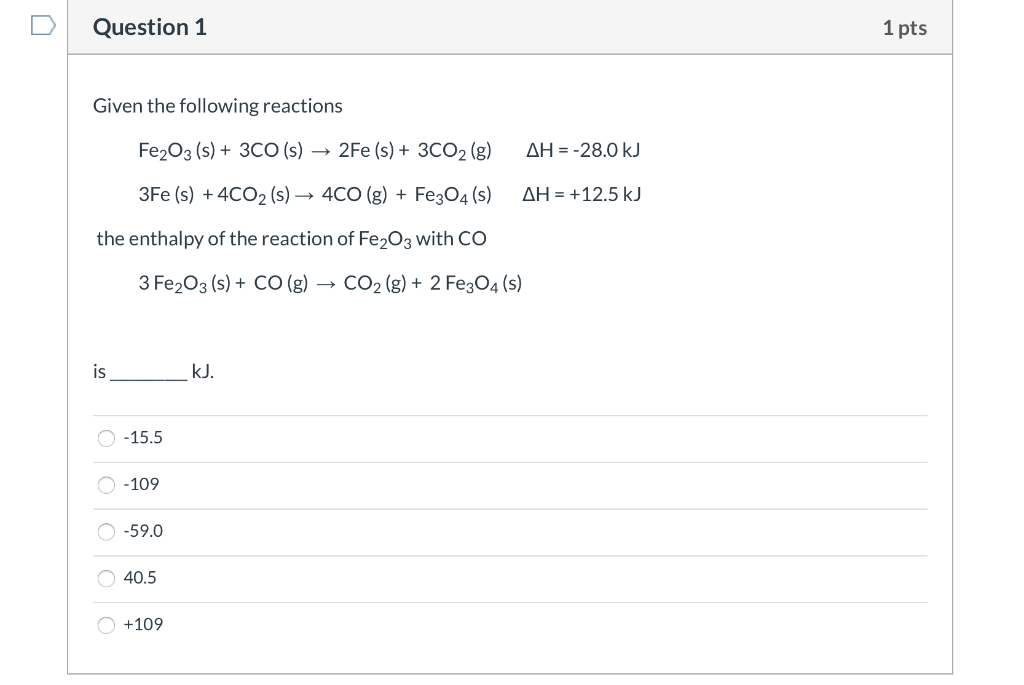
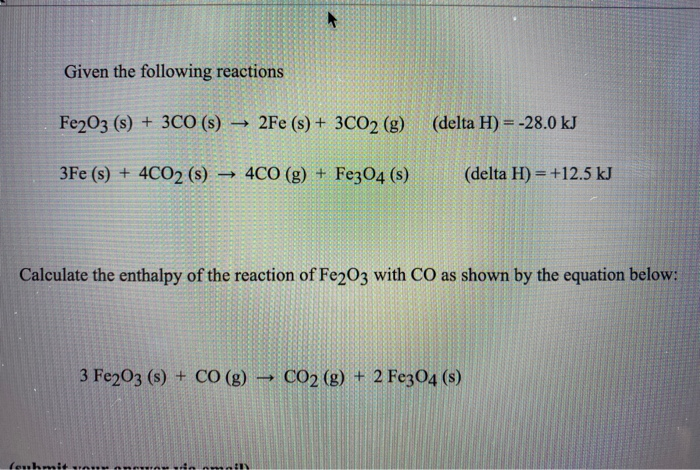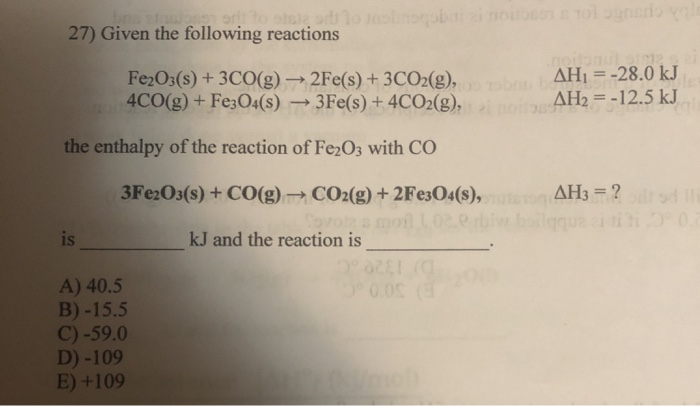Given The Following Reactions Fe2o3 + 3co
Ever find yourself staring into the fiery abyss of your grill, wondering what magical transformation is happening to that sad, grey lump of iron ore? Well, my friends, you're not alone. We're about to dive headfirst into a chemical party, and our VIP guests are iron(III) oxide (that's rust, in its most common form) and carbon monoxide (a gas we definitely don't want to be breathing in large quantities, so let's keep this strictly theoretical, folks!). The reaction? Fe2O3 + 3CO. Sounds a bit like a secret code, doesn't it? Like something you'd whisper to your tech-savvy friend when you need to bypass a stubborn firewall. But nope, this is just good old-fashioned chemistry, and surprisingly, it's got more in common with your daily life than you might think.
Think about it. We've all seen rust. It's that unsightly orange-brown coating that appears on everything from your garden tools to that vintage bicycle you swore you'd restore someday. It's the universe's way of saying, "Hey, this metal has seen better days and is slowly but surely returning to its earthy origins." Iron(III) oxide, or Fe2O3, is basically the fancy name for this everyday phenomenon. It's like calling your slightly burnt toast "artisanal carbonized bread." Sounds more sophisticated, but it's still toast that’s a bit too well-done.
And then there's carbon monoxide, CO. Now, this one’s a bit of a sneaky character. It’s odorless, colorless, and generally unwelcome. Imagine a guest who shows up uninvited, doesn't say a word, but subtly ruins the entire party by making everyone feel a bit woozy. That's CO. It’s the byproduct of incomplete combustion, which means when things don't burn completely, this gas tends to pop out and say hello. Think of your old gas stove that sometimes smelled a bit funny, or even a car idling in a closed garage – not ideal, to say the least!
Must Read
So, when Fe2O3 meets CO, what happens? It's like a cosmic dance where the grumpy, oxidized iron is looking for a way to shed its rusty coat, and the carbon monoxide is just itching to grab some oxygen. They get together, and suddenly, the iron is like, "You know what? This whole rust thing is getting old. You, CO, you look like you've got energy to spare. How about you take this pesky oxygen off my hands?" And CO, being the oxygen-hungry gas it is, is all too happy to oblige.
The chemical equation, Fe2O3 + 3CO, tells us that for every one molecule of iron(III) oxide, we need three molecules of carbon monoxide to perform this oxidative makeover. It's like a tiny, very specific recipe for transformation. Imagine you’re trying to bake a cake, and you need exactly three eggs and a cup of flour. Too much or too little, and things get… well, messy. In this case, the "messy" is actually a good thing because we end up with something desirable: pure iron and carbon dioxide.
The iron(III) oxide (Fe2O3) is the ingredient that's been through it. It's been exposed to air and moisture, and it's become chemically bound with oxygen. It's like that friend who’s been through a tough breakup and is feeling a bit brittle and oxidized. They're not their shiny, pure selves anymore.

Carbon monoxide (CO) is the energetic, slightly reckless friend who's looking to make a change. It's got a carbon atom and an oxygen atom, and it's got this strong desire to bond with more oxygen. It's like that friend who's always trying to "fix" things, sometimes with a bit too much enthusiasm. In this reaction, CO acts as a reducing agent, meaning it takes oxygen away from other substances. It’s the ultimate oxygen thief, and in this context, it's stealing from the rust.
When these two meet, under the right conditions (usually high heat, like in a blast furnace – more on that later!), a chemical reaction kicks off. The carbon monoxide molecules essentially "steal" the oxygen atoms from the iron(III) oxide. It’s a bit like a game of tag, but with oxygen atoms. CO tags the oxygen on Fe2O3, and they run off together, forming carbon dioxide (CO2).
So, the Fe2O3, which was all bundled up with oxygen, starts to let go. It’s like it’s saying, "Phew, finally! I can breathe!" The oxygen atoms leave the iron, and the carbon monoxide molecules happily scoop them up, turning into carbon dioxide (CO2). This is the part where the iron gets to be its true, unadulterated self again. It sheds its rusty shell and becomes pure, metallic iron. Think of it as a spa day for iron, but instead of cucumber slices and mud masks, it’s getting a good scrub-down with carbon monoxide.
The equation continues: Fe2O3 + 3CO → 2Fe + 3CO2. This is the full story. For every Fe2O3 and three COs, we get two atoms of pure iron (Fe) and three molecules of carbon dioxide (CO2). The iron is the prize, the shiny reward for this chemical exchange. And the carbon dioxide? Well, that's just the exhaust. It's the stuff that floats away, like the grateful sighs of the now-liberated iron.

This, my friends, is the fundamental principle behind how we get iron out of iron ore. Most of the iron we use in our world, from the steel beams in skyscrapers to the cast iron skillet you probably own, starts its life as iron(III) oxide, buried deep within the earth. And to get that pure iron out, we need to perform this very specific, very energetic, and frankly, very hot, chemical process.
Imagine the sheer scale of this. We're not talking about a single rusty bolt here. We're talking about mountains of iron ore being processed. This is where the real-life connection gets a bit more industrial, a bit more… fiery. The most common place this reaction happens is in a blast furnace. And let me tell you, a blast furnace is not your average backyard barbecue. It's more like the fiery belly of a dragon, spewing heat and molten metal.
Inside a blast furnace, you’ve got layers upon layers of iron ore, coke (which is a purified form of coal that also acts as a fuel and a source of carbon for the reaction), and limestone. Then, hot air is blasted in, making everything glow with an intensity that would make the sun jealous. The coke burns, producing carbon monoxide, and this CO is the star player that starts to strip the oxygen away from the iron ore.
It's a multi-stage process, really. As the materials descend into the furnace, they heat up. The carbon monoxide is formed in the lower, hotter parts and then rises up to meet the iron ore. It’s like a perfectly choreographed performance. The carbon monoxide is the lead dancer, twirling and grabbing oxygen, while the iron ore is the graceful partner, slowly shedding its oxygen and revealing its true metallic form.
Think about the iron(III) oxide as a stubborn shell around a precious pearl. The CO is the skillful jeweler who, with a bit of heat and effort, cracks open that shell to reveal the gleaming pearl – the pure iron.
At the top of the furnace, the temperature is cooler. Here, the coke itself can react directly with the iron ore, also helping to remove oxygen. But the real heavy lifting, the most efficient oxygen-stripping, happens with the carbon monoxide. It’s the workhorse of this whole operation.
The limestone? That's like the helpful cleaning crew. It decomposes and forms calcium oxide, which then reacts with impurities in the iron ore (like sand and clay) to form a molten slag. This slag floats on top of the molten iron, making it easier to separate. So, while the CO is busy turning rust into iron, the limestone is making sure everything else gets cleaned up and out of the way. It’s a well-oiled (or rather, well-melted) machine!
When you see steel products, from your car to your kitchen appliances, remember that they all started as Fe2O3, that humble rust, and CO, that sneaky gas, having a very hot, very intense conversation in a blast furnace. It’s a process that’s been around for centuries, and it’s fundamentally what allows us to build, to create, and to shape the modern world.
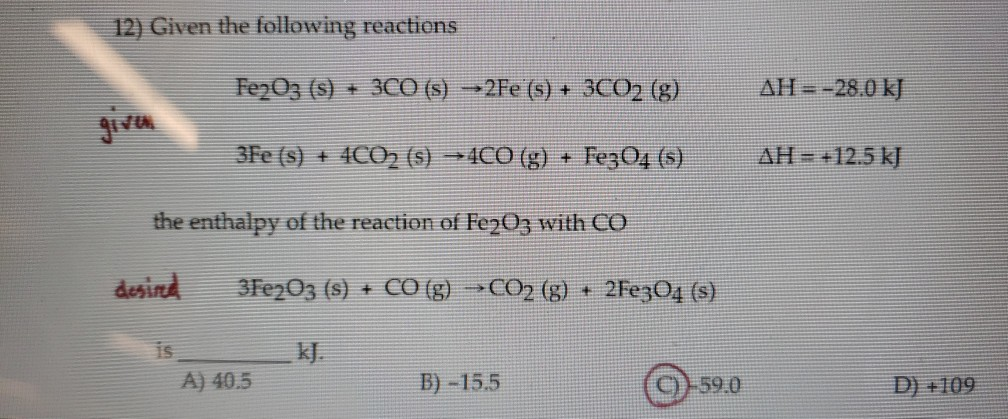
It’s fascinating to think about how something as simple as rust and a gas we try to avoid can be the key to unlocking one of the most important metals on Earth. It’s like finding out your annoying neighbor’s weird collection of bottle caps is actually the secret ingredient to the world’s best jam. Who knew?
The purity of the iron produced depends on the efficiency of the reaction and the quality of the raw materials. Sometimes, the iron that comes out of the blast furnace is still not quite pure enough for certain applications. It might have a bit too much carbon in it, making it brittle. That's where further refining processes come in, turning that "pig iron" into steel, which is much more versatile. But the initial step, the magical transformation of Fe2O3 with CO, is the crucial foundation.
So, the next time you see a rusty car or hear about the steel industry, you can smile to yourself, knowing the secret handshake between Fe2O3 and CO that made it all possible. It’s a testament to the power of chemistry, turning the ordinary into the extraordinary, one oxygen atom at a time. It's proof that even the most mundane-looking substances can hold the potential for something truly remarkable, especially when they have a little help from their friends, even if one of them is a bit of a gas guzzler (of oxygen, that is!). It’s a beautiful, fiery symphony of atoms, all working together to forge the world we live in.
And as for the carbon dioxide produced? Well, that's another story for another day. But for now, let's just appreciate the incredible feat of turning rust into the backbone of our civilization, all thanks to a reaction that, at its core, is about iron and carbon monoxide having a very energetic chat.