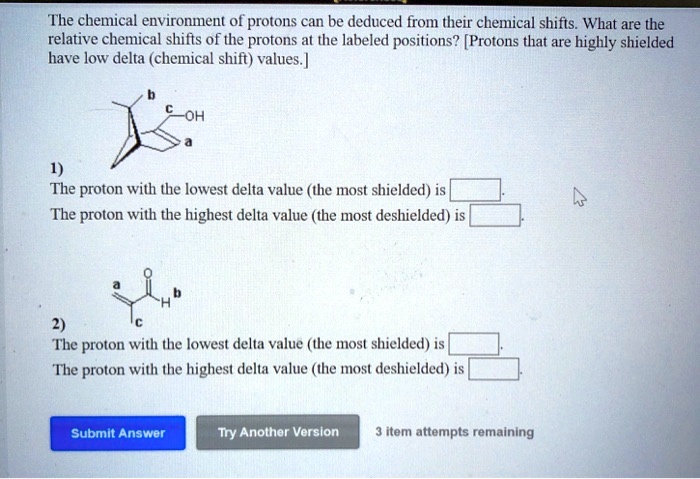
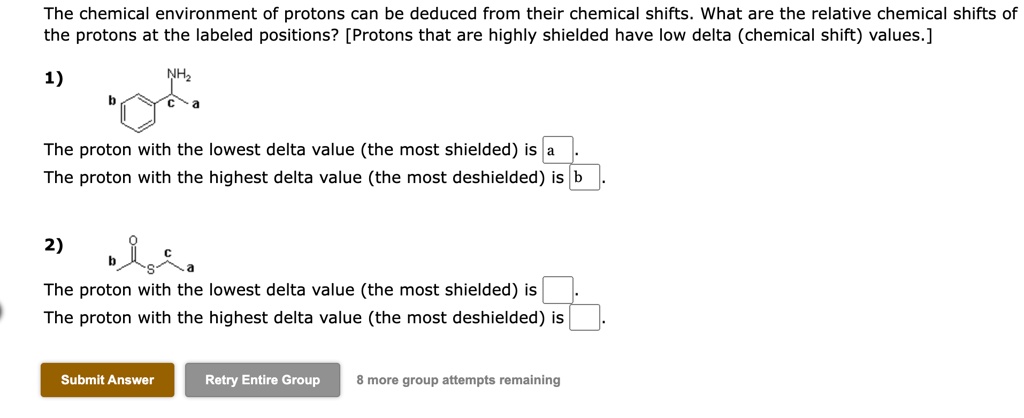
For Chemical Shifts Of The Protons Labeled

Ever found yourself staring at a complex molecule, perhaps doodling it absentmindedly on a napkin during a particularly inspired coffee break, and wondered what makes each tiny proton within it tick? It's a question that might sound super niche, even a little intimidating, but trust us, understanding the "chemical shifts" of these protons is actually a bit like cracking the code to their personalities. Think of it as eavesdropping on their private conversations, learning about their neighbors, and even their general vibe in the molecular world. And the best part? This isn't just for lab coat enthusiasts; it's a fascinating peek into the building blocks of everything around us, from your morning smoothie to that iconic shade of blue in Picasso's Guernica.
We're talking about Nuclear Magnetic Resonance, or NMR spectroscopy, a superhero of analytical chemistry. It's the technique that lets scientists see these incredibly detailed molecular portraits. And the star of the show? The humble proton. These little guys, positively charged and buzzing with energy, are everywhere in organic molecules. But they aren't all created equal, and that's where the magic of chemical shift comes in. It’s essentially a measurement of how much a proton’s signal is shifted or deshielded from a reference point. Why does this happen? Because protons don't exist in a vacuum. They're constantly interacting with their molecular environment, and especially with the electrons that surround them.
Imagine our protons are like people at a bustling party. Some are chilling in quiet corners, surrounded by their close pals (electron density), feeling pretty relaxed and unbothered. Others are right in the thick of it, chatting with people from different social circles, perhaps near someone a bit loud or flashy (electronegative atoms). These latter protons are going to feel a bit more exposed, a little more changed by the interactions. This change in their "electromagnetic atmosphere" is what we call the chemical shift.
Must Read
The "labeled" part in our headline is key. It means we're specifically pointing to protons that are attached to certain atoms or in particular chemical environments. So, when we talk about the chemical shifts of labeled protons, we’re being super precise. We're not just talking about any random proton; we're focusing on the ones that give us the most diagnostic information. It's like having a guest list and knowing exactly who you're looking for at that party – maybe you’re trying to find the quiet artist or the life of the party.
The Electron Shield: Our Proton's Personal Space Bubble
Let's dive a little deeper into this "shielding" concept. Protons are often bonded to carbon atoms, which are the backbone of organic chemistry, the stuff of life itself. But these protons can also be near other atoms, like oxygen, nitrogen, or halogens (think chlorine or bromine). These elements are known as electronegative. In simpler terms, they have a strong appetite for electrons. When a proton is near an electronegative atom, the electrons tend to flock towards that atom, leaving the proton feeling a bit less "electron-rich."
This withdrawal of electron density is what causes the proton to become deshielded. Think of it as the electron cloud around the proton shrinking. In the presence of a strong external magnetic field, this reduced electron cloud means the proton is more exposed to the field. This makes it easier to flip its magnetic spin, and it requires a slightly higher frequency of radio waves to do so. This higher frequency corresponds to a larger chemical shift value. Confusing? Not really, once you get the hang of it. It’s like needing a louder shout to get someone's attention when there's a lot of background noise.
Conversely, if a proton is surrounded by lots of electron density, perhaps in a very saturated hydrocarbon chain with no electronegative neighbors, it's considered shielded. The electron cloud is robust, acting like a cozy blanket. This shielding reduces the proton's exposure to the external magnetic field, meaning it requires a lower frequency of radio waves to flip its spin. This translates to a smaller chemical shift value. Easy peasy, right? It’s the difference between a whisper in a quiet library and a shout across a stadium.
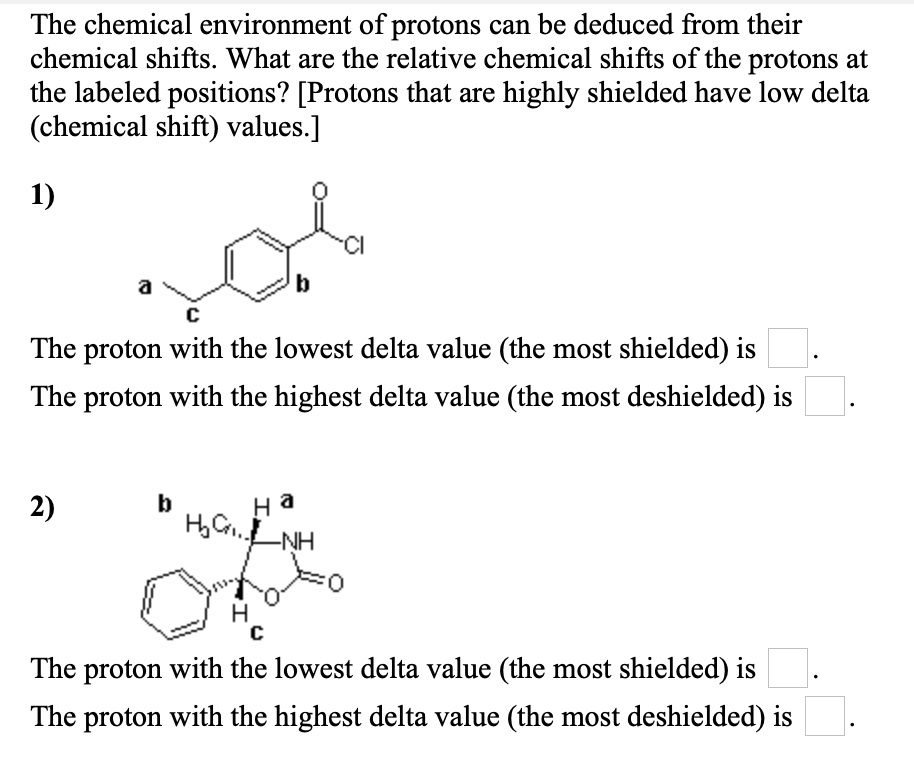
Decoding the Numbers: The Delta Scale
So, how do we quantify this? Scientists use a scale called the delta (δ) scale, measured in parts per million (ppm). This scale is conveniently normalized, meaning it's independent of the specific magnetic field strength of the NMR instrument. This makes the data comparable across different labs and machines.
On this scale, highly shielded protons appear at lower delta values, often close to 0 ppm. The quintessential example of a highly shielded proton is one in a simple alkane, like methane (CH4). Its protons are generally found around 0.8-0.9 ppm. Now, consider something like water (H2O). The protons in water are highly influenced by the electronegative oxygen atom, making them much more deshielded. Their signal typically appears around 1.5-5.0 ppm, depending on factors like concentration and temperature. Pretty neat how a simple molecule can have such a different signature!
On the other hand, highly deshielded protons appear at higher delta values. Protons directly attached to electronegative atoms, or those in regions of high electron deficiency, will show up further downfield. For instance, protons in a carboxylic acid group (-COOH) are very deshielded due to the adjacent oxygen atoms and are typically found in the range of 10-13 ppm. That's a huge jump from our simple alkane proton!
Think of the delta scale as a timeline of proton "stress levels." Lower numbers mean a chill, protected proton; higher numbers mean a proton that’s feeling the heat, so to speak. This simple numbering system is the bedrock of interpreting NMR spectra.

Context is Key: What's Around Our Proton?
It's not just the immediate atom a proton is attached to that dictates its shift. The entire molecular neighborhood plays a role. A proton attached to a carbon atom might have a different shift if that carbon is part of a double bond (an alkene) versus a single bond. The electrons in a double bond are more dispersed and can influence nearby protons differently than those in a single bond.
For example, protons attached to sp2 hybridized carbons (like those in alkenes, C=C) are generally more deshielded than those attached to sp3 hybridized carbons (like those in alkanes, C-C). This is because the electrons in the pi system of the double bond are more easily influenced by the external magnetic field, creating a deshielding effect. So, a proton in ethylene (C2H4) typically appears around 5.65 ppm. That's a significant shift from our alkane proton!
Then we have aromatic rings, like the benzene ring, which is a staple in many organic molecules. The delocalized pi electrons in an aromatic ring create a special magnetic field. Protons attached to these rings are highly deshielded, often appearing in the range of 7-8 ppm. This is a really diagnostic signal! It's like recognizing a specific type of music; you just know it's rock and roll when you hear those aromatic protons.
Consider the difference between a simple methyl group (-CH3) attached to a saturated carbon, which might be around 0.9 ppm, and a methyl group attached to an aromatic ring, which could be around 2.3 ppm. That's a substantial shift, telling us the proton's environment has changed dramatically. It's these subtle, yet significant, shifts that allow chemists to piece together complex molecular structures.

Fun Facts and Cultural Touches
Did you know that the discovery of NMR actually led to Nobel Prizes for multiple scientists? Felix Bloch and Edward Purcell were awarded the Nobel Prize in Physics in 1952 for their work on nuclear magnetic resonance. Later, in 1991, Richard Ernst won the Nobel Prize in Chemistry for his development of high-resolution NMR spectroscopy, revolutionizing how we study molecules. It’s a testament to how fundamental this technique is!
Think about how much of our modern world relies on understanding these molecular interactions. Pharmaceuticals, new materials, even food science – all these fields benefit immensely from NMR. The precise identification of drug molecules, the characterization of novel polymers, or ensuring the authenticity of food products all depend on the information gleaned from chemical shifts.
It’s like learning a new language, but instead of words, we're learning the "words" that protons speak through their chemical shifts. Each shift is a clue, a piece of a larger puzzle. And once you learn the basic vocabulary – the typical ranges for different functional groups and environments – you can start to "read" molecules.
Ever admired a beautifully designed piece of jewelry or a intricate piece of architecture? The stability and form of those structures, at the most fundamental level, are governed by the way atoms, and their protons, interact. Chemical shifts are a window into that incredibly precise, atomic-level arrangement that gives things their form and function.
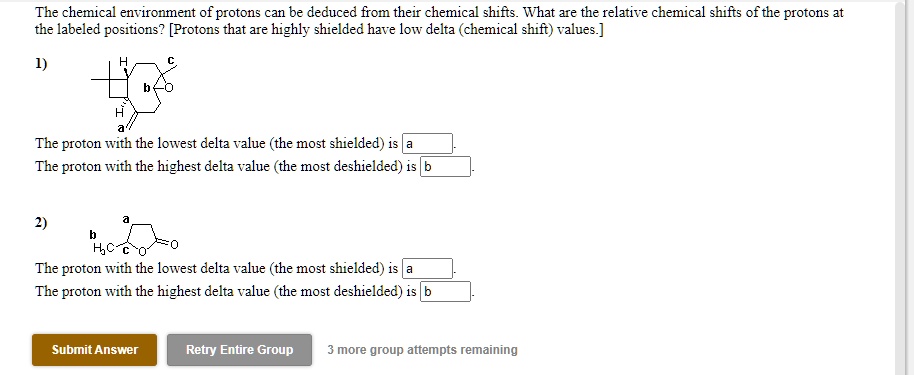
And for you pop culture buffs, the ability to determine molecular structures using NMR is so powerful that it’s often portrayed in detective shows or scientific dramas. It’s the ultimate forensic tool for chemists, allowing them to identify unknown substances or confirm the composition of complex mixtures. It’s less about dramatic car chases and more about the quiet, intense deduction in a lab!
Putting It All Together: Practical Tips for the Curious Mind
If you’re a student delving into chemistry, here are some friendly tips for navigating chemical shifts:
- Get a Reference Chart: The most important tool is a chemical shift chart. These charts list the typical ppm ranges for protons in various common functional groups and environments. Keep one handy, almost like a cheat sheet for proton personalities!
- Start Simple: Don't try to tackle a complex natural product on day one. Begin with simple molecules like ethanol, acetone, or ethyl acetate. These have distinct and well-understood proton signals that will help you build your understanding.
- Look for Patterns: Notice how protons in similar environments tend to have similar shifts. For example, all the methyl protons in a series of similar alkanes will be grouped together.
- Consider Electronegativity: Always ask yourself: "What electronegative atoms are near this proton?" This is your biggest clue to whether it will be shielded or deshielded.
- Think about Double/Triple Bonds and Aromatic Rings: These features significantly impact electron density and therefore chemical shifts. Protons near them will almost always be found at higher delta values.
- Don't Forget Integration: While chemical shifts tell us what kind of proton it is, the integration (the area under the signal) tells us how many protons are giving that signal. This is crucial for determining the number of protons in each environment.
- Practice, Practice, Practice: Like learning any new skill, the more NMR spectra you look at and interpret, the more intuitive it becomes.
Even if you’re not a chemist, understanding this concept can give you a new appreciation for the intricate world of molecules. The next time you’re enjoying a piece of fruit, marveling at the vibrant colors of a flower, or even just taking a deep breath of air, remember that these phenomena are all governed by the precise interactions of atoms and their protons, with their chemical shifts telling a subtle yet powerful story.
Beyond the Lab: A Daily Reflection
It’s fascinating how these tiny, unseen forces dictate so much of our reality. The chemical shift, a concept born in the precise world of spectroscopy, ultimately speaks to a universal truth: everything is influenced by its surroundings. Just like us, protons are not isolated entities. Their behavior, their "signal" in the molecular world, is a direct reflection of their interactions with others. The electron density around them, the nearby atoms, the overall molecular architecture – all contribute to their unique chemical signature.
In our own lives, we too are shaped by our environments. The people we surround ourselves with, the cultures we immerse ourselves in, the challenges we face – they all influence our perspectives, our actions, and ultimately, who we become. We are constantly interacting, constantly adjusting. Sometimes we are shielded, finding comfort and stability in familiar surroundings. Other times, we are pushed into new, more challenging situations that "deshield" us, forcing us to grow and adapt in ways we never expected. The chemical shift, in its own quiet, scientific way, is a beautiful metaphor for this constant dance of influence and interaction that defines both the molecular world and our own human experience. It’s a reminder that even in the smallest building blocks of existence, connection and context are everything.