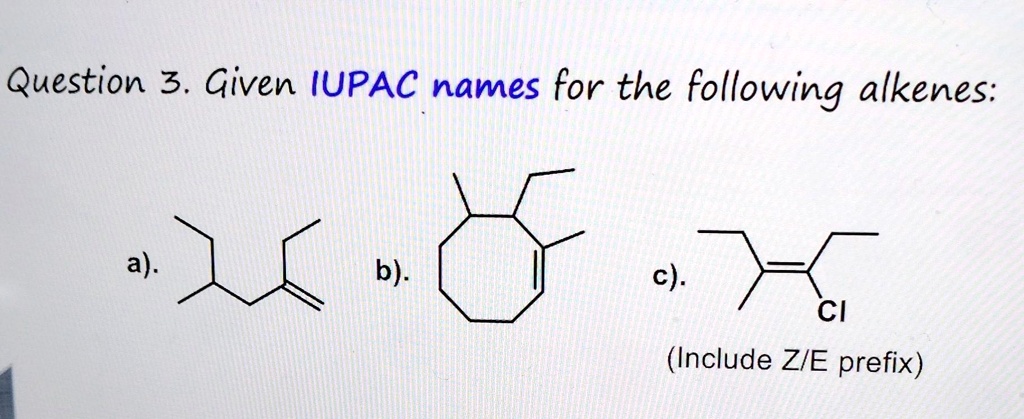
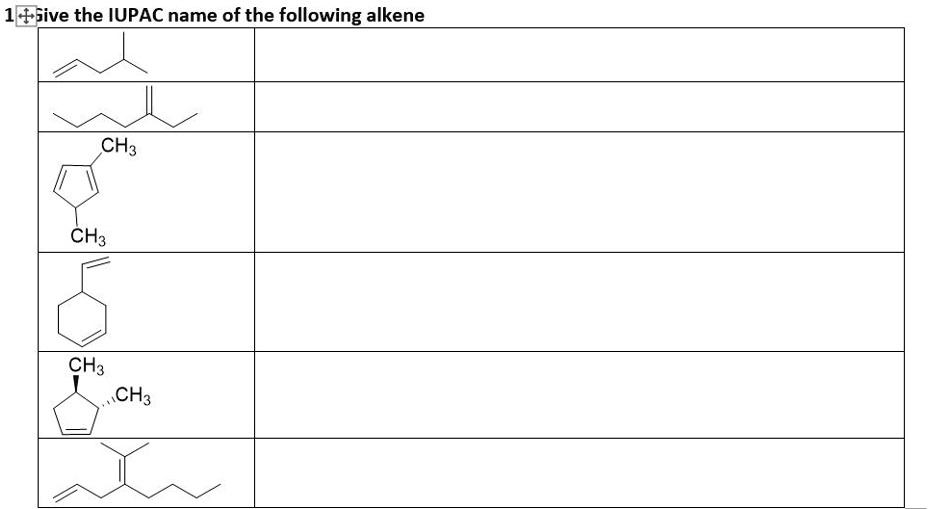
Give The Iupac Name For The Following Alkene
Ever looked at a chemical name and thought, "That looks complicated!"? Well, get ready to unlock a little bit of the magic behind those seemingly daunting labels. Today, we're diving into the fun world of naming alkenes, a group of molecules that are a big deal in everything from the food we eat to the plastics we use. It's like cracking a secret code, and once you know the basics, it becomes surprisingly easy and even rewarding!
So, why is learning to name alkenes a cool thing to do? Think of it as a superpower for understanding the building blocks of our world. For beginners, it’s a fantastic introduction to the systematic way scientists communicate. It builds a foundation for understanding more complex chemistry without feeling overwhelmed. For families looking for a fun educational activity, it can be a great way to explore science together. Imagine a little chemistry detective game where you're all solving puzzles! And for hobbyists, whether you're into cooking, gardening, or even DIY projects, a basic understanding of these molecular names can offer fascinating insights into how different substances behave and interact.
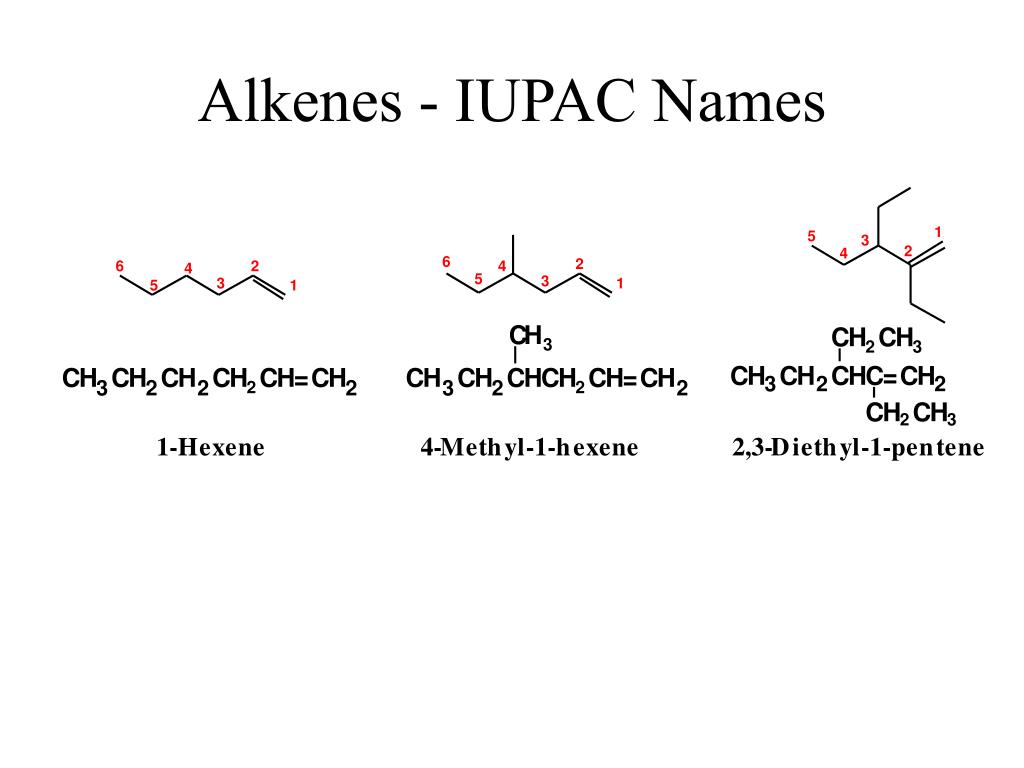
Alkenes are characterized by a double bond between two carbon atoms. This double bond is the key feature that dictates their naming. The International Union of Pure and Applied Chemistry, or IUPAC, has a set of rules that ensures every molecule has a unique and unambiguous name. This is crucial for scientists worldwide to be on the same page. For instance, a simple alkene like ethene (which has two carbons with a double bond) is the basis for polyethylene, a plastic found in countless everyday items. Understanding its name helps you appreciate the origin of those materials!
Must Read
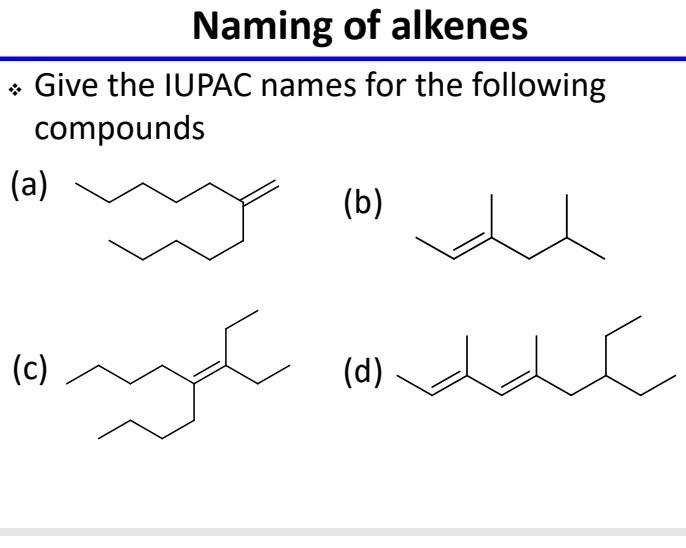
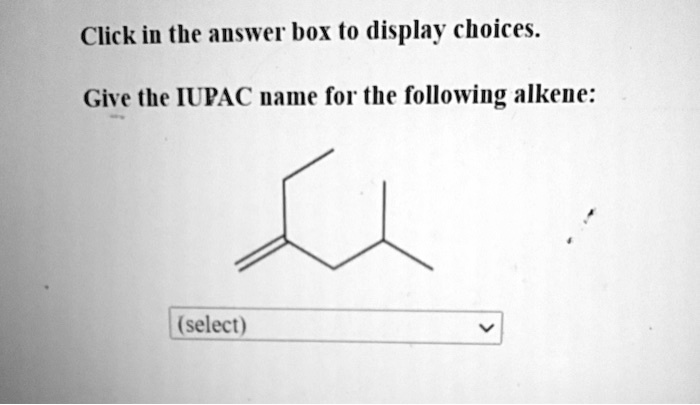
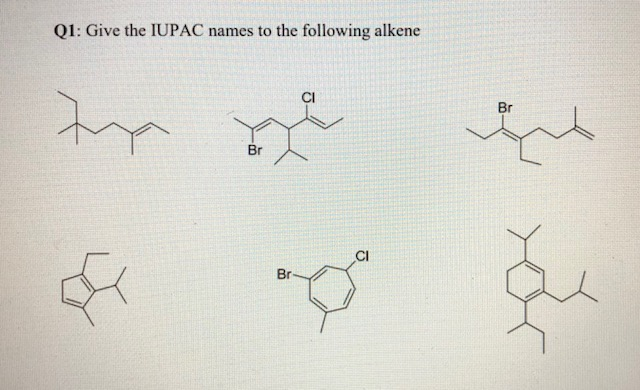
Let's look at a simple example to get you started. Imagine a molecule with three carbon atoms, and somewhere in the chain, there's that tell-tale double bond. Following IUPAC rules, we first identify the longest carbon chain containing the double bond, which in this case would be three carbons, giving us the "prop" prefix. Then, we identify the position of the double bond. If the double bond is between the first and second carbon, we'd call it prop-1-ene. If it's between the second and third carbon (which is the same as the first and second in a three-carbon chain, just counted from the other end), it would still be prop-1-ene. The key is to number the carbons so the double bond gets the lowest possible number. For a molecule with four carbon atoms and a double bond, it could be but-1-ene or but-2-ene, depending on where that double bond sits.
Getting started is easier than you might think! Tip number one: Familiarize yourself with the basic prefixes for the number of carbon atoms: meth- (1), eth- (2), prop- (3), but- (4), pent- (5), and so on. Tip number two: Remember that alkenes end in "-ene". Tip number three: The most important part is locating that double bond and numbering the carbon chain so that the double bond has the smallest number. Don't be afraid to draw it out! Visualizing the molecule makes naming so much simpler.
So, there you have it! Learning to give IUPAC names to alkenes is a fun and practical skill. It demystifies chemical nomenclature and offers a glimpse into the organized world of molecular science. It's a small step that opens up a bigger understanding, making the world around you just a little bit more fascinating.