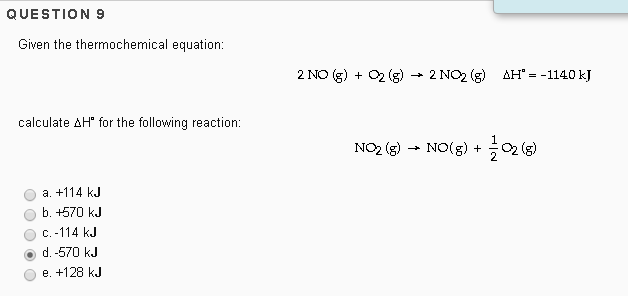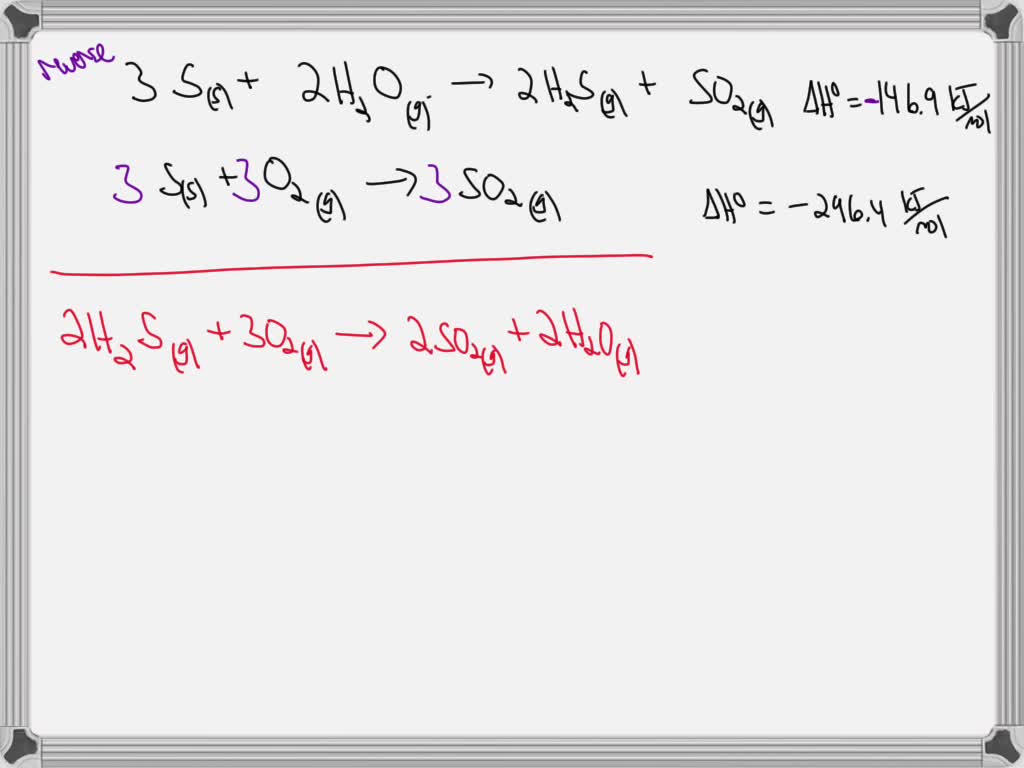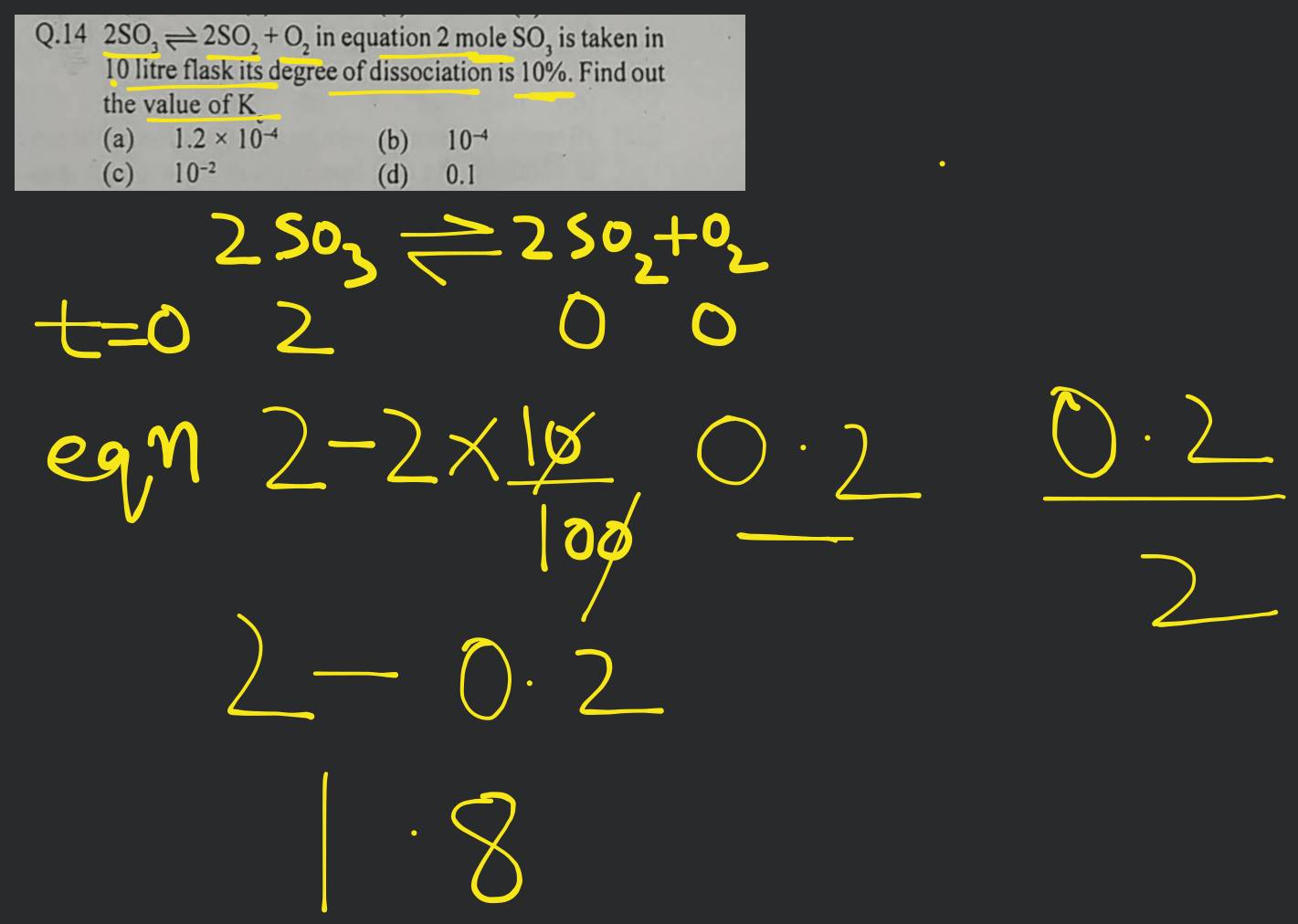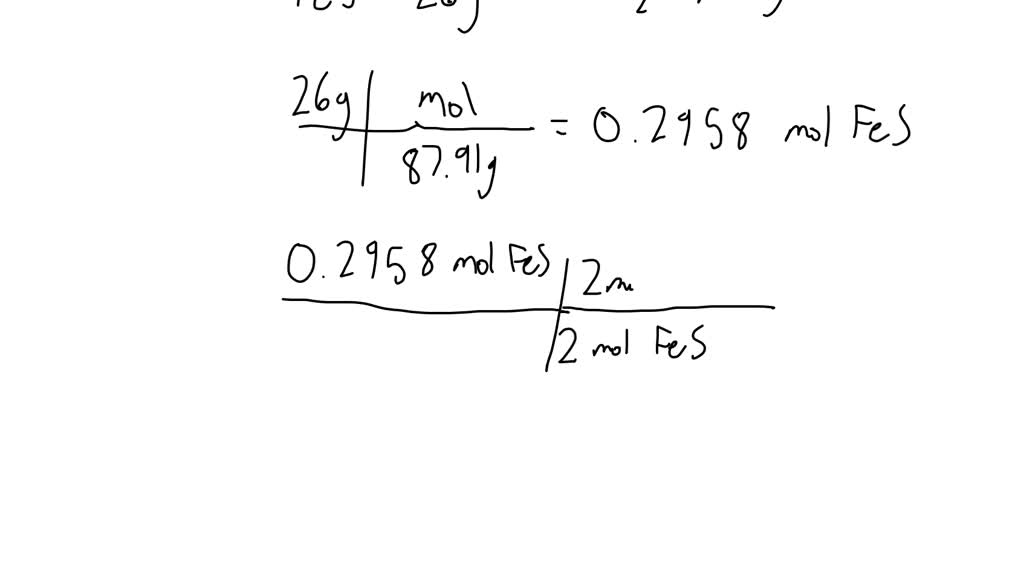Given The Thermochemical Equation 2so2 + O2

Ever wondered what makes things burn, rust, or even breathe? It all comes down to the magical world of chemical reactions, and today, we're going to peek behind the curtain at a particularly fascinating one: the formation of sulfur trioxide from sulfur dioxide and oxygen. This might sound like something straight out of a science lab, and in a way, it is! But understanding this reaction, represented by the thermochemical equation 2SO2 + O2 → 2SO3 + energy, isn't just for chemists in white coats. It's about understanding the fundamental processes that shape our world, from the industrial furnaces that power our cities to the subtle transformations happening in nature every single day.
Think of it this way: every time you see a campfire crackling, or a metal object developing a patina, or even when your body uses energy to digest food, you're witnessing chemical reactions at play. These reactions are the unsung heroes of our existence, constantly rearranging matter and releasing or absorbing energy. The reaction we're diving into today, the one that transforms sulfur dioxide (SO2) and oxygen (O2) into sulfur trioxide (SO3), is a prime example of this energy exchange. It’s a process that's not only crucial for industrial applications but also has significant implications for our environment.
The beauty of this particular thermochemical equation lies in its ability to tell us more than just what chemicals are involved. The "thermochemical" part is key! It means we're not just looking at the ingredients and the final product, but also at the energy that's either released or absorbed during the transformation. In the case of 2SO2 + O2 → 2SO3, it's an exothermic reaction. That means it gives off heat, like a little fiery burst as it happens. This release of energy is what makes the reaction so useful and, frankly, so interesting to study. It’s like getting a bonus – not only do you get new stuff (sulfur trioxide), but you also get a little bit of warmth!
Must Read
So, why should you care about sulfur trioxide? Well, it’s not just some obscure chemical compound. Sulfur trioxide (SO3) is a really important intermediate in the production of sulfuric acid (H2SO4). And sulfuric acid? That’s one of the most widely produced chemicals in the world! It's a workhorse in so many industries, from the manufacturing of fertilizers that help grow our food to the production of detergents, dyes, and even in oil refining. Imagine trying to make all those essential products without sulfuric acid – it would be a very different world indeed! Therefore, understanding the reaction that creates its precursor, SO3, is fundamental to understanding a huge chunk of modern industry.
The benefits of this reaction go beyond just industrial output. On a more environmental note, controlling the formation of sulfur oxides is crucial. While this specific reaction leads to a useful product, uncontrolled burning of fossil fuels can release sulfur dioxide into the atmosphere, which can then react with oxygen and water to form sulfuric acid, a major component of acid rain. Acid rain can damage forests, lakes, and buildings. So, by understanding and managing reactions like this, scientists and engineers can work towards cleaner industrial processes and mitigating environmental damage. It's a perfect example of how chemistry can be both a tool for progress and a means to protect our planet.

Let's break down the equation a bit more without getting too technical. On the left side, we have our reactants: two molecules of sulfur dioxide (2SO2) and one molecule of oxygen (O2). They're like the ingredients ready to be mixed. When they come together under the right conditions – perhaps with a catalyst to speed things up and a bit of heat to get them going – they transform into two molecules of sulfur trioxide (2SO3). This is our product. And as we mentioned, this transformation doesn't happen in a vacuum; it releases energy. This energy release is often represented by a positive value after the arrow, indicating heat is being given off.
The beauty of thermochemical equations is that they provide a quantitative measure of this energy change. For this specific reaction, the enthalpy change (often symbolized by ΔH) is typically around -198 kJ/mol. That negative sign is your cue that energy is being released – it's exothermic. This number tells us precisely how much heat is produced per mole of sulfur trioxide formed. This precise knowledge is invaluable for engineers designing chemical plants. They need to know how much heat will be generated so they can design systems to manage it – either to harness it for other processes or to prevent overheating.

Imagine a giant chemical reactor. If this reaction releases a lot of heat, you need to make sure the reactor can handle that temperature safely. You might need cooling systems, or you might even be able to use that heat to boil water and generate steam for electricity. It’s a fascinating interplay between the microscopic world of molecules and the macroscopic world of industrial engineering. This reaction, therefore, is not just an abstract concept; it's a practical consideration that impacts safety, efficiency, and economics in large-scale chemical production.
So, the next time you hear about a chemical reaction, remember that there's a whole story behind it. It’s a story of transformation, of energy, and of how these tiny molecular events can have a huge impact on the world around us. The equation 2SO2 + O2 → 2SO3 + energy is a tiny window into this grand narrative, revealing the power and utility hidden within the fundamental processes of chemistry. It’s a testament to the fact that even the seemingly simple act of rearranging atoms can be both incredibly useful and deeply fascinating.