How Can Metals Be Placed In Order Of Their Reactivity

So, picture this: I'm about twelve, and my grandad, bless his heart, decides it's high time I learned a thing or two about science. He's got this shed that smells vaguely of old oil and forgotten dreams, and in it, he has all these bits of metal lying around. Copper pipes, rusty iron nails, some shiny bits of aluminum foil I probably used for a school project. He pulls out this jar of clear liquid and says, "Right, let's see which of these lads is the loudest."
Now, to my young mind, "loudest" meant something dramatic happening. He drops a piece of iron into the liquid, and nothing. Then, he drops in a piece of zinc, and poof! Tiny bubbles start to appear, like the metal was trying to whisper secrets. Then he drops in some magnesium, and it's a bit more vigorous, a proper fizzy party. I was utterly captivated. It was like a tiny, contained volcano erupting, all thanks to some random metals and a mysterious liquid. Little did I know, Grandad wasn't just showing me a cool science trick; he was subtly introducing me to the concept of the reactivity series.
You see, that "mysterious liquid" was likely a dilute acid, and the "loudness" of the reaction – the bubbling – was a pretty good indicator of how readily those metals were willing to lose electrons and react. It’s all about how eager a metal is to get involved in a chemical shindig. Some metals are total party animals, jumping into any reaction they can. Others are more introverted, preferring to hang back and observe.
Must Read
And that, my friends, is where the seemingly complex idea of ordering metals by their reactivity comes in. It’s not some abstract scientific musing; it’s a practical way to understand how these elements behave when they meet other substances. Think of it like a social hierarchy, but for elements. Who’s the most popular? Who’s the loner? Who’s always looking for a fight (or, you know, a chemical reaction)?
The Grand Unveiling: The Reactivity Series
So, how do we actually put these metals in order? Scientists, being the wonderfully curious and sometimes slightly obsessive bunch they are, have figured this out. They've devised something called the Reactivity Series (or sometimes the Activity Series). It's basically a list, arranged from the most reactive metal to the least reactive. Imagine it as a leaderboard, where the top spot is reserved for the metal that throws the biggest chemical tantrum.
What determines this order? It boils down to a few key factors, but the most important one is their tendency to lose electrons. Metals are like magpies; they love to give away their shiny electrons to form positive ions. The easier it is for a metal to shed those electrons, the more reactive it is. It’s a bit like how some people are naturally more inclined to share their toys (or their electron shells, in this case).
Let's break down what makes a metal "reactive." It's not just about whether it rusts or tarnishes (though that's often a symptom of reactivity!). True reactivity in this context refers to its ability to undergo chemical reactions, particularly with acids, water, or other elements. The more readily it reacts, the higher up it sits on the reactivity series.
The Usual Suspects: A Glimpse at the Top and Bottom
At the very top of the reactivity series, you'll find the alkali metals. Think lithium, sodium, and potassium. These guys are the ultimate social butterflies of the metal world. They are so eager to lose an electron that they can react explosively with water. Seriously, drop a piece of potassium into water, and you'll get a fireworks display that would make Grandad proud. They are the rockstars of the reactivity world, constantly seeking attention (and a chemical partner).
These metals are characterized by their single valence electron, which is easily lost. This makes them incredibly strong reducing agents, meaning they readily donate electrons in a redox reaction. They're like the generous friend who always picks up the tab, except instead of money, they're handing out electrons. It's a good thing they are usually stored under oil or kerosene; otherwise, they'd be reacting with the moisture in the air!
On the other end of the spectrum, chilling at the bottom, we have the noble metals. Gold, platinum, silver – these are the introverts. They are so content with their electron configurations that they hardly ever react. They're like the people who politely decline every invitation, preferring their own company. Gold, in particular, is famously unreactive. That's why it's been used for jewelry for millennia without tarnishing or losing its shine. It’s the ultimate aloof celebrity of the periodic table.
These metals have full electron shells or stable electron configurations, making it very difficult for them to lose or gain electrons. They are chemically inert, meaning they resist chemical change. You can leave a piece of gold out in the rain, expose it to air, or even dip it in most acids, and it will likely just sit there, looking smug and unfazed. It's the definition of chemical chill.
How Do We Actually Measure This "Loudness"?
Okay, so Grandad used acids. That's a common way to test reactivity. Acids contain hydrogen ions (H⁺), and more reactive metals can displace these hydrogen ions, forming a salt and releasing hydrogen gas. The faster the hydrogen gas is produced (the more vigorous the bubbling), the more reactive the metal.
Imagine you have a metal, let's call it 'M'. If you put it in an acid, and it reacts to form M²⁺ (a metal ion) and H₂ gas, then:
M + 2H⁺ → M²⁺ + H₂
The faster this reaction happens, the higher 'M' is on the reactivity series. So, in my grandad's shed, the magnesium that fizzes more than zinc is higher up on the list. It's like a race: who can produce the most bubbles the quickest? The winner gets bragging rights (and a higher position on the reactivity series).
But it's not just about acids. We can also test reactivity by seeing if one metal can displace another from its salt solution. This is where things get really interesting and a bit like a chemical game of musical chairs. If a more reactive metal is added to a solution containing ions of a less reactive metal, the more reactive metal will "steal" the ions, kicking the less reactive metal out.
For example, let's say we have a solution of copper sulfate (CuSO₄), which contains Cu²⁺ ions. If we drop a piece of zinc (Zn) into it, something cool happens:
Zn(s) + CuSO₄(aq) → ZnSO₄(aq) + Cu(s)
What’s going on here? Zinc is more reactive than copper. So, the zinc atoms readily give up their electrons to the copper ions (Cu²⁺). These copper ions gain electrons and turn back into solid copper, which will start to coat the zinc. The zinc sulfate (ZnSO₄) stays dissolved in the solution. You’ve effectively “displaced” the copper from its compound using zinc. It's like a chemical takeover!
If you were to try the reverse – putting copper into a zinc sulfate solution – absolutely nothing would happen. Copper isn't reactive enough to steal the zinc ions. It's like a smaller, less popular kid trying to take a toy from a bigger, more popular kid. No dice!

The Digital Age of Reactivity
Nowadays, we don't always need to get our hands dirty with bubbling solutions (though it’s still pretty neat!). Scientists use sophisticated techniques and electrochemistry to determine the standard electrode potentials of metals. These values are essentially a precise measure of a metal's tendency to gain or lose electrons under specific conditions.
The higher (more positive) the standard electrode potential, the less reactive the metal. Conversely, the lower (more negative) the standard electrode potential, the more reactive the metal. It’s a numerical way of ranking them, removing any ambiguity from visual cues like bubbling. It’s like having a definitive score in a video game, rather than just judging by how loud the player is yelling at the screen.
These electrode potentials are measured against a standard reference electrode (usually the standard hydrogen electrode, SHE). So, for every metal, there's a number that tells us its inherent "desire" to react. It’s a bit like assigning a personality trait score to each element.
Putting the Pieces Together: The Order Matters!
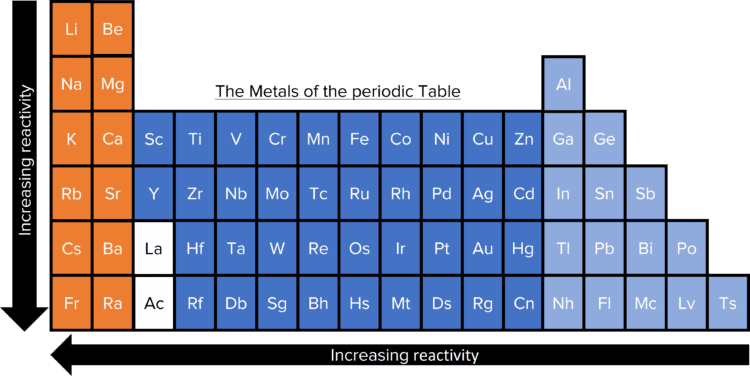
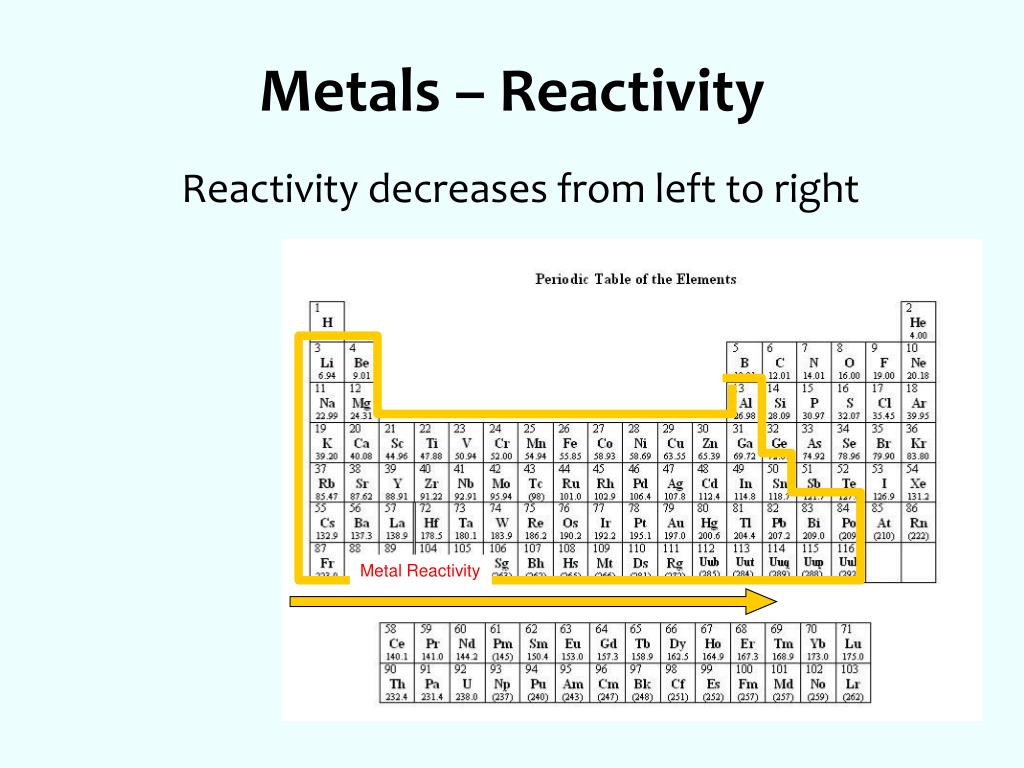
So, what does this reactivity series actually look like? It’s a gradient, a spectrum of chemical enthusiasm. While the exact order can vary slightly depending on the conditions or the specific test used, the general consensus is something like this (from most reactive to least reactive):
Potassium (K) Sodium (Na) Lithium (Li) Calcium (Ca) Magnesium (Mg) Aluminum (Al) Zinc (Zn) Iron (Fe) Nickel (Ni) Tin (Sn) Lead (Pb) Hydrogen (H) (Often included as a reference point, not a metal, but shows how metals react with acids) Copper (Cu) Silver (Ag) Gold (Au) Platinum (Pt)
Woah, that’s a lot of metals! And yes, it can be a bit of a memory challenge to recall the whole thing. But the key is to understand the trend. The top ones are the rebels, the bottom ones are the conformists. Everything in between falls somewhere on that scale of willingness to participate in a chemical reaction.

You might notice hydrogen in there. It's not a metal, but it's often placed in the series because it represents the point at which metals will react with acids to produce hydrogen gas. Metals above hydrogen can displace it, while metals below hydrogen cannot. It’s like the dividing line between metals that are "acid-chasers" and those that are not.
Why Should You Even Care About This? (Besides Impressing Your Grandad)
This isn't just trivia for chemistry geeks. Understanding the reactivity series has some pretty cool real-world applications. Think about it:
- Corrosion and Rusting: Why does iron rust but gold doesn't? Because iron is much higher up the reactivity series. It readily reacts with oxygen and water to form rust (iron oxide). Gold, being so unreactive, just laughs in the face of oxidation. This knowledge helps us choose the right materials for bridges, cars, and pretty much anything exposed to the elements.
- Batteries: Batteries are all about chemical reactions, specifically redox reactions where electrons are transferred. The difference in reactivity between the metals used in a battery is what drives the flow of electrons, creating electricity. The greater the difference in reactivity, the higher the voltage. So, the reactivity series is a fundamental concept in battery design!
- Extraction of Metals: Metals like aluminum are very reactive, so they are often found in nature combined with other elements (in ores). To extract them, you need to use very strong chemical processes. Less reactive metals, like copper, are sometimes found in their native state or in simpler compounds that are easier to extract. The reactivity series dictates the difficulty and cost of getting pure metals from their ores.
- Alloys: Mixing metals to create alloys can change their properties. Sometimes, by combining metals, you can make them less reactive. For instance, stainless steel is an alloy of iron, chromium, and nickel, and the addition of chromium significantly increases its resistance to corrosion compared to pure iron.
So, the next time you see a rusty fence, a shiny gold ring, or a battery powering your phone, you’re witnessing the practical implications of the reactivity series in action. It’s a fundamental principle that governs how the world of metals behaves around us.
The Takeaway: It's All About the Electrons
At its heart, the reactivity series is a way to organize metals based on their inherent tendency to give away electrons. It’s a ranking of their chemical "boldness." The more willing a metal is to part with its electrons, the higher it sits on the list, and the more readily it will participate in chemical reactions.
It’s a spectrum, from the explosively reactive alkali metals to the stoic noble metals. And understanding this order helps us predict how metals will behave, choose the right materials for our needs, and even harness their power in technologies like batteries.
So, next time you’re near some bits of metal, don't just see them as dull chunks of stuff. See them as participants in a grand chemical dance, each with its own unique level of enthusiasm. And maybe, just maybe, you'll feel a tiny bit like my grandad, seeing the world of chemistry unfold before your eyes, one bubbling reaction at a time!


