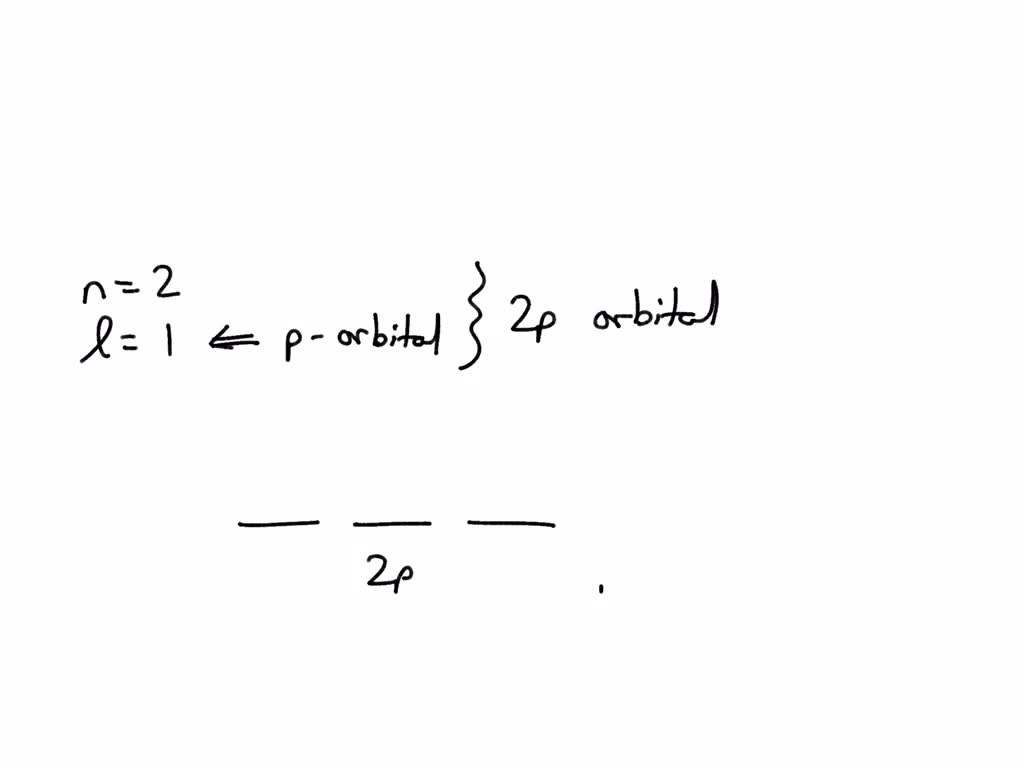How Many Electrons Can Fit Into An Orbital

Hey there, science explorer! Ever wonder about the teeny-tiny universe inside atoms? It's a wild place, full of whizzing particles. Today, we're zooming in on one of its most fundamental building blocks: the electron. And the burning question on everyone's lips (well, maybe just mine, but you're curious now, aren't you?) is: how many of these little guys can cram into a spot called an orbital? Get ready for some electron-tastic fun!
So, first things first, what exactly is an orbital? Think of it less like a perfectly defined parking spot and more like a fuzzy, three-dimensional probability cloud. It’s the region around the atom’s nucleus where you’re most likely to find an electron. It's not like a planet orbiting the sun, where you know its path. Electrons are more… elusive. They're kind of everywhere in that cloud, all at once, until you try to pinpoint them. Spooky, right? But also, pretty cool.
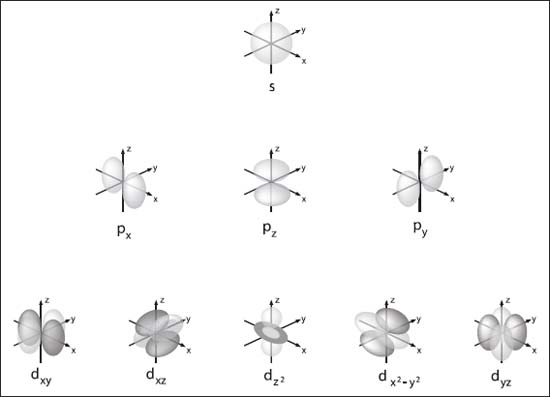
Imagine you’re at a party, and the electron is the super-energetic guest who just can't stand still. The orbital is the dance floor. They're definitely on the dance floor, but good luck telling me exactly where they are at any given millisecond. Scientists figured out how to describe these dance floors, giving them shapes and sizes. We've got the simple, spherical ones called 's' orbitals, the dumbbell-shaped ones called 'p' orbitals, and then things get even crazier with 'd' and 'f' orbitals. Think of them as different kinds of dance moves – some are basic, some are show-stopping.
Must Read
Now, the big question: how many electrons can we invite to the party on a single orbital dance floor? Brace yourselves, because the answer is surprisingly simple and, frankly, a little restrictive. An orbital can hold a maximum of two electrons. Yep, just two. That’s it. No mosh pits, no huge electron rave. Just a cozy duo.
Why only two? Well, it all boils down to some really fundamental rules of quantum mechanics. These rules are like the bouncers at our electron party, ensuring everything stays orderly. The most important rule here is called the Pauli Exclusion Principle. It sounds fancy, I know, but it’s actually quite logical. It basically says that no two electrons in an atom can be in the exact same quantum state.
What does "quantum state" even mean? Good question! Think of it as the electron's unique ID card. This ID card has a few pieces of information, like its energy level and its "spin." Now, electrons have this property called spin, which is a bit like an intrinsic angular momentum. You can picture it as the electron spinning on its own axis, either clockwise or counter-clockwise. For our party analogy, it's like each electron has to wear a different colored hat – one red, one blue. You can't have two guests wearing the same color hat in the same dance floor spot.

So, if an orbital is our dance floor, and two electrons can be there, how do they avoid being in the exact same quantum state? They do it by having opposite spins! One electron will have its spin "up" (let's say the red hat), and the other will have its spin "down" (the blue hat). This tiny difference in spin is enough for them to be considered in different quantum states, allowing them to share the same orbital.
It's like a perfectly matched pair, each with their own little quirk that makes them unique, yet they can still vibe together in the same space. They're not exactly identical twins; they're more like fraternal twins who are still super close. This pairing up is actually quite stable and energetically favorable for atoms. They like having their electrons neatly organized, with pairs filling up orbitals before moving on to the next one.
Think about it this way: if an orbital could hold, say, five electrons, imagine the chaos! They'd be bumping into each other, repelling each other with their negative charges, and generally making a mess of things. Nature, thankfully, prefers order. The Pauli Exclusion Principle is the universe’s way of saying, "Let's keep things tidy, shall we?"

This "two electrons per orbital" rule is a cornerstone of chemistry. It dictates how atoms bond together to form molecules. It's why we have the periodic table organized the way it is. Every single chemical property you can think of, from why water is wet to why gold is shiny, can ultimately be traced back to how these electrons are arranged in their orbitals, following that strict "max two" rule.
Let’s dive a little deeper into those orbital shapes, just for fun. 's' orbitals are the simplest. They're spheres, like tiny little bubblegum balls around the nucleus. You can fit a maximum of two electrons in an 's' orbital. Easy peasy. Then you have the 'p' orbitals. These are shaped like dumbbells, with two lobes on either side of the nucleus. For any given energy level, there are actually three of these 'p' orbitals oriented along different axes (x, y, and z). Since each 'p' orbital can hold two electrons, you can have a total of 3 x 2 = 6 electrons in the 'p' subshell.
After 'p' come the 'd' orbitals. These have more complex shapes, often described as cloverleafs, plus one that looks like a dumbbell with a donut around the middle. There are five 'd' orbitals in each subshell, so that's a potential 5 x 2 = 10 electrons. And then we have the 'f' orbitals, which are even more convoluted and there are seven of them, holding a whopping 7 x 2 = 14 electrons!

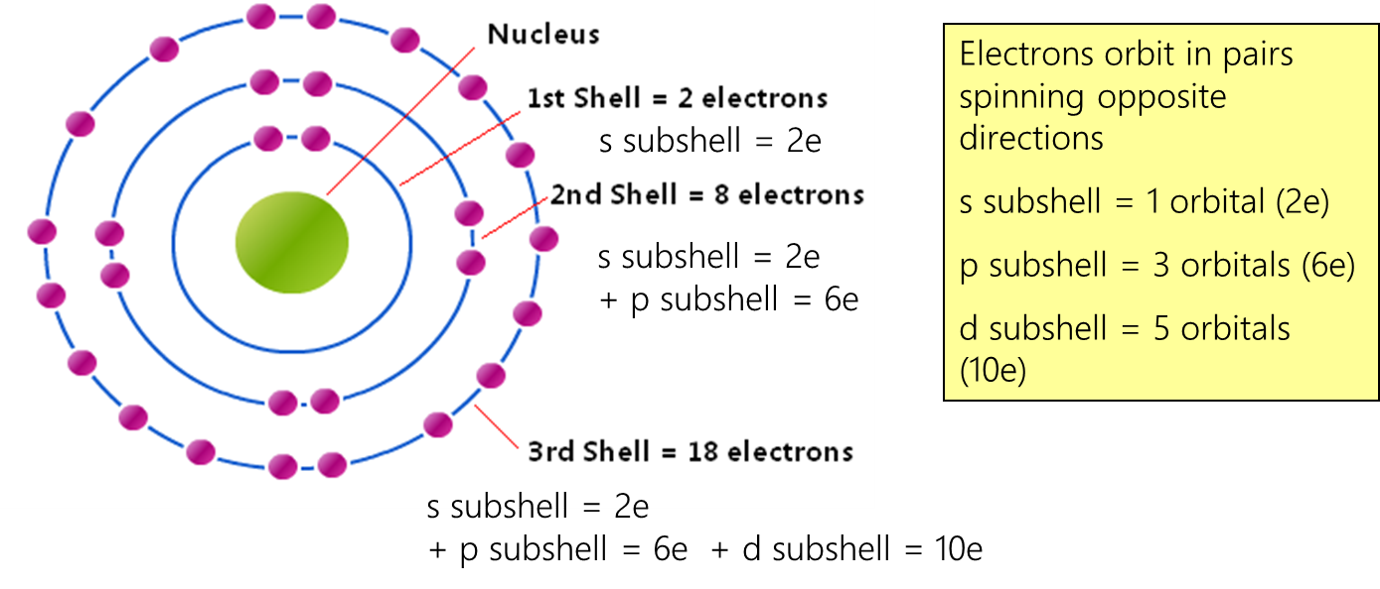
So, while a single orbital is a cozy spot for just two electrons, atoms have many different orbitals at different energy levels. These orbitals are grouped into something called subshells (like the 's' subshell, 'p' subshell, etc.), and then these subshells are grouped into electron shells. It's like a set of Russian nesting dolls, each layer containing smaller ones, all contributing to the atom's overall electron configuration.
The arrangement of electrons in these shells and subshells is what gives each element its unique identity. For instance, hydrogen, the simplest atom, has just one electron. It sits happily in the lowest energy 's' orbital. Helium has two electrons, and they both pair up in that same 's' orbital, with opposite spins. They're like the ultimate electron power couple!
When we get to lithium, the third element, things get interesting. We've already filled that first 's' orbital with our two helium electrons. So, lithium's third electron has to go to the next available energy level, in a new 's' orbital. It's like the party is getting bigger, and we need a new dance floor!
This filling of orbitals, strictly adhering to the "max two electrons with opposite spins per orbital" rule, is the fundamental basis for understanding chemical behavior. When atoms interact to form bonds, they're essentially trying to achieve a more stable electron configuration, often by filling up their outermost orbitals or by sharing electrons.
It’s a beautiful, elegant system, isn't it? This simple rule – two electrons per orbital, each with opposite spin – governs the vastness of chemical diversity we see in the universe. From the air we breathe to the stars we gaze at, it all comes down to these tiny, energetic particles dancing in their probabilistic clouds.
So, next time you look at anything around you, remember the intricate dance happening at the atomic level. Remember the cozy orbitals holding their paired-up electrons, obeying the universe’s gentle but firm rules. It's a testament to the incredible order and complexity that can arise from such fundamental principles. And even though we can't see them, these electrons are the unsung heroes of our reality, making everything possible. Pretty amazing, right? Keep exploring, keep wondering, and let that curiosity spark your own inner glow!