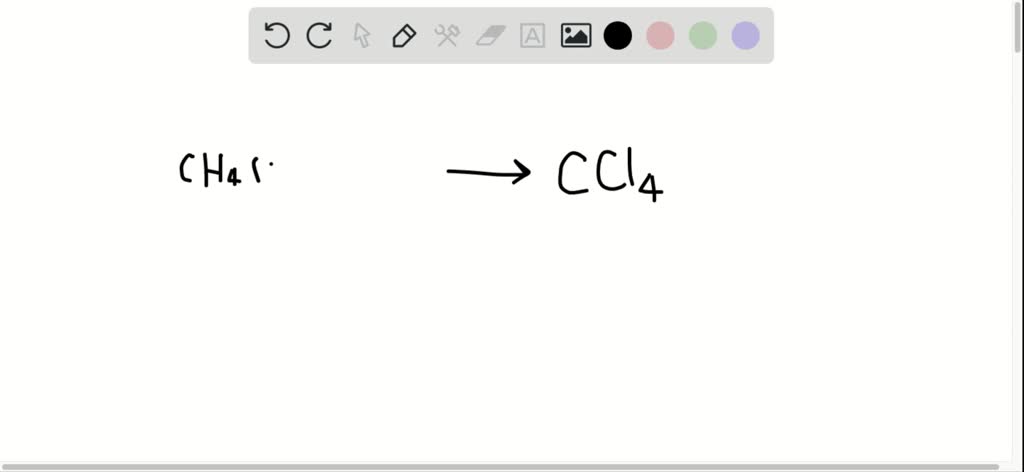
Given The Equation For A Reaction C4h10+cl2
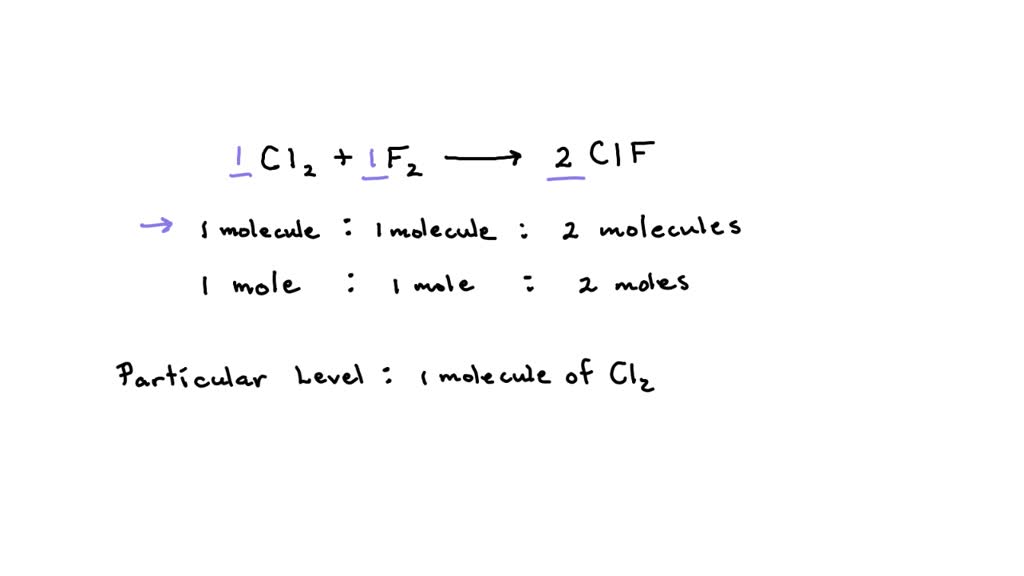
Ever stumbled upon a chemical equation and thought, "What in the world is that all about?" We've all been there! Today, let's dive into a seemingly simple reaction: C4H10 + Cl2. This isn't just a jumble of letters and numbers; it's a little glimpse into how the world around us works, and understanding it can be surprisingly fun and relevant.
So, what's the big deal with C4H10 + Cl2? Well, C4H10 is the chemical formula for butane, the stuff you might find in a lighter or a camping stove. And Cl2? That's chlorine gas, a common chemical with various industrial uses. When these two get together, they undergo a chemical reaction, meaning they rearrange their atoms to form something entirely new. It’s like a molecular dance, with atoms swapping partners!
The primary purpose of understanding reactions like this is to grasp the fundamental principles of chemistry. It helps us predict what will happen when different substances mix, which is crucial for everything from developing new medicines to creating everyday materials. The benefits are immense: it fuels innovation, helps us understand safety protocols (like how to handle chlorine gas!), and allows us to appreciate the complex processes happening constantly, even in our own bodies.
Must Read
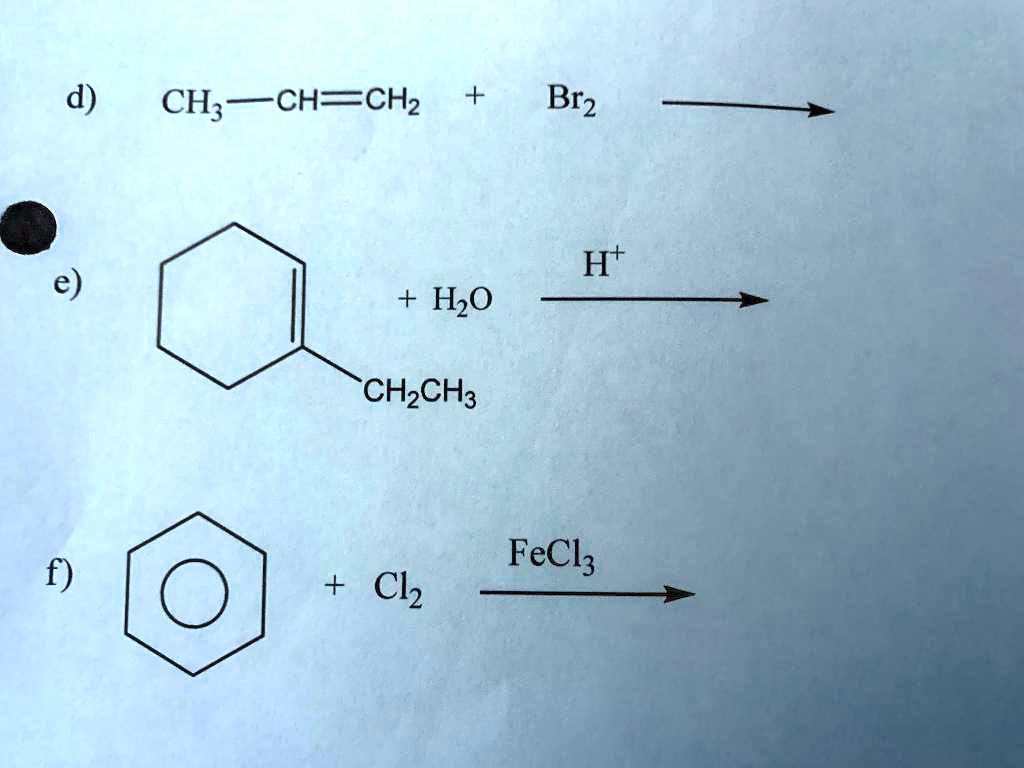
In education, this equation is a fantastic starting point for teaching about alkane halogenation. It’s a classic example used to introduce concepts like free radical substitution, where atoms are swapped out one by one. Think of it as replacing a passenger on a bus with someone from the sidewalk. You might see this in high school chemistry classes, explaining how we can modify hydrocarbons for different purposes.
But what about daily life? While you won't likely be mixing butane and chlorine gas in your kitchen, the principles are at play. When fuels burn (like butane in your grill), they react with oxygen. When plastics are made, chemical reactions are involved. Even the way your body digests food is a series of complex chemical reactions. Understanding C4H10 + Cl2 helps build that foundational knowledge.
Ready to explore this a bit more? You don't need a full lab! A great starting point is to simply learn the names of the compounds involved: butane and chlorine. Then, you can look up what happens when they react. You'll discover that the reaction isn't a simple one-step process; it often involves different stages and can produce several different products depending on the conditions. You can even watch animations online that visualize these molecular changes. It’s a wonderfully accessible way to peek behind the curtain of chemistry and see the amazing transformations that shape our world!