How Many Electrons Can Each P Orbital Hold
Ever found yourself staring at a blank canvas, a dusty sketchbook, or even just a spare piece of paper, and felt that familiar creative itch? That desire to bring something new into the world, to express a thought, a feeling, or a fantastical scene? Well, guess what? You might be closer to unlocking your inner artist than you think, and the secret weapon we're talking about today isn't a fancy brush or a expensive pigment. It's a fundamental concept from the world of chemistry: how many electrons can each p orbital hold. Now, before you picture complicated molecular diagrams, let's demystify this. It's actually a surprisingly accessible and inspiring idea that can spark some serious creative fireworks!
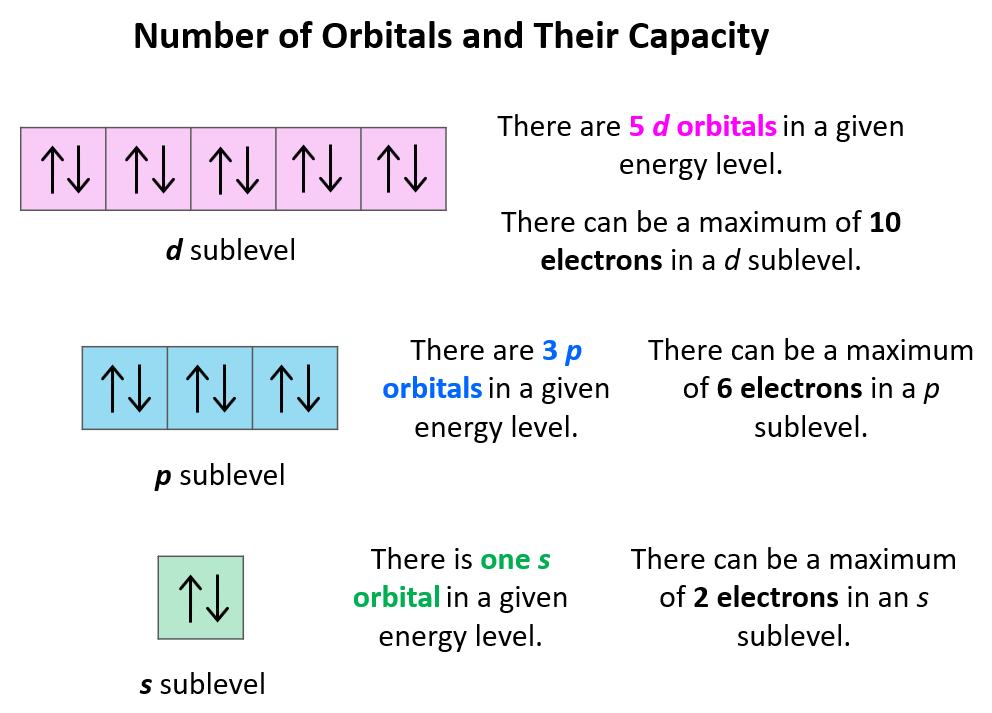
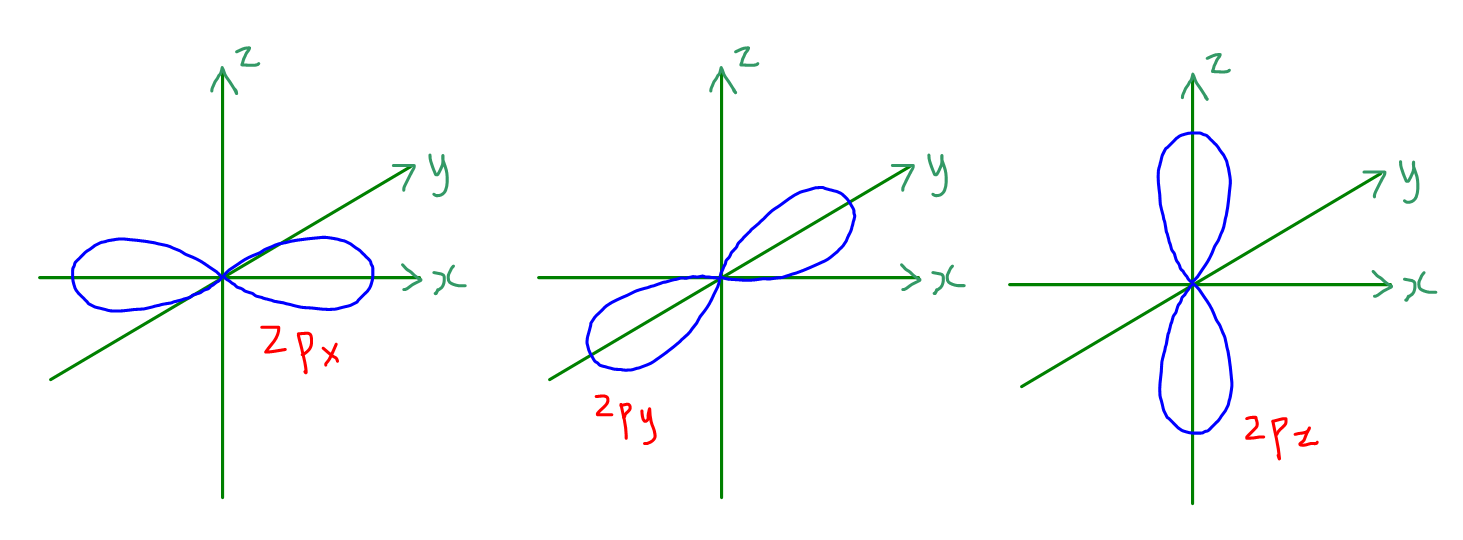
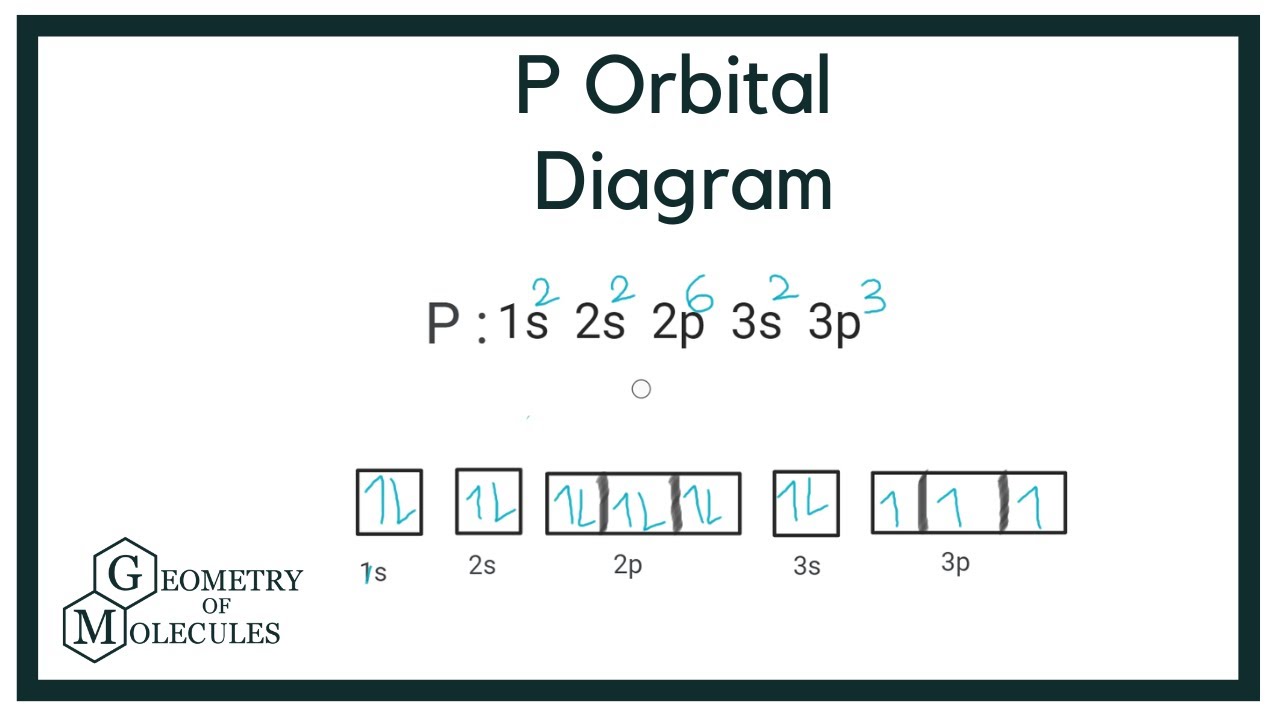
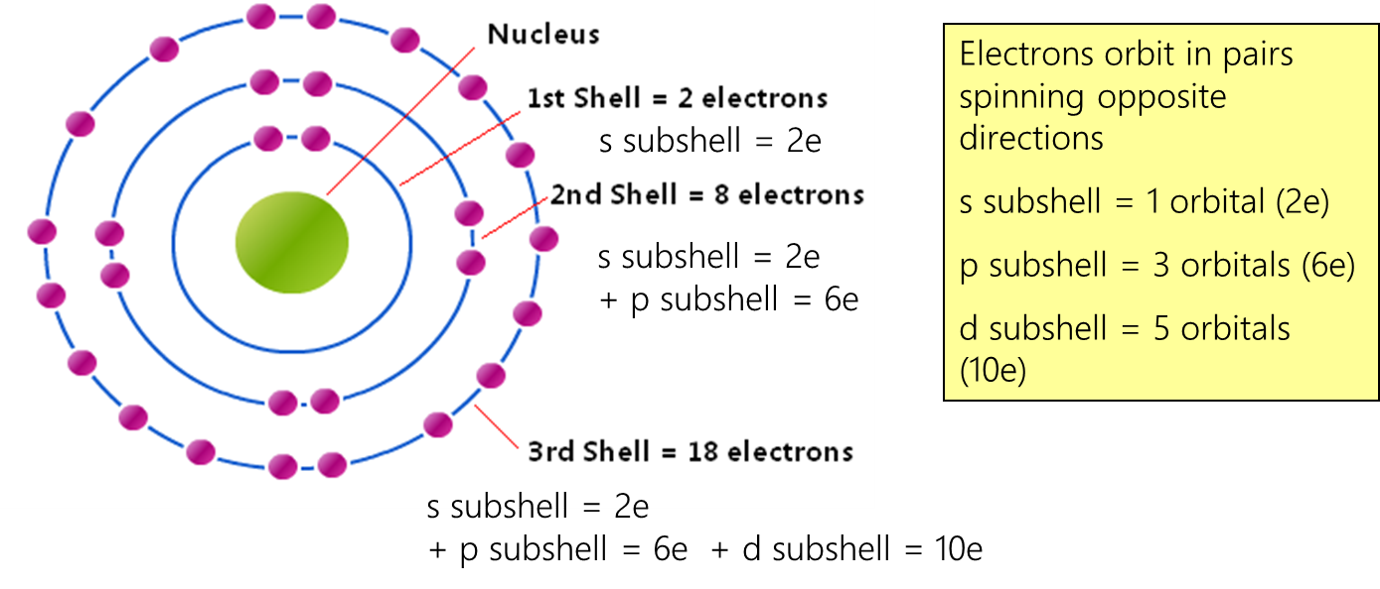
You might be wondering, "How can a science concept help my art?" The beauty of this lies in its simplicity and its structural elegance. Think of atomic orbitals as tiny, invisible containers within atoms. The p orbitals, in particular, have a distinctive dumbbell shape. Now, here's the key: each individual p orbital can hold a maximum of two electrons. That's it! Two. This fundamental rule of quantum mechanics, known as the Pauli Exclusion Principle, isn't just for scientists; it's a fantastic little nugget for anyone looking to build, create, or even just understand patterns. For artists and hobbyists, this concept offers a framework for understanding balance, composition, and limiting factors. For casual learners, it’s a gateway to appreciating the underlying order of the universe in a way that’s both intellectually stimulating and visually relatable.
Imagine using this idea in your creative endeavors. For a visual artist, you could explore themes of duality or pairing. Think of two figures in a painting, perfectly balanced, representing those two electrons. Or perhaps you could create abstract compositions based on the symmetry of the p orbital shape itself. For a writer, this concept can inspire character dynamics: a pair of opposing characters, or two halves of a whole. Even for a crafter, you could design patterns with pairs of elements, or experiment with how two identical objects interact. Think of pottery with twin handles, or a quilt block featuring two contrasting colors. The possibilities are as vast as your imagination!
Must Read
Ready to give it a try? It’s easier than you think! Start small. Grab a pencil and paper. Try sketching the basic dumbbell shape of a p orbital. Now, within that shape, draw two simple dots, representing the electrons. Then, let your mind wander. What happens if you duplicate that shape? What if you arrange them in different orientations? You could create repeating patterns, explore negative space, or even try to anthropomorphize the shapes. Don't be afraid to experiment with different mediums too – colored pencils, digital drawing software, or even collage!
Ultimately, the enjoyment comes from finding order and inspiration in the fundamental building blocks of the universe. It’s about recognizing that even the most complex creations are built upon simple, elegant rules. The fact that each p orbital can hold precisely two electrons is a tiny piece of that puzzle, but it’s a piece that can unlock a world of creative potential. So, next time you’re seeking inspiration, remember the humble p orbital and its capacity for two. It might just be the spark you need!