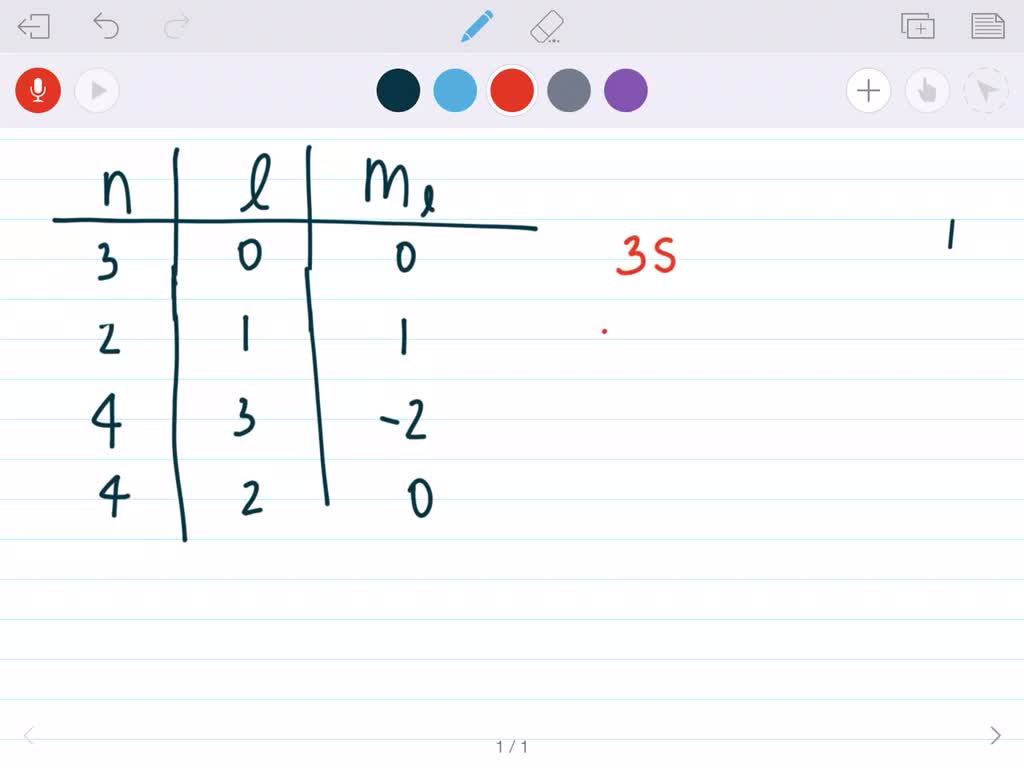
Give The Numbers For Ml For An F Orbital.

Hey there, curious minds! Ever found yourself staring up at the night sky, wondering about all the tiny, invisible building blocks that make up everything around us? We're talking about atoms, those little dynamos of the universe. And within those atoms, things get even more interesting. Today, we're diving into a concept that might sound a little sci-fi at first, but trust me, it's surprisingly down-to-earth and, dare I say, even a little bit cool.
We're going to chat about the humble "f orbital". Now, if you've ever been in a science class, you might have heard of s, p, and d orbitals. Think of them like different types of rooms in an atomic hotel, where electrons like to hang out. The s orbitals are like cozy studio apartments, simple and round. The p orbitals are like slightly bigger one-bedroom places, with a bit more shape. The d orbitals? Well, they're getting a little more complex, like a junior suite with a few extra nooks and crannies.
But the f orbital? Oh, they're the penthouse suites of the atomic world! They're the most complex, the most intricate, and frankly, the most fascinating when it comes to electron behavior. And the question of the hour is: what are the numbers that describe these f orbitals? In the world of quantum mechanics, which is basically the super-detailed instruction manual for the tiny universe, these numbers are called quantum numbers.
Must Read
Think of it like this: imagine you're trying to find a specific book in a massive library. You wouldn't just say "I want a book." You'd need more details, right? You'd need the section, the shelf number, the specific book on that shelf. Quantum numbers are the library catalog for electrons in atoms. They tell us precisely where an electron is likely to be and what its energy is like.
For an f orbital, there are four key quantum numbers. Let's break them down, nice and easy, like unwrapping a present.
The First Quantum Number: The Principal Quantum Number (n)
This is the big boss, the main descriptor. It tells us the energy level or the shell the electron is in. Think of it like the floor of our atomic hotel. Higher numbers mean higher floors, more energy, and a greater distance from the atom's nucleus (which is like the central lobby). So, an electron in the first shell (n=1) is super close to the nucleus, while an electron in the fifth shell (n=5) is chilling way up on the top floor.
For f orbitals, the principal quantum number n must be at least 4. That's right, these fancy f orbitals don't even show up until the fourth floor of our atomic hotel! You won't find any f orbitals on the first, second, or third floors. They're just too complex and energetic for those lower levels. Imagine trying to fit a grand piano into a tiny studio apartment – it just doesn't fit! So, n can be 4, 5, 6, and so on. The higher the n, the bigger and more energetic the f orbital can be.
The Second Quantum Number: The Azimuthal or Angular Momentum Quantum Number (l)
This number tells us about the shape of the orbital. It's like describing the layout of that atomic hotel room. We already know that s orbitals have l=0 (round, studio apartment), p orbitals have l=1 (dumbbell shape, one-bedroom), and d orbitals have l=2 (more complex, junior suite). So, what do you think it is for an f orbital?
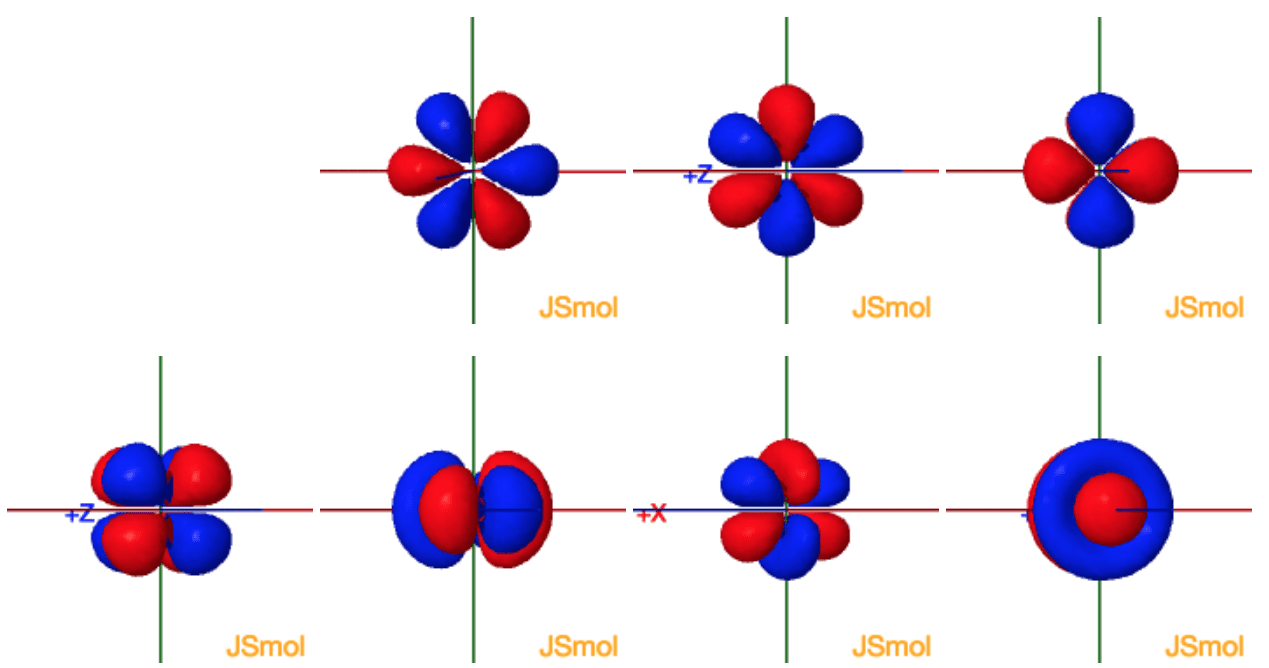
You guessed it! For an f orbital, the azimuthal quantum number l is always 3. This is the defining characteristic that says, "Yep, this is an f orbital!" It signifies a shape that's even more complicated than the d orbitals. Think of it like trying to describe a really elaborate, multi-room suite with all sorts of weird angles and alcoves. It's not a simple sphere or a dumbbell; it's something much more intricate.
This l=3 value means that f orbitals have a particularly complex spatial distribution. They aren't just one simple blob or two lobes; they can have multiple lobes and nodes (areas where an electron is unlikely to be found) arranged in specific, intricate patterns. Imagine a really intricate origami design – that's the kind of complexity we're talking about!
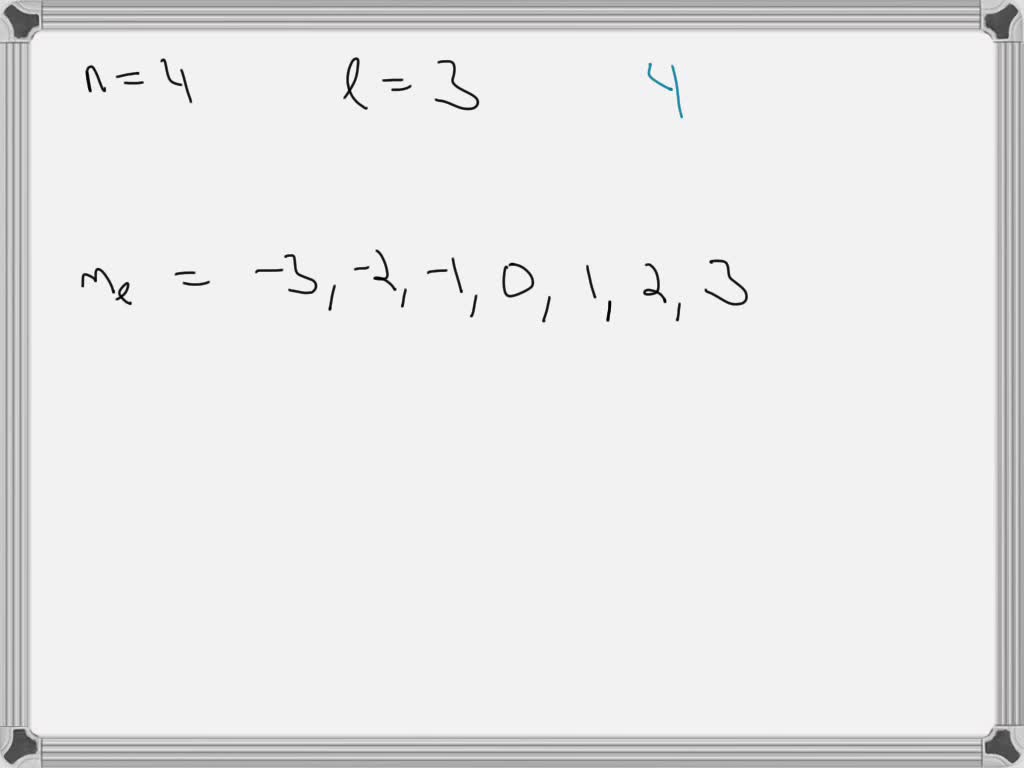
The Third Quantum Number: The Magnetic Quantum Number (ml)
Okay, so we know the energy level (n) and the shape (l). Now, what about the orientation of that shape in three-dimensional space? That's where the magnetic quantum number, ml, comes in. Think of it as telling you which way the atomic hotel room is facing. Is it facing north, south, east, or west? Or maybe it has a view of the mountain, or the ocean!
For a given value of l, the possible values of ml range from -l to +l, including zero. Since for f orbitals, l=3, the possible values for ml are:
- -3
- -2
- -1
- 0
- +1
- +2
- +3
That's a total of seven different possible orientations for an f orbital! This is a big jump from the p orbitals (which have 3 orientations) and d orbitals (which have 5 orientations). So, for each energy level (n) where f orbitals exist (n ≥ 4), there are seven distinct f orbitals. They're all the same shape (defined by l=3), but they're pointing in different directions in space.
Imagine you have a really fancy, multi-faceted diamond. Each facet is like an f orbital, same basic shape, but angled differently. These seven orientations are crucial because they allow electrons to arrange themselves in ways that best minimize their repulsion and fill up the available space efficiently. It’s like arranging furniture in a room – you want to make sure it all fits nicely and doesn’t bump into each other too much!
The Fourth Quantum Number: The Spin Quantum Number (ms)
Finally, we have the spin quantum number, ms. This one's a bit like giving each electron its own tiny internal compass. Electrons are not just sitting there; they're constantly spinning, and they can spin in one of two directions: up or down. These spins are represented by:
- +1/2 (spin up)
- -1/2 (spin down)
This might seem like a small detail, but it's incredibly important. It’s governed by the Pauli Exclusion Principle, which basically says that no two electrons in an atom can have the exact same set of all four quantum numbers. So, even if two electrons are in the same f orbital (same n, l, and ml), they must have opposite spins (different ms) to coexist.
Think of it like assigning beds in a dorm room. If you have two students sharing a bunk bed (our f orbital), they can’t both lie on their backs with their heads at the same end! One has to sleep on their back, and the other on their stomach, or one’s head at one end, the other’s at the other. That’s the essence of the spin quantum number – it's a way for electrons to distinguish themselves and avoid occupying the exact same quantum state.

So, Why Should We Care About F Orbitals?
You might be thinking, "This is all very interesting, but does it really matter in my everyday life?" Absolutely! The behavior of electrons in these f orbitals is responsible for some of the most fascinating and useful properties of certain elements, particularly the lanthanides and actinides, which are found at the bottom of the periodic table. These are the "heavyweights" of the element world, and their electrons are filling those f orbitals!
Elements like uranium (which powers nuclear reactors) and plutonium are actinides. Their unique properties, including their radioactivity and how they interact with other substances, are directly tied to the electrons in their f orbitals. Even elements that seem less dramatic, like gadolinium, which is used in MRI contrast agents to help doctors see inside your body with incredible detail, owe their abilities to the intricate dance of electrons in their f orbitals.
Understanding f orbitals helps scientists design new materials with specific magnetic, optical, or chemical properties. It's crucial for developing advanced technologies, from catalysts that make industrial processes more efficient to the rare-earth magnets that power your phone and electric car. Without the knowledge of these complex orbital shapes and their electron occupants, we wouldn't have many of the modern conveniences we rely on.
So, the next time you hear about f orbitals, don't think of them as just a dry scientific concept. Think of them as the intricate, multi-faceted spaces where some of the universe's most important electron players reside. They're the reason why certain elements behave the way they do, leading to incredible innovations and a deeper understanding of the world around us. It's a little bit of atomic magic, explained by a few simple numbers!