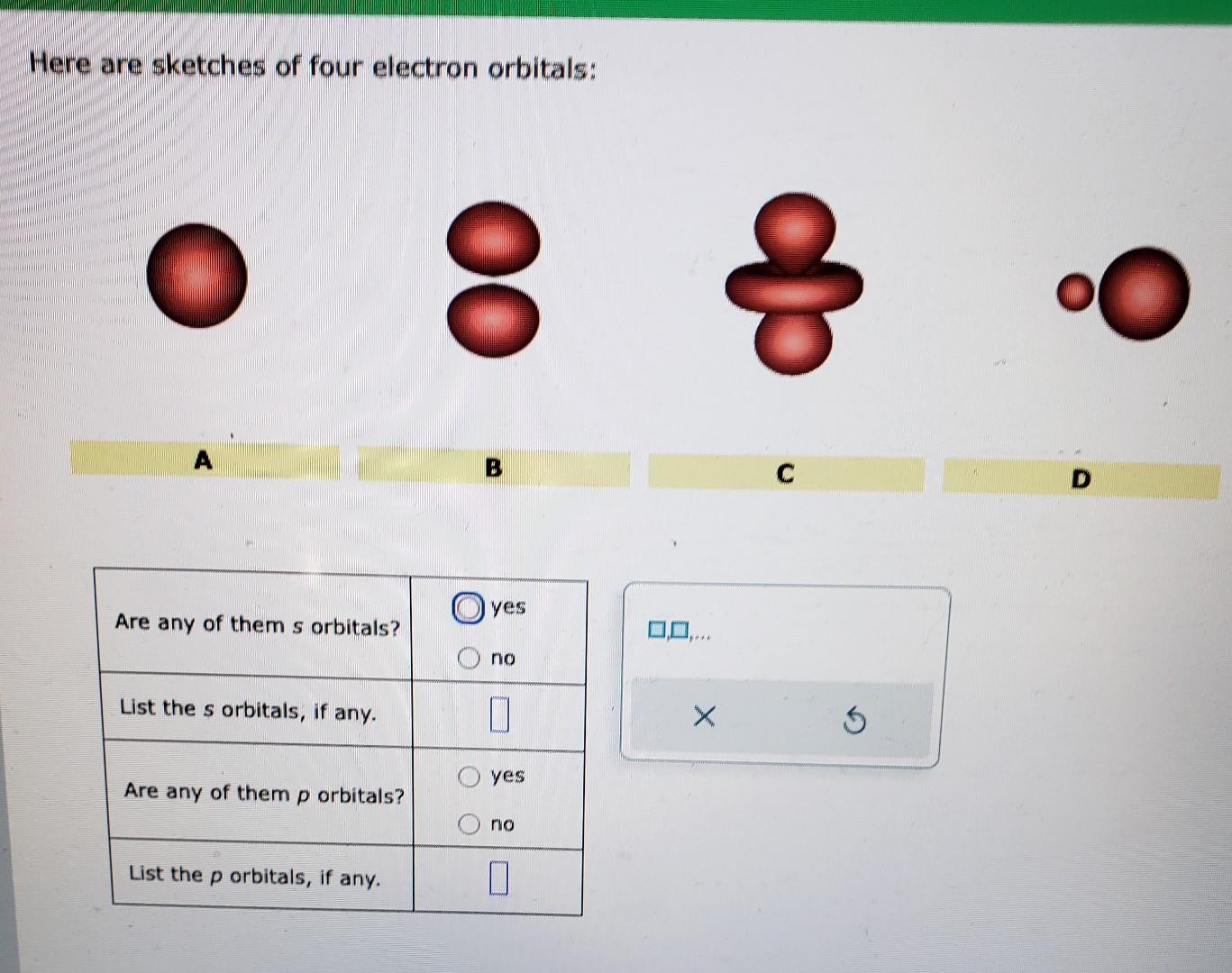
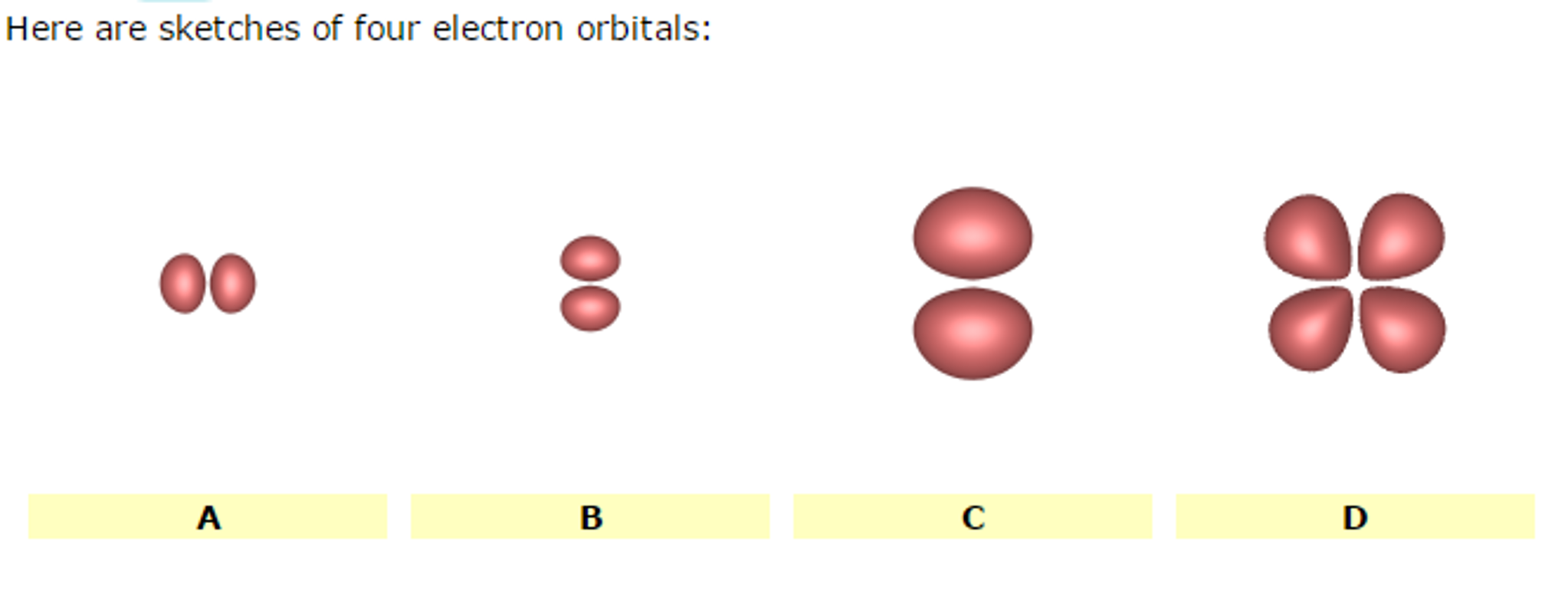
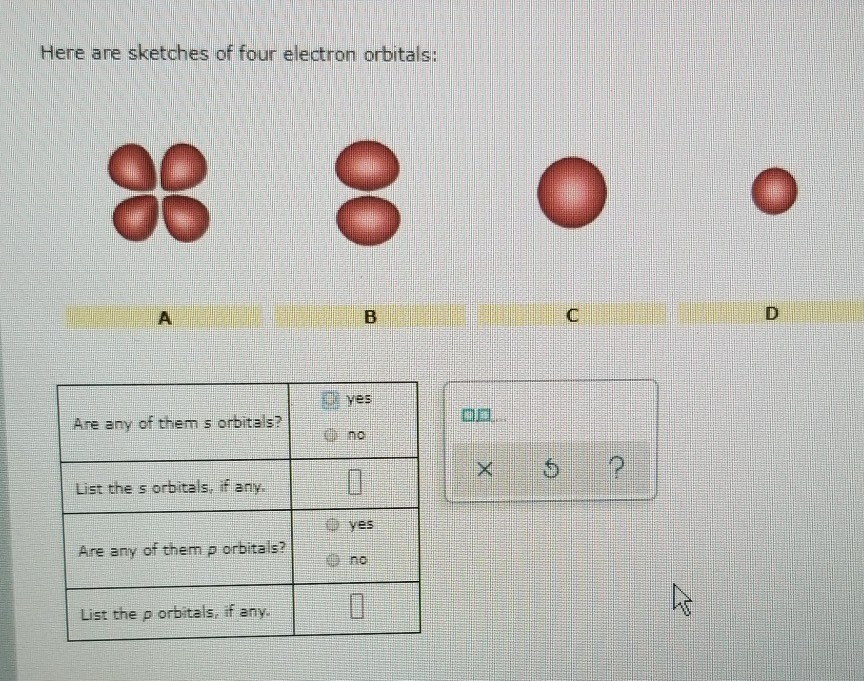
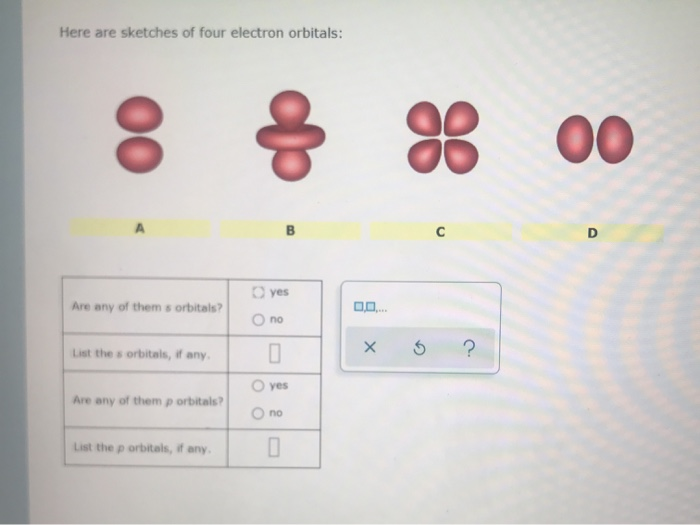
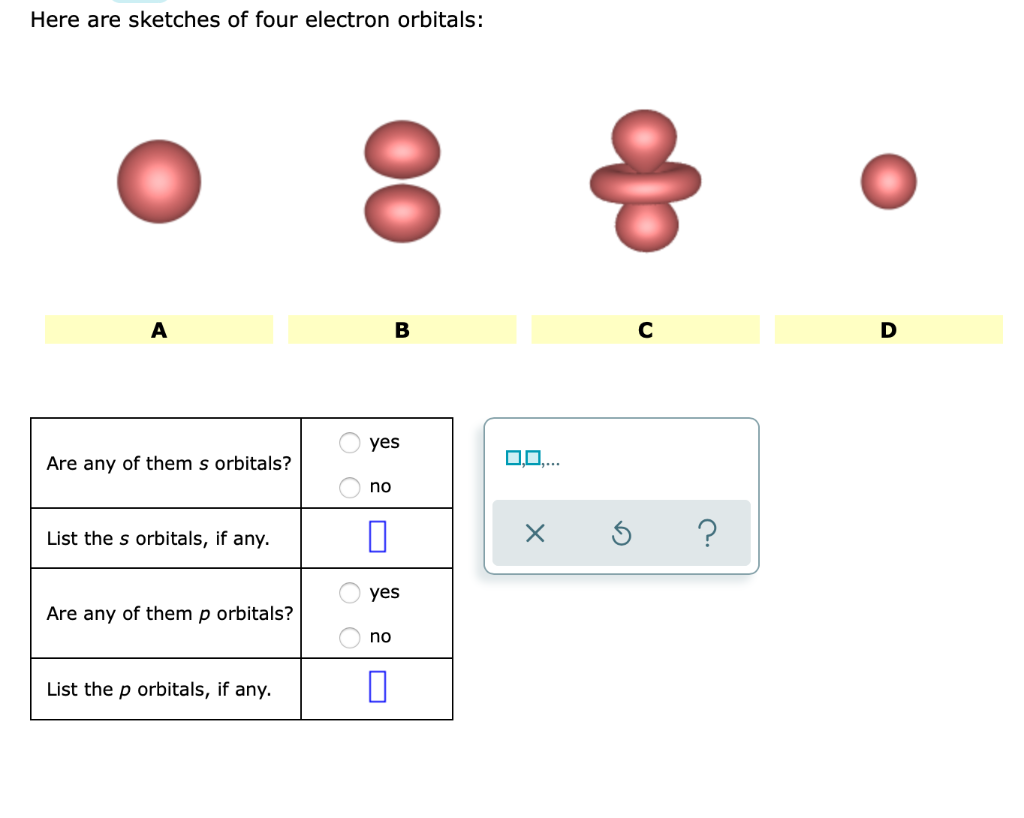
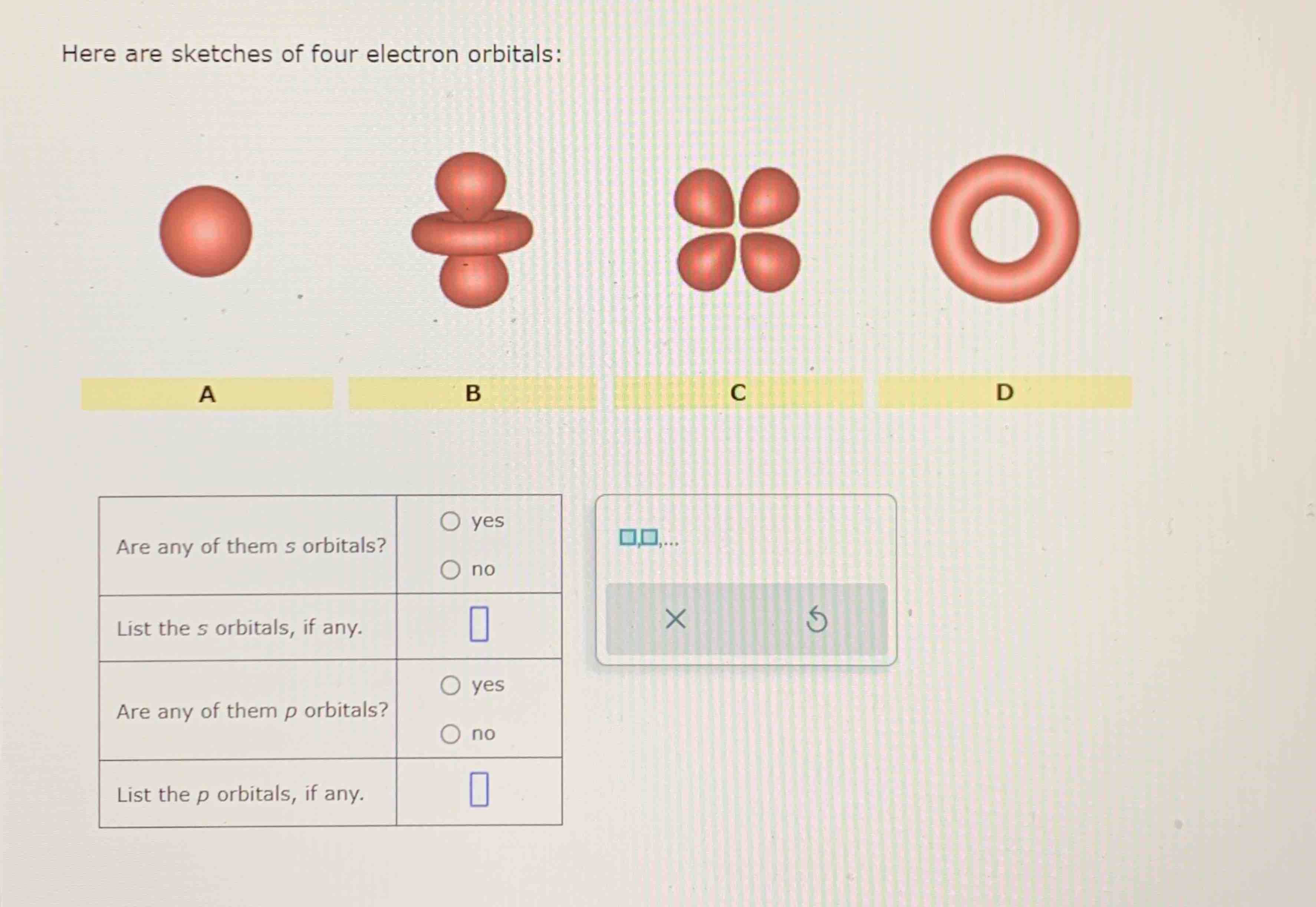
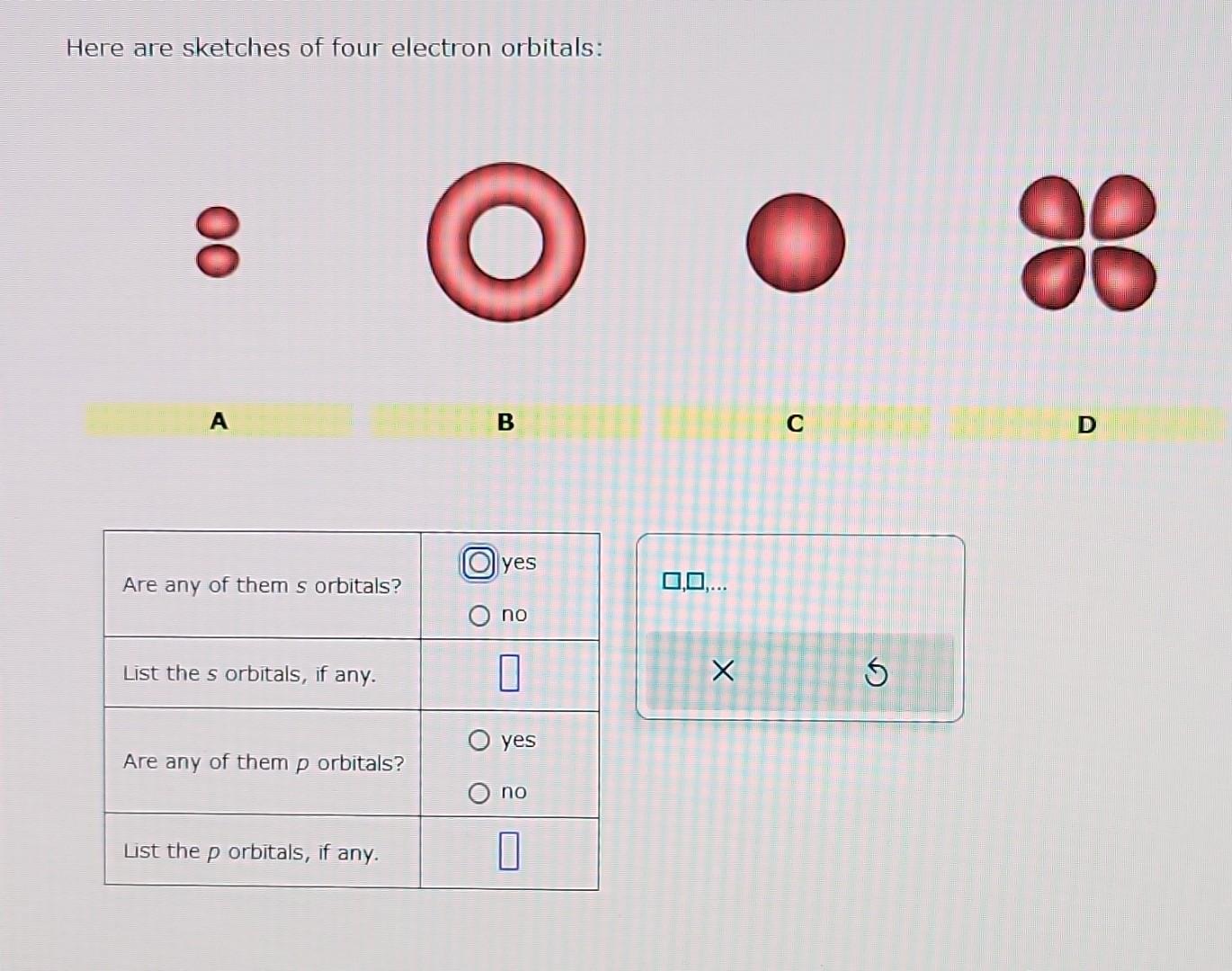
Here Are Sketches Of Four Electron Orbitals
So, the other day, I was trying to explain to my niece, who’s about seven, what atoms are. You know, the tiny, fundamental building blocks of everything. She looked at me with those big, curious eyes, and I started blabbering about protons, neutrons, and electrons whizzing around. Then came the dreaded question: “What do they do? Where do they go?”
I’d prepared for this, sort of. I’d seen those cool diagrams online, you know, the ones with the little fuzzy clouds or these weird, defined shapes. I pulled up a picture on my tablet, picturing myself as some sort of wizened science guru, ready to impart atomic wisdom. “See,” I said, pointing at a diagram that looked suspiciously like a balloon, “this is where an electron might be!”
Her response? A perfectly innocent, “But it looks like a bubble.” And you know what? She wasn’t wrong. It did look like a bubble. My carefully constructed explanation about electron clouds and probability distributions sort of deflated like a leaky balloon. It made me realize that sometimes, the simplest analogies are the most deceiving, and the most accurate descriptions can sound utterly bizarre.
Must Read
This little interaction got me thinking. We’re taught about electrons being in orbitals, right? But what does that really mean? Is it like planets orbiting a sun? Or something much, much weirder? Well, buckle up, because today we’re diving into the wonderfully strange world of electron orbitals. And to help us out, we’ve got some sketches. Not like, artistic masterpieces, but actual representations of these elusive electron haunts.
The Classic Analogy: A Not-So-Helpful Planet
For ages, the go-to image for atoms was the Bohr model. Think of a tiny solar system. The nucleus is the sun, and electrons are little planets zipping around in fixed, circular paths. It’s tidy, it’s easy to visualize, and it works okay for explaining basic chemical bonding, like how hydrogen likes to hold hands with oxygen. But for a more accurate picture? Nah, not really. It’s like trying to describe a bustling city by only showing a single, empty street. It misses so much of the nuance.
The problem with the planetary model is that it implies electrons have a definite location and a predictable path at any given moment. But quantum mechanics, bless its complicated heart, tells us that’s just not how it works at the atomic scale. Electrons are… more like a hazy fog or a mischievous ghost. They’re somewhere, but pinning them down is a real challenge.
This is where the concept of orbitals comes in. Instead of a path, think of an orbital as a 3D region of space around the nucleus where there’s a high probability of finding an electron. A high probability, mind you. Not a 100% guarantee. It’s like saying, “There’s a 90% chance you’ll find your keys in your bag.” You might find them on the counter, or under the sofa, but the bag is your best bet.
Introducing the Orbital Gang: S, P, D, and F
So, if electrons aren’t circling like tiny satellites, what are these orbital shapes we see? Well, they’re not arbitrary doodles. They represent the shapes of these probability clouds. And there are different types, each with its own distinct personality and spatial orientation. The most common ones we talk about are the s, p, d, and f orbitals.
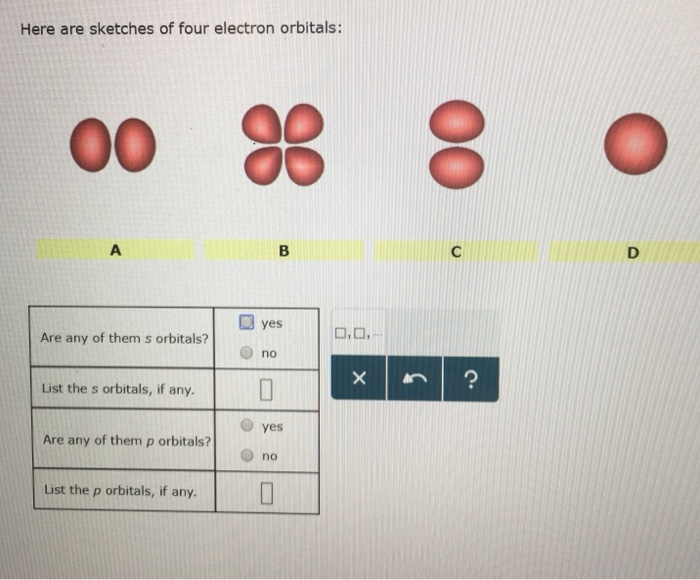
Let’s start with the simplest, the s orbital. These are like the dependable, no-nonsense members of the orbital family. Every atom has at least one s orbital. And guess what? They’re spherical. Like a perfect, perfectly symmetrical ball. Imagine a tiny, invisible bubble. That’s your s orbital.
You can have multiple s orbitals in an atom, each one just a bigger version of the one before it. So, there’s the 1s orbital (closest to the nucleus), then the 2s, 3s, and so on. The higher the number, the larger the sphere and the further away from the nucleus the electron is likely to be found. Easy peasy, right? Almost too easy, which is usually a red flag in chemistry.
The S-Orbital: The Humble Sphere
Let’s look at the sketch of an s orbital. See that? It’s just… a circle. Or, in 3D, a sphere. No lobes, no strange twists, just a uniform probability of finding the electron in any direction from the nucleus. It’s the atomic equivalent of a perfectly round cheese ball. And while it might seem boring, these s orbitals are fundamental. They’re the foundation upon which other, more complex orbitals are built.
Think about the 1s orbital. It’s the lowest energy orbital in an atom. It’s where the electron wants to be, closest to the attractive pull of the nucleus. When an atom has only one electron, like hydrogen, it sits in this 1s orbital. It’s the ultimate cozy spot. If you were an electron, and you could pick a place to hang out, you’d probably aim for the lowest energy state, right? Less effort involved. Smart thinking, electrons!
The 2s orbital is also spherical, but it’s bigger. And it has a slightly more complex structure within that sphere. If you could see it in more detail, it’s not just a solid ball. It’s more like nested spheres with regions of probability. But for a general sketch, we often just show it as a larger sphere. Don’t let the simplicity of the sketch fool you; even the humble s orbital has its quantum quirks.
The P-Orbitals: The Dumbbell Duo (and a Trio!)
Now, things start to get a bit more interesting. When we move up in energy levels, we encounter the p orbitals. Unlike the s orbitals, p orbitals are not spherical. They’re shaped more like dumbbells. You know, those weights people lift at the gym? Yeah, like that, but with two lobes.
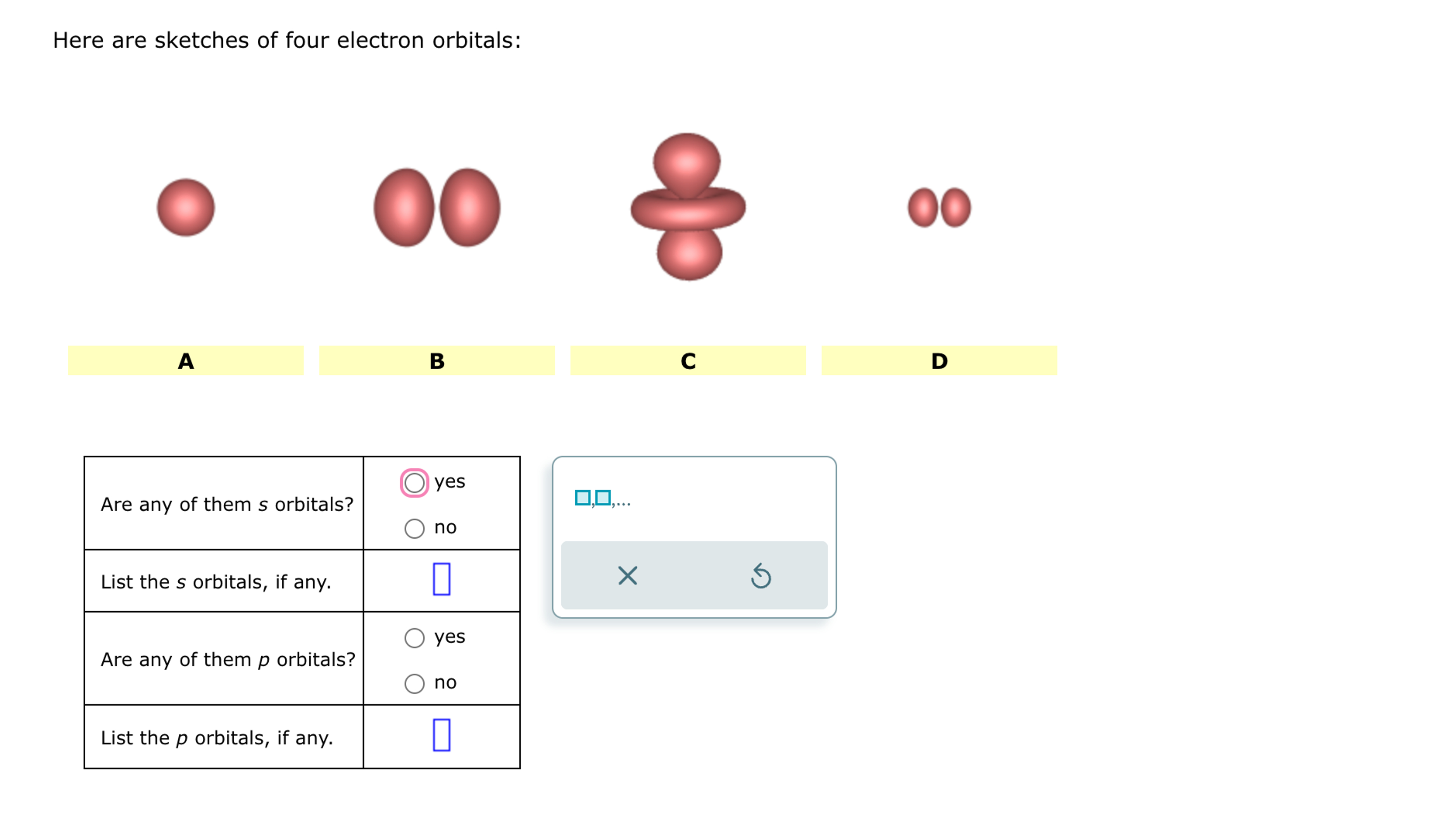
And here’s the kicker: for any given energy level (starting from the second energy level, n=2), there are three p orbitals. Why three? Because they are oriented along the three perpendicular axes of space: the x, y, and z axes. So, you have a px orbital, a py orbital, and a pz orbital. Imagine three dumbbells, each pointing in a different direction, all centered on the nucleus.
When you see a sketch of a p orbital, you'll typically see two opposing lobes, with the nucleus sitting right at the pinched-off center between them. It's like the electron has decided it likes being in two places at once, as long as they're on opposite sides of the nucleus. This shape has a huge impact on how atoms interact and form bonds. It’s not just about having an electron; it’s about where and in what shape that electron likes to reside.
The P-Orbital Sketches: A Directional Dance
Let’s look at the p orbital sketches. You’ll see them depicted as these symmetrical, two-lobed shapes. The nucleus is at the origin, the point where the two lobes meet. The px orbital will have its lobes along the x-axis, the py along the y-axis, and the pz along the z-axis. If you were to see all three simultaneously, it would look like a starburst of dumbbells.
Each of these p orbitals can hold a maximum of two electrons. So, in a given energy level (like n=2), you can have up to six electrons distributed among the three p orbitals (2 electrons in px, 2 in py, and 2 in pz). This is why the second energy shell can hold up to 8 electrons: 2 in the 2s orbital and 6 in the 2p orbitals.
The dumbbell shape is fascinating because it means that the electron density is concentrated in specific directions. This directional nature is crucial for understanding chemical bonding. When atoms approach each other to form molecules, the overlapping regions of these p orbitals are where the magic happens, forming covalent bonds. It’s not a fuzzy, undefined interaction; it’s quite specific and directional.
The D-Orbitals: Getting Fancy with Five
Alright, things are about to get a bit more complicated, and I’m warning you now, the shapes are going to start looking less intuitive. We’re moving onto the d orbitals. These become relevant starting from the third energy level (n=3). And just when you thought three p orbitals was a lot, how many d orbitals do you think there are? Five! Yes, five.
The shapes of the d orbitals are more varied and complex. Most of them look like double dumbbells, sort of like four lobes. But there's one that’s a bit different, looking like a dumbbell with a donut around its middle. It’s like the universe decided to get creative with its electron housing options.
These d orbitals are important for transition metals, those elements in the middle of the periodic table that have all sorts of cool and varied chemistry. Their d orbitals are partially filled, and this is what gives them their unique properties, like forming colorful compounds and having multiple oxidation states. It’s like they have extra rooms in their atomic mansions, and they can furnish them in all sorts of interesting ways.
The D-Orbital Sketches: A Cloverleaf Convention (Mostly)
Let’s check out the d orbital sketches. You’ll notice that four of them have a shape that resembles a four-leaf clover. Each leaf is a lobe, and the nucleus is at the center. These are the dz² (which looks a bit different, more like a dumbbell with a torus), dx²-y², dxy, dxz, and dyz orbitals.
The labels tell you their orientation. For example, dx²-y² has its lobes along the x and y axes, while dxy has its lobes in between the x and y axes. The dz² orbital is unique, with two lobes along the z-axis and a ring or torus shape around the middle in the xy plane. It’s a bit of an anomaly, but it’s part of the set.
These d orbitals contribute to the electron configurations of elements, especially the transition metals. Their involvement in bonding can be quite intricate. The fact that they have these specific, directional shapes influences the geometry of molecules and the way electrons are shared. Imagine trying to build a complex Lego structure – the shape of the individual bricks matters a lot!
The F-Orbitals: The Pinnacle of Complexity
And finally, we have the f orbitals. These are even more complex and appear from the fourth energy level (n=4) onwards. There are seven f orbitals for each energy level where they exist. Their shapes are incredibly intricate and difficult to visualize, often described as having multiple lobes, sometimes eight. They are the most spatially complex of the standard orbitals.
The f orbitals are crucial for the chemistry of the lanthanides and actinides, the two rows of elements at the bottom of the periodic table. These elements have unique properties due to the involvement of their f electrons. Think of them as having really elaborate, multi-room mansions where the electrons can spread out in very unusual ways.
While the sketches of f orbitals are often highly stylized and simplified, they represent regions of space with a high probability of finding an electron. The complexity of their shapes means they can participate in bonding in very subtle and nuanced ways. Honestly, trying to draw or even accurately describe an f orbital shape can make your brain feel like it’s doing a triple backflip.
The F-Orbital Sketches: Beyond Comprehension (Almost!)
If you look at sketches of f orbitals, prepare for a visual overload. They often appear as complex, multi-lobed structures. Some might look like starbursts, others like more abstract shapes. They’re a testament to the fact that nature, at the quantum level, can be incredibly intricate and defy easy intuition. For most general chemistry purposes, we don’t delve too deeply into the exact shapes of f orbitals, but it’s good to know they exist and play a role.
The key takeaway from all these shapes – the spheres, the dumbbells, the cloverleafs, and the bewildering f orbitals – is that electrons don’t just randomly float around. They occupy specific regions of space dictated by their energy and quantum numbers. These regions, the orbitals, have distinct shapes and orientations.
And these shapes are not just academic curiosities. They are the very foundation of chemistry. The way atomic orbitals overlap when atoms bond dictates the structure of molecules, the properties of materials, and the way chemical reactions proceed. So, next time you see one of these seemingly abstract shapes, remember that it’s a window into the fundamental forces that shape our universe.
So, back to my niece and her bubble. She wasn't entirely wrong. The s orbital is a bit like a bubble. But the other orbitals? They’re more like intricate, directional furniture arrangements in the atomic house. And understanding these arrangements helps us understand everything from how water molecules stick together to why metals conduct electricity. It's a strange and beautiful world, this atomic landscape. And we've only just peeked at the blueprints!