How Do You Calculate The Rate Of Photosynthesis

Picture this: I'm a kid, maybe seven or eight, and my grandma, bless her green thumb, has this huge tomato plant in her garden. It's practically a monster, sprawling everywhere, and absolutely dripping with the most delicious tomatoes I'd ever tasted. I remember asking her, totally serious, "Grandma, how does it grow so much?" She just smiled, winked, and said, "Oh, it's a little bit of sunshine, a little bit of water, and a whole lot of magic."
Magic, huh? Well, as I got older, I realized Grandma's "magic" had a name: photosynthesis. And it's not just about tomatoes, is it? It's about everything green and growing. Trees, grass, that little succulent you're probably trying not to kill on your windowsill – they're all doing this incredible thing, turning sunlight into food. But how, exactly, do we put a number on that whole process? How do we calculate the rate of photosynthesis? That's what we're diving into today, no magic required, just a bit of scientific sleuthing.
So, What Exactly IS Photosynthesis? (A Quick Refresher, Don't Worry)
Okay, deep breaths. We're not going back to high school biology exam cram sessions. Photosynthesis, in its simplest, most delicious form, is how plants make their own food. They take in carbon dioxide (CO2) from the air, water (H2O) from the soil, and use energy from sunlight to create glucose (sugar – the plant's fuel!) and oxygen (O2 – the stuff we breathe!). Pretty neat, right?
Must Read
Think of it like a tiny solar-powered factory inside every leaf. The chlorophyll, that green pigment we all associate with plants, is like the solar panel, capturing the sunlight's energy. Then, in a series of super-complex reactions (which we are definitely not going into today, unless you're a biochemist, in which case, hi!), the plant whips up its sugary meals.
But here's the kicker: this isn't a static process. It's happening constantly, and its speed can change. That's where calculating the rate comes in. It's like asking, "How fast is that factory churning out those sugary snacks?"
Why Bother Calculating the Rate?
Honestly, it sounds a bit niche, doesn't it? Like, who wakes up in the morning thinking, "I need to know the precise rate of photosynthesis in my petunias"? Well, scientists, farmers, and even people trying to figure out how to grow plants in space (yes, that's a thing!) are very interested in this. Understanding the rate helps us:
- Optimize Crop Yields: If you can figure out what conditions make a plant photosynthesize faster, you can grow more food. Simple as that. Farmers are always tweaking things – light, water, fertilizer – to get the best harvest.
- Understand Plant Health: A slow rate might mean the plant is stressed, diseased, or not getting what it needs.
- Study Climate Change: Plants are massive carbon sinks, absorbing CO2. Knowing how fast they do it helps us model and understand how our changing planet affects them, and vice-versa.
- Develop New Technologies: Think artificial photosynthesis – creating fuel or chemicals using sunlight. You have to know the rate to make that work!
So, it's not just about Grandma's tomatoes anymore. It's about feeding the world, understanding our environment, and maybe even powering our future. Pretty important stuff!
The Nitty-Gritty: How Do We Actually Measure It?
Alright, so how do we go from Grandma's magic to actual, measurable numbers? Well, there are a few ways, and they all hinge on measuring the inputs or the outputs of photosynthesis.
Remember our equation?
CO2 + H2O + Sunlight → Glucose + O2
We can either measure how fast the plant is sucking up CO2 or spitting out O2, or sometimes, indirectly, how fast it's making that glucose. The most common methods focus on gases because they're a bit easier to track.

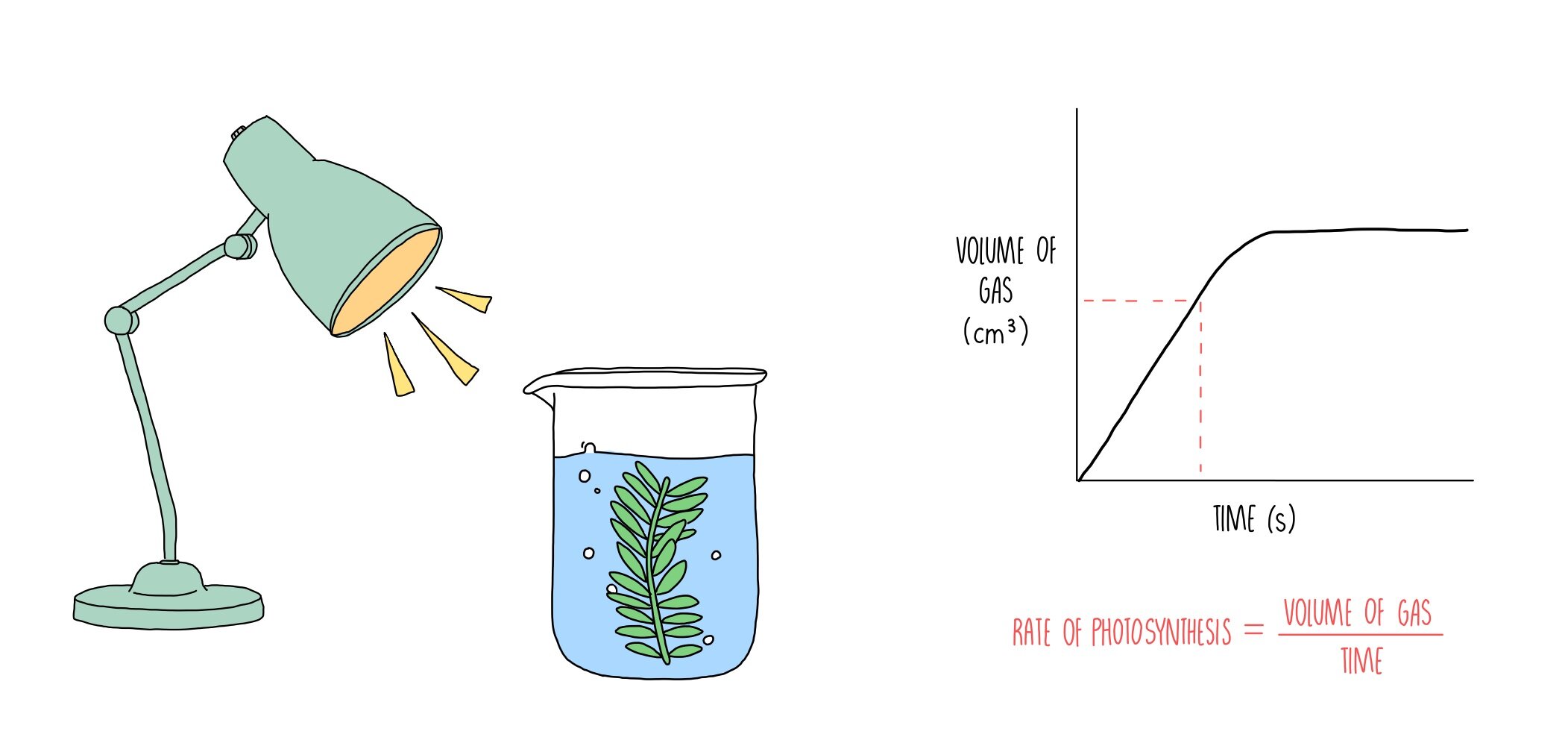
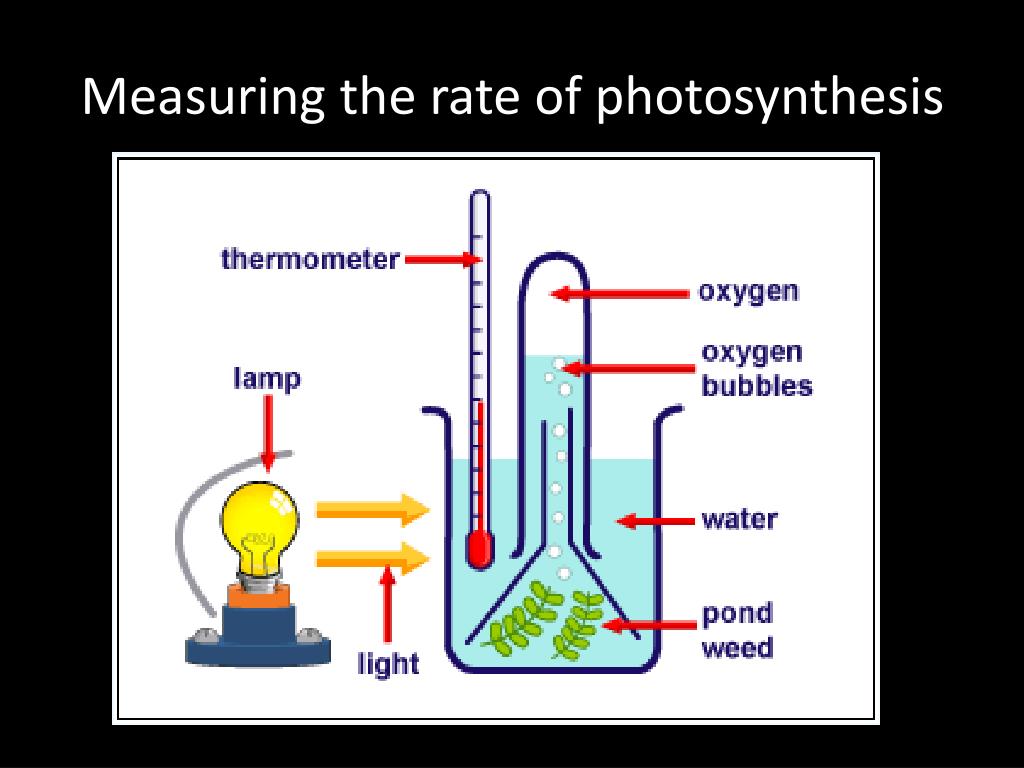
Method 1: Measuring Oxygen Production
This is a classic. Since oxygen is a product of photosynthesis, if we see the amount of oxygen increasing, we know the plant is busy doing its thing. Here’s the general idea:
Imagine you have a plant, maybe a sprig of Elodea (that common aquarium plant that seems to thrive on neglect – you know the one!). You put it in a sealed container filled with water. You expose it to light, and you’ve got this little setup that can measure the oxygen bubbling out. Or, more precisely, you can measure the change in oxygen concentration in the air above the water over a specific time.
What you need:
- A plant (Elodea is a good beginner choice)
- A sealed container (like a flask or a bell jar)
- A light source
- A way to measure oxygen concentration (this can be fancy sensors or even more basic chemical tests, though sensors are way more common for rate calculations).
- A timer, obviously!
The Process (Simplified):
- Get your plant sample ready in its container with water.
- Ensure the container is sealed to trap the gases.
- Expose it to a controlled light source.
- Measure the initial oxygen concentration.
- Leave it for a set period (say, 30 minutes).
- Measure the final oxygen concentration.
- Calculate the difference.
So, if you started with 20% oxygen and ended up with 20.5% oxygen after 30 minutes, your plant produced 0.5% more oxygen in that time. To get the rate, you'd then divide this amount of oxygen produced by the time taken. You might express it as "% oxygen per hour" or something similar.
The tricky bits:
- Respiration: Uh oh. Plants don't only photosynthesize. They also respire, meaning they use up oxygen to break down sugars for energy. This happens all the time, day and night. So, the oxygen you measure is actually the net oxygen production (photosynthesis minus respiration). To get the gross photosynthesis rate (how much was actually made), you'd ideally measure oxygen production in the light and then oxygen consumption in the dark (during respiration) and do some math. It’s like trying to track your bank account: you see the final balance, but you need to know how much came in and how much went out to truly understand the flow.
- CO2 levels: Plants need CO2 to photosynthesize. If you're in a closed system, they might run out of CO2, which would slow down the rate even if there's plenty of light. So, sometimes you have to ensure a constant supply of CO2.
- Temperature: Enzymes involved in photosynthesis are sensitive to temperature. Too hot or too cold, and the rate drops.
Method 2: Measuring Carbon Dioxide Uptake
This is the flip side of the coin. If oxygen is a product, then CO2 is a reactant. So, if the plant is photosynthesizing, it's sucking up CO2 from its surroundings. We can measure this decrease.
The setup is often similar: a sealed environment with the plant. But instead of measuring oxygen, we're measuring the decrease in CO2 concentration.
What you need:
- A plant
- A sealed container
- A light source
- A way to measure CO2 concentration (fancy sensors are key here – they're called NDIR sensors, Non-Dispersive Infrared, if you want to impress your friends).
- A timer.
The Process (Simplified):
- Place the plant in the sealed container.
- Expose it to light.
- Measure the initial CO2 concentration.
- Wait for a set period.
- Measure the final CO2 concentration.
- Calculate the difference (the amount of CO2 consumed).
If you started with, say, 400 parts per million (ppm) of CO2 and ended with 380 ppm after an hour, your plant took up 20 ppm of CO2. The rate would then be 20 ppm per hour. This is often expressed in units like micromoles of CO2 per square meter per second (µmol CO2 m⁻² s⁻¹), which sounds super scientific and is the standard for serious research.
The tricky bits:
- Respiration again! Just like with oxygen, plants also respire and release CO2. So, again, you're measuring the net CO2 uptake. To get gross photosynthesis, you'd need to account for respiration by measuring CO2 release in the dark.
- Water vapor: Plants transpire, releasing water vapor. This can sometimes interfere with CO2 measurements, so it needs to be managed.
- Light intensity and CO2 levels are still king: Just like before, these factors heavily influence the rate.
Method 3: Measuring Biomass Increase (The Slow and Steady Approach)
This is a much less immediate way to gauge photosynthesis, but it's fundamental. Photosynthesis produces glucose, which the plant uses to build its tissues – its leaves, stems, roots, everything. So, if a plant is growing and gaining weight (biomass), it's a sign of photosynthesis happening over time.
The Process (Very Simplified):
- Take a plant (or a population of plants) and measure its initial dry weight.
- Grow it under controlled conditions for a significant period (weeks, months).
- Measure its final dry weight.
- The difference in weight represents the accumulated biomass, a portion of which came from photosynthesis.
The tricky bits:

- Slow: This method takes a loooong time. You can't get an hourly rate this way.
- Multiple factors: Biomass increase isn't just photosynthesis. It also involves nutrient uptake, water, and respiration. It's a cumulative effect, not an instantaneous rate.
- Drying: You need to dry the plant material to get a true weight, which destroys the sample.
This method is more for understanding long-term growth and productivity rather than moment-to-moment rates.
Method 4: Using Specialized Equipment (The High-Tech Way)
For serious research, scientists use fancy gadgets called photosynthesis measurement systems or leaf porometers. These are often clamp-like devices that attach to a leaf.
They create a small, controlled environment around the leaf and measure gas exchange (CO2 and H2O) in real-time. They can precisely control light intensity, temperature, and humidity within that little chamber.
This is the gold standard for getting accurate, instantaneous rates under specific conditions. You can literally move from leaf to leaf and get readings. It's like having a mini, portable photosynthesis lab!
Putting It All Together: The "Rate" Part
So, we've measured changes in oxygen, CO2, or biomass. How do we get to a "rate"? It's all about amount per unit of time. No matter what you're measuring (oxygen, CO2, or even sugar if you could measure it directly!), you divide that amount by the time over which the change occurred.
For example, if a leaf sample consumed 10 micromoles (µmol) of CO2 in 10 minutes, the rate is:
10 µmol / 10 minutes = 1 µmol/minute
If you want to be really precise, you'd usually normalize this to the leaf's surface area (so you're comparing apples to apples, not a big leaf to a small one) or the plant's dry weight. So, the rate might be expressed as:
1 µmol CO2 / cm² / minute (CO2 uptake per square centimeter of leaf area per minute)
Or:
5 µmol O2 / g leaf dry weight / hour (Oxygen production per gram of leaf dry weight per hour)
These units tell you how efficient the plant is at converting light and CO2 into energy and biomass.
Factors That MESS With the Rate
It’s never just one thing, is it? The rate of photosynthesis is influenced by a whole bunch of environmental factors:
- Light Intensity: More light generally means a faster rate, but only up to a point. Too much light can actually damage the photosynthetic machinery (photoinhibition). It's like trying to charge your phone with a lightning bolt – not effective!
- Carbon Dioxide Concentration: Higher CO2 levels usually lead to higher rates, but again, there's a saturation point.
- Temperature: Photosynthesis relies on enzymes, and enzymes have optimal temperature ranges. Too cold, and they slow down; too hot, and they can denature (basically, fall apart).
- Water Availability: Plants need water, not just as a reactant, but also to keep their stomata (tiny pores on leaves that let CO2 in and O2 out) open. If they're dry, they'll close their stomata to conserve water, which also stops CO2 from getting in, thus slowing photosynthesis.
- Nutrient Availability: Plants need nutrients like nitrogen and magnesium to build chlorophyll and enzymes. A lack of these can limit photosynthesis.
So, when we calculate the rate, we're usually doing it under specific, controlled conditions to see how one factor (like light intensity) affects the rate, or we're measuring it in a natural setting where all these factors are interacting. It's a constant dance between the plant and its environment.
Wrapping It Up (Without Getting Too Technical!)
Calculating the rate of photosynthesis isn't rocket science, but it does require a bit of careful observation and measurement. Whether you're using simple bubbling experiments with aquatic plants to see oxygen production or sophisticated sensors measuring gas exchange on a leaf, the core idea is to quantify how quickly a plant is converting light energy into chemical energy.
It's about understanding those tiny factories within the leaves, working tirelessly to feed the plant and, in turn, support life on Earth. So, next time you see a lush green plant, remember Grandma’s magic and all the science that goes into understanding just how much life-giving work it’s doing. It's a beautiful, intricate process, and figuring out its speed is pretty cool, if you ask me!


