something called an atomic mass. This atomic mass tells us, roughly, how much one mole of that specific element weighs in grams. It’s like a price tag for each type of atom.

For sulfuric acid (H₂SO₄), we need to add up the atomic masses of all the atoms in its formula. Let’s break down H₂SO₄:
Hydrogen (H):
We have two hydrogen atoms (that’s what the little ‘2’ after H means). Hydrogen’s atomic mass is about 1.01 grams per mole. So, for our two hydrogens, we have 2 * 1.01 = 2.02 grams.
Sulfur (S):
We have one sulfur atom. Sulfur’s atomic mass is about 32.07 grams per mole. Easy peasy, just 32.07 grams.

Oxygen (O):
And we have four oxygen atoms (that’s the ‘4’ after O). Oxygen’s atomic mass is about 16.00 grams per mole. So, for our four oxygens, we have 4 * 16.00 = 64.00 grams.
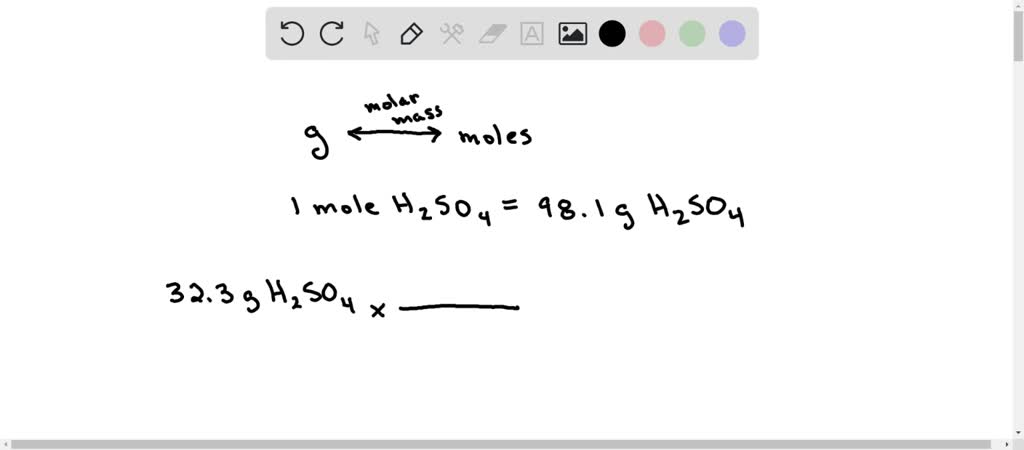
Now, we just add these all up to get the molar mass of sulfuric acid. This is the weight of one mole of H₂SO₄. 2.02 (Hydrogen) + 32.07 (Sulfur) + 64.00 (Oxygen) = 98.09 grams per mole.
Think of it like this: If you were going to bake a cake that specifically needed 1 mole of flour, and you knew 1 mole of flour weighed 98.09 grams, you'd measure out 98.09 grams. Simple, right? The molar mass is our conversion factor, our bridge between the abstract idea of "moles" and the tangible measurement of "grams."
So, we know that one mole of sulfuric acid weighs 98.09 grams. Our question is about 6.50 moles. This is where a little bit of multiplication comes in. It’s like if you knew one fancy artisanal chocolate bar cost $3, and you wanted to buy 6.50 of them. You’d just multiply the price of one by the number you want, right?

In our case, we multiply the weight of one mole by the number of moles we have:
6.50 moles * 98.09 grams/mole = 637.585 grams.
And there you have it! 6.50 moles of H₂SO₄ is equal to approximately 637.59 grams (we can round it a bit for everyday talk). That’s roughly the weight of a medium-sized bag of sugar, or about three big cans of soda. That's a decent amount of sulfuric acid!

Why should you care about this? Well, understanding these conversions is like having a secret superpower in the world of science. It helps scientists and engineers to accurately measure out chemicals for everything from developing new medicines to creating the materials for your smartphone. It ensures that reactions happen correctly, that products are safe, and that experiments are repeatable.
It’s also about appreciating the tiny, invisible world that makes up everything around us. When you understand that a "mole" is just a way to count these minuscule building blocks, and that you can figure out their weight, you start to see the universe in a slightly different, and perhaps more magical, light. It’s the difference between just seeing a finished product and understanding a little bit about how it was made, from its most fundamental parts.
So, the next time you hear about moles or chemical formulas, don't panic! Remember our baker’s yeast analogy, think about the chemists’ gigantic dozen, and know that with a little help from the periodic table and some basic math, you can unlock the secrets of how much of anything there really is. It's all about finding those handy conversion factors, those everyday equivalences, that turn abstract numbers into things we can actually imagine holding in our hands.
It's a reminder that even the most complex-sounding scientific concepts can often be broken down into relatable steps, much like figuring out how many eggs you need for a double batch of cookies. And who doesn't love cookies? So, there you have it – a little bit of chemistry, a little bit of math, and a whole lot of understanding about the building blocks of our world. And it all started with a question about 6.50 moles of H₂SO₄. Pretty neat, huh?