How Many Electrons In An Atom Could Have N 2

Hey there, fellow travelers on this wild cosmic ride! Ever catch yourself staring up at the stars, or maybe just at your phone screen, and wondering about the tiny, invisible building blocks that make up everything? Yeah, me too. It’s like the ultimate reality show, happening constantly, right under our noses. Today, we’re diving into the electrifying world of atoms, and specifically, we’re going to unravel a little mystery: How many electrons in an atom could have N=2?
Now, before your brain starts doing that sci-fi movie intro thing, let’s take a deep breath. This isn’t about rocket science degrees or needing a lab coat. Think of it more like understanding the basic rules of the universe’s coolest game. We're talking about electrons, those zippy little guys that orbit the atomic nucleus like tiny planets around a sun. And that ‘N=2’ we’re throwing around? That’s just a fancy way of talking about their energy level, their… vibe, if you will.
Imagine an atom as a tiny apartment building. The nucleus is the lobby, and the electrons are the residents living on different floors. Each floor represents a specific energy level, and the higher the floor, the more energy the resident has. Electrons aren’t just randomly scattered; they’re organized, like tenants paying rent and sticking to the rules.
Must Read
So, when we talk about N=2, we’re essentially asking: how many electrons can crash on the second floor of this atomic apartment building?
The Quantum Leap of Understanding
This whole idea comes from the fascinating world of quantum mechanics. Don’t let the word intimidate you; it’s just the set of rules that govern the super-duper tiny stuff. In this realm, things don’t behave like billiard balls; they’re a bit more… fuzzy and probabilistic. Electrons exist in specific energy levels, also known as shells or orbitals. These are like designated zones where an electron is most likely to be found.
Think of it like a concert venue. You have different seating sections – the general admission floor, the lower boxes, the premium VIP seats. Each section has its own price (energy) and capacity. Electrons, in their atomic homes, are similar. They occupy specific energy levels, and each level can only hold a certain number of them.
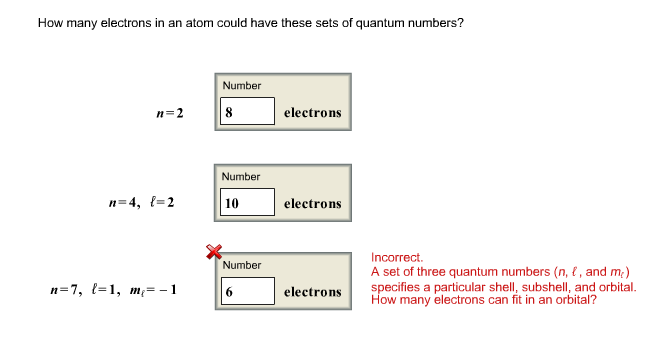
The first energy level, N=1, is like the ground floor. It’s the closest to the nucleus (the ‘lobby’) and therefore the most stable. It’s also the most exclusive, only able to house a maximum of two electrons. Think of it as a cozy studio apartment – only room for two.
Level Up: Entering the N=2 Zone
Now, we ascend to the second energy level, N=2. This is where things get a bit more spacious. The second energy level has sub-levels, kind of like different types of apartments within a floor. These sub-levels are called orbitals.
For N=2, there are actually two sub-levels: the 2s orbital and the 2p orbitals. The 2s orbital is like a single, comfortably sized studio on the second floor. The 2p orbitals? They’re like a suite with multiple rooms. Specifically, there are three 2p orbitals.
Here’s where the magic happens, and where the number ‘N=2’ really gets its meaning in terms of electron capacity. Each orbital, regardless of whether it’s s, p, or d, can hold a maximum of two electrons. This is a fundamental rule in quantum mechanics, often referred to as the Pauli Exclusion Principle. It’s like a strict housing code: no more than two residents per room, and they must have opposite ‘spins’ – a quantum property that’s a bit like them facing opposite directions.
Crunching the Numbers for N=2
So, let’s add it up for our N=2 energy level:

- The 2s orbital can hold 2 electrons.
- The three 2p orbitals can each hold 2 electrons, so that’s 3 x 2 = 6 electrons.
Therefore, the total number of electrons that can occupy the second energy level (N=2) is the sum of electrons in its sub-levels: 2 (from 2s) + 6 (from 2p) = 8 electrons.
So, to answer our burning question: In an atom, the second energy level (N=2) can accommodate a maximum of eight electrons. It's like the second floor of our atomic apartment building can host up to eight residents comfortably.
Beyond the Numbers: The Dance of Electrons
This isn't just an abstract number. The way electrons fill these energy levels and orbitals dictates an element’s chemical properties. It’s like the residents on different floors of the apartment building having different personalities and social circles. Elements with full outer shells are super stable and tend not to react much – they’re the contented, solitary types. Elements with nearly full outer shells are eager to bond and share electrons to achieve that stability – they’re the social butterflies.
Think about it like dating in the digital age. We have profiles (electron configurations), we make connections (chemical bonds), and we aim for a sense of balance and satisfaction (stability). The number of electrons in the outer shells is the key to an element’s reactivity, just like a person’s social skills and availability might influence their dating prospects.

For instance, Neon (atomic number 10) has its electrons perfectly filling up to the N=2 shell. It has 2 electrons in N=1 and 8 electrons in N=2. Because its outer shell (N=2) is full, Neon is a noble gas, meaning it’s incredibly unreactive. It’s the atom equivalent of someone perfectly happy chilling on their own, not needing to interact with anyone else. Very Zen.
Contrast this with Oxygen (atomic number 8). Oxygen has 2 electrons in N=1 and 6 electrons in N=2. It has space for two more electrons in its N=2 shell to reach that magical number of eight. This desire to fill its outer shell is why oxygen is so reactive, readily bonding with other elements, including forming water (H₂O) with hydrogen. It’s like oxygen is always looking for a plus-two to complete its perfect apartment.
Fun Facts to Spark Your Curiosity
- The concept of electrons having specific energy levels was a radical idea when proposed by Niels Bohr. Before that, scientists thought electrons might spiral into the nucleus, causing atoms to collapse! Thankfully, quantum mechanics saved the day.
- The 'spin' of an electron, often visualized as spinning clockwise or counterclockwise, is a purely quantum mechanical property. It doesn't mean the electron is literally spinning like a top. It's more like an intrinsic angular momentum. Think of it as a built-in barcode that allows two electrons to share an orbital.
- The arrangement of electrons in orbitals is described by quantum numbers. N is the principal quantum number (energy level). There are also azimuthal (l), magnetic (ml), and spin (ms) quantum numbers, each adding another layer of detail to an electron's identity. It’s like an electron has a full social security number!
- Elements in the same column of the periodic table (groups) often have similar chemical properties because they have the same number of valence electrons – the electrons in their outermost energy level. This is why the periodic table is such a powerful organizational tool, a chemist's dream Pinterest board.
A Practical Tip for the Atomically Curious
If you're ever trying to visualize this, grab some LEGOs or different colored beads. Use one color for the nucleus, then different colors or sizes for each energy level. You can even try to represent the orbitals within those levels. It’s a fun, hands-on way to grasp these abstract concepts, and a great activity to do with kids (or just to unleash your inner child!).
Think about the periodic table as a map. Each element is a destination, and its position tells you a lot about its electronic makeup and how it might interact with others. Understanding the electron shells is like learning the basic language of this map.

From Atoms to Everyday Life
So, why should you care about how many electrons can fit on the N=2 level? Because this fundamental principle underpins so much of our modern world. The way atoms bond dictates the properties of materials we use every day – from the plastic in your phone case to the metal in your car, to the molecules in the food you eat and the air you breathe.
Every chemical reaction, every biological process, is a complex ballet of electrons moving between atoms, seeking stability. When you see vibrant colors, taste delicious flavors, or feel the warmth of the sun, you’re witnessing the consequences of these intricate atomic interactions. It’s a constant, unseen performance of nature’s most basic laws.
Even something as simple as plugging in your phone is a testament to our understanding of how electrons behave. The flow of electricity, the very lifeblood of our digital existence, is all about the movement of these tiny, charged particles. It’s a reminder that even the most advanced technology is built on the foundations of tiny, energetic dancers in their atomic homes.
So next time you’re marveling at the intricate design of a snowflake, or the vibrant hues of a sunset, take a moment to appreciate the silent, unseen dance of electrons. They’re the unsung heroes, the fundamental rhythm section of our universe, and understanding their little world, even just a little bit, makes our own world feel a whole lot more magical.