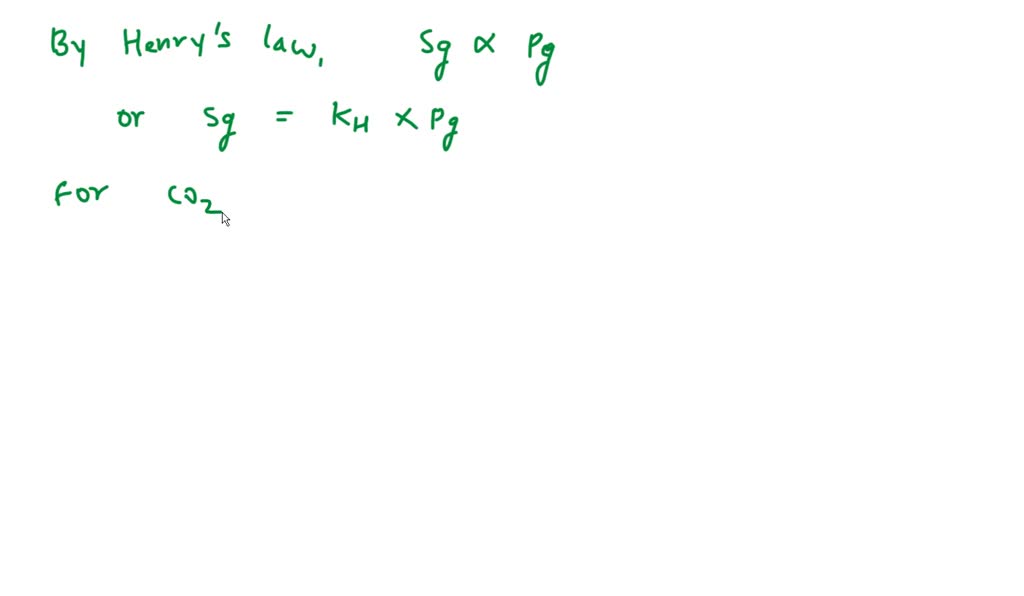Henry's Law Constant For Co2 At 25 Degrees C

Hey there, science enthusiast (or just someone who stumbled upon this page and is now mildly curious)! Ever wonder why your fizzy drinks are, well, fizzy? Or how all that carbon dioxide (CO2) manages to hang out in the ocean? Well, buckle up, because we’re about to dive into a super cool concept that explains a lot of this everyday magic: Henry's Law Constant for CO2 at 25 degrees Celsius. Yeah, I know, it sounds like something out of a mad scientist's lab, but trust me, it's way less intimidating than it sounds. Think of it as the secret recipe for how gases love to mingle with liquids.
So, imagine you've got a nice, refreshing soda. You pop the top, and what happens? Fizzzzzzzzz! That glorious sound is CO2 escaping its liquid prison. This isn't some random act of gas rebellion; it's all thanks to a brilliant scientific principle named after a super smart dude named William Henry. Back in the day (early 1800s, to be precise), he basically figured out a nifty relationship between how much gas you can dissolve in a liquid and the pressure of that gas sitting on top of the liquid. Pretty neat, right? It’s like he cracked the code for how gases behave when they decide to take a dip.
Now, what exactly is Henry's Law? In plain English (because who needs fancy jargon when we can have fun?), it states that the concentration of a gas dissolved in a liquid is directly proportional to the partial pressure of that gas above the liquid. Translation alert! This means if you increase the pressure of the gas, more of it will dissolve into the liquid. And if you decrease the pressure, some of it will decide to bail and go back into the gas phase. It’s all about balance, people! Think of it like a cosmic tug-of-war between the gas molecules trying to escape and the liquid molecules politely inviting them in. The pressure is the muscle on the gas side of the rope.
Must Read
So, where does our star player, CO2, come into this? Well, CO2 is a gas that loves to play in liquids, especially water. It's responsible for the tartness in carbonated beverages and plays a massive role in the Earth's climate system (more on that later, maybe!). And the temperature? Ah, yes, 25 degrees Celsius. This is a pretty standard temperature, often considered room temperature or a nice, comfortable day. It’s the kind of temperature where you’d happily sit on a park bench, perhaps with a fizzy drink in hand. So, when we talk about Henry's Law Constant for CO2 at 25°C, we're essentially looking at a specific "sweet spot" where this gas-liquid dance is happening under particular conditions.
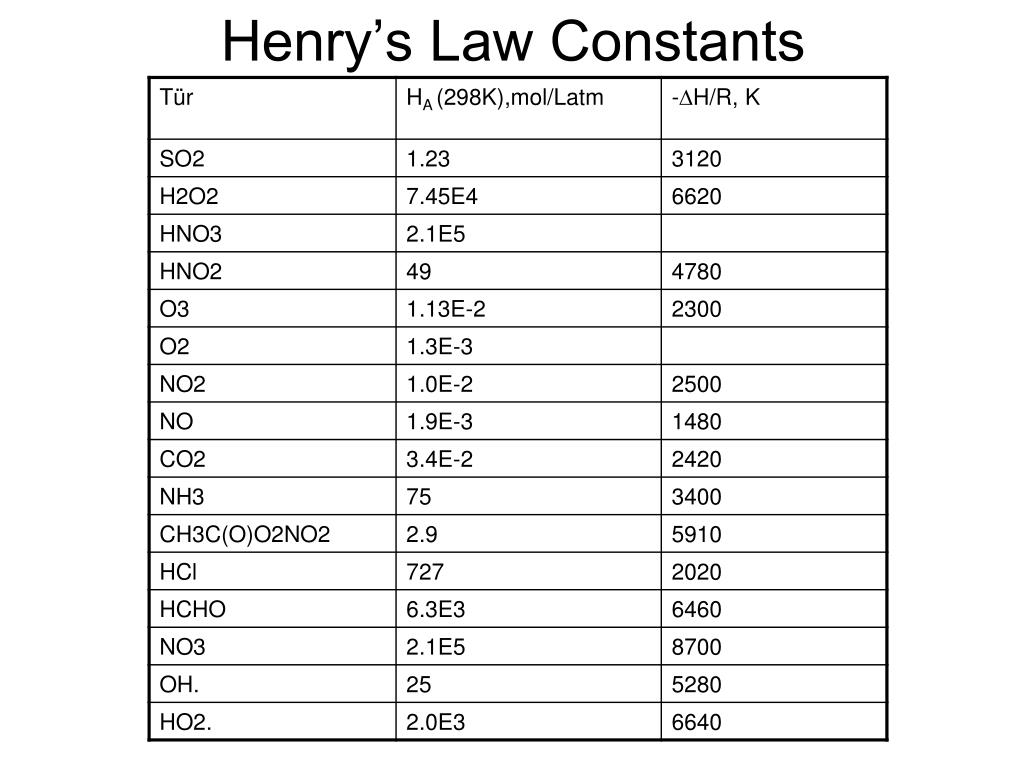
Let’s get a little more technical, but don't worry, we'll keep it light! The Henry's Law Constant, often represented by the letter 'H' or 'kH', is basically a proportionality constant. It tells us how much gas will dissolve at a given pressure and temperature. It’s like a special conversion factor, a little numerical secret that unlocks the relationship. Different gases have different constants because some are naturally "stickier" to liquids than others. For example, ammonia is super soluble in water, while nitrogen is not so much. CO2 is somewhere in the middle, and its constant at 25°C is what we’re zooming in on today.

Now, here’s where it gets interesting. The value of this constant isn't just some random number pulled out of a hat. It's determined experimentally. Scientists get their lab coats on, grab their beakers (probably filled with lovely 25°C water), and carefully measure how much CO2 dissolves at different pressures. It’s a bit like baking – you need the right ingredients and the right measurements to get the perfect result. And the result for CO2 at 25°C? It’s a number that tells us, for every atmosphere of CO2 pressure above water, how many moles of CO2 will be dissolved per liter of water. I’m not going to give you the exact number just yet because, honestly, it can be expressed in different units, and that can get a tad confusing. But rest assured, it's a well-established and important value in the scientific world.
Why 25 degrees Celsius, you ask? Well, as I mentioned, it's a common reference point. Most scientific data is collected and reported at specific, standardized conditions to make comparisons easier. Think of it like a universal "hello, nice to meet you" temperature for gases and liquids. If you change the temperature, the constant changes too. Generally, as temperature increases, the solubility of gases in liquids tends to decrease. So, that fizzy drink might go a little flat faster on a hot day compared to a cool one. It's like the gas molecules get a bit too energetic and decide it's time for an adventure outside the liquid. Henry's Law Constant is temperature-dependent, making our 25°C snapshot super valuable for understanding those specific conditions.
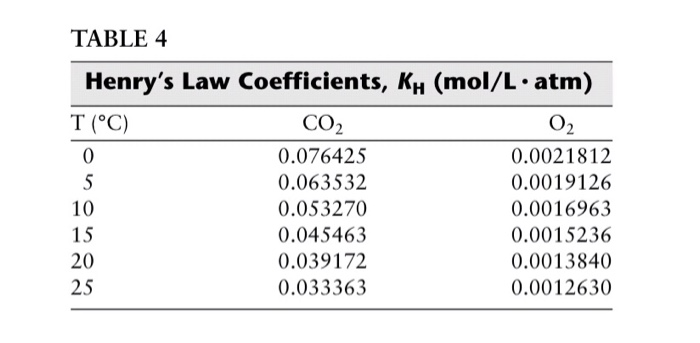
Let's talk about the units for a sec. Because science likes to be thorough (sometimes too thorough, am I right?), Henry's Law Constant can be expressed in a few ways. You might see it in units of atm/mol/L, or mol/(kg·atm), or even Pa·m³/mol. They all essentially describe the same relationship, just with different ways of measuring pressure, moles, and volume. It's like saying "a dozen" or "12" – same quantity, different name. For CO2 at 25°C, the commonly cited value is around 0.034 mol/(L·atm) or, if you're working with mole fraction and partial pressure, something like 1.0 x 10-2. It’s this little number that helps engineers design carbonation machines, helps oceanographers understand CO2 uptake, and generally explains why your soda doesn't just taste like flat water.

So, what does this constant actually mean in practice? If you have pure CO2 gas at a pressure of 1 atmosphere sitting above water at 25°C, approximately 0.034 moles of CO2 will dissolve in every liter of that water. That might sound like a tiny amount, but remember, even a small concentration of dissolved gas can create a big fizz! It's the cumulative effect. And if you double the pressure of CO2 to 2 atmospheres, you'll dissolve roughly double the amount of CO2. See? Directly proportional! Henry’s Law is your friendly guide to these gas-liquid interactions. No complex calculus required, just a simple proportional relationship. It's like a scientific handshake.
Why is this important beyond just enjoying a bubbly beverage? Well, CO2 is a bit of a big deal for our planet. It's a greenhouse gas, so understanding how it dissolves in the oceans is crucial for climate modeling. The oceans are like a giant sponge for CO2, absorbing a significant amount from the atmosphere. Henry's Law helps scientists predict how much CO2 the oceans can take up under different atmospheric conditions. It's a vital piece of the puzzle in understanding our changing climate. So, next time you're thinking about CO2, remember it's not just in your soda; it's also a major player in global environmental science. And our friend, the Henry's Law Constant at 25°C, is a key to understanding its behavior.

Think about it: if the Henry's Law Constant for CO2 were drastically different, our world would be a very different place. Maybe our oceans would be full of undissolved gas, or perhaps they’d absorb too much, leading to even more extreme climate changes. This constant is a small but significant number that shapes our planet's chemistry and our everyday experiences. It's a testament to how even the seemingly mundane aspects of science can have profound implications. It’s like discovering a tiny cog that, when turned just right, keeps a giant machine running smoothly.
So, to recap, Henry's Law Constant for CO2 at 25 degrees Celsius is our handy dandy guide to how much CO2 loves to hang out in water when the temperature is nice and pleasant. It’s all about pressure and proportion. It explains your fizzy drinks, it helps scientists understand our oceans, and it's a beautiful example of a simple scientific principle with far-reaching consequences. It's a reminder that the world around us is full of fascinating, interconnected processes, and sometimes, the most complex phenomena can be understood through elegant, straightforward laws.
Isn't science just the coolest? It takes something as simple as a bubble in your drink and connects it to the vastness of our oceans and the health of our planet. So, the next time you hear that satisfying pssst of a soda can opening, give a little nod to Henry and his constant. You'll know that a little bit of scientific magic is at play, making your world a more bubbly and understandable place. And that, my friends, is a reason to smile, even if you don't have a beaker in sight!