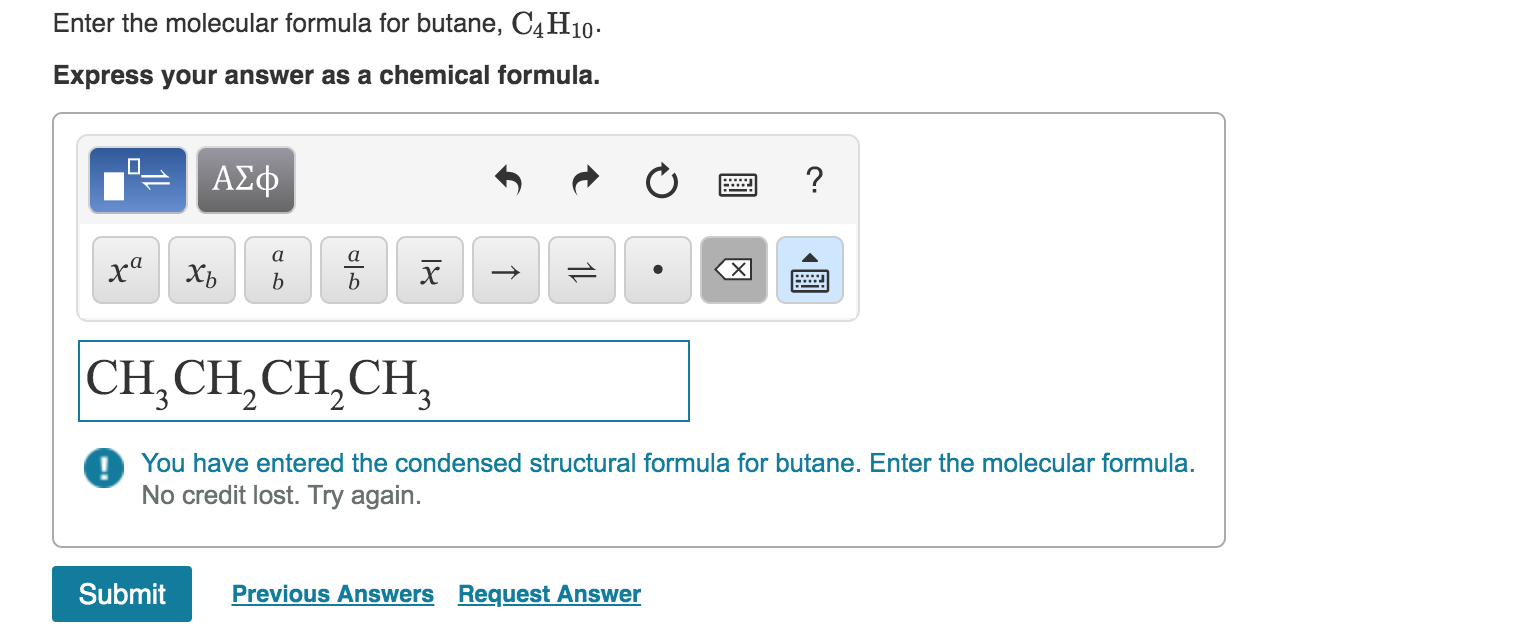
Express Your Answer As A Chemical Formula
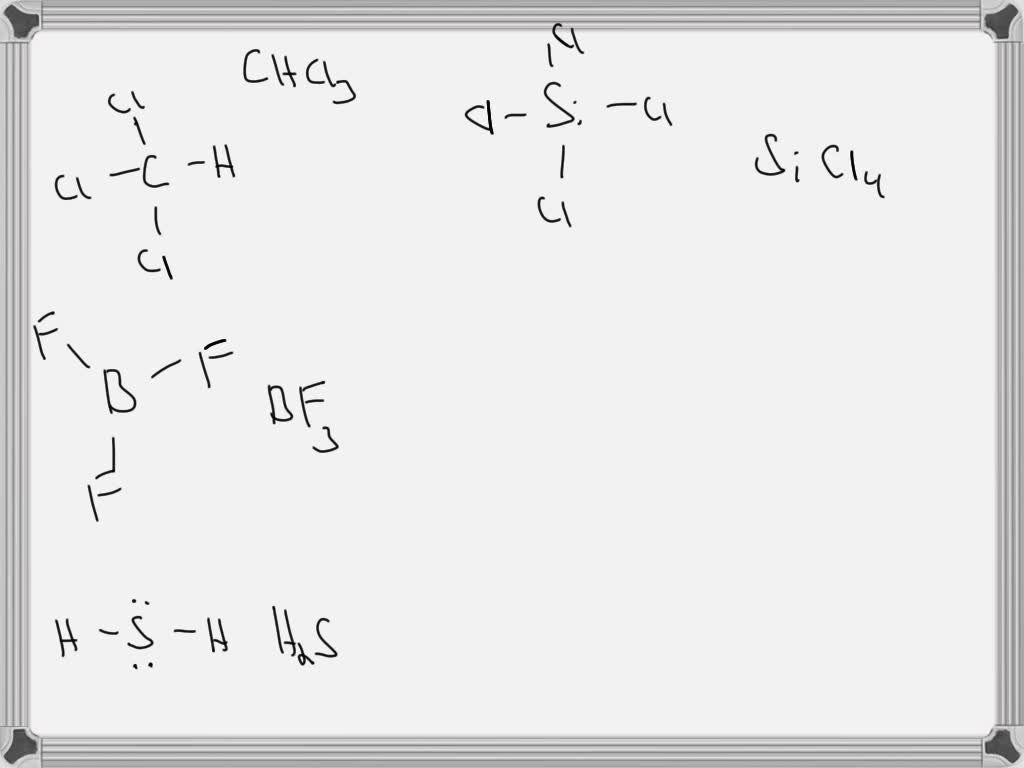
Alright, settle in, grab your imaginary latte, and let's talk about something that sounds terrifying but is actually, dare I say, fun? We're diving headfirst into the wild world of chemical formulas, but don't freak out! Think of me as your friendly neighborhood chemist, minus the intimidating lab coat and the slightly unsettling aroma of unknown experiments. My mission today is to convince you that these cryptic little collections of letters and numbers are, in fact, the secret handshake of the universe.
You see, everything around you, from the air you're breathing (which is, by the way, a delightful cocktail of nitrogen and oxygen, mostly) to the suspiciously delicious donut you're probably craving right now, is made of tiny, invisible building blocks called atoms. And these atoms? They love to hang out together, forming little molecular families. A chemical formula is basically the family portrait of these molecules, telling you exactly who's in the gang and how many of each are present.
Take water, for instance. Everyone knows water, right? H2O. Seems simple enough. But oh, the stories that little H2O can tell! It means you've got two hydrogen atoms (H) doing a little jig with one oxygen atom (O). They're basically inseparable, like a celebrity couple you can't get enough of. And here's a mind-blower: if you change just one little thing, say, swap out one hydrogen for a different atom, you don't get water anymore. You might get something that smells like rotten eggs (sulfur dioxide, anyone? No thanks!). It’s a stark reminder that sometimes, the smallest changes have the biggest impact. Like forgetting your keys. Or wearing two different socks.
Must Read
So, how do these formulas work? It's like a super-efficient shorthand. The letters, usually capitalized, represent the element symbols. Think of them as the first initial of each atom's name. Hydrogen is H, Oxygen is O, Carbon is C (for "Carbon-tastic!"). Then, you have the numbers, called subscripts. These little fellas, chilling down low to the right of the symbol, tell you how many atoms of that element are in the molecule. If there's no number, it's like saying "one." So, O is just one oxygen atom. But H2? That's two hydrogen atoms, ready to party!
The Drama of Molecules: More Than Just Pretty Letters
Honestly, these formulas are the ultimate gossip columns of the chemical world. They reveal who's bonding with whom, and why. Take table salt, your everyday NaCl. It's a sodium atom (Na) and a chlorine atom (Cl) holding hands. Sounds innocent, right? But the story behind their bond is fascinating! Sodium is a super reactive metal that would happily explode if you splashed water on it (don't try this at home, seriously, the firefighters will not appreciate it). Chlorine? Well, it's a toxic gas that’s used in swimming pools to keep things… hygienic. Yet, when they get together, forming that ionic bond? Boom! You get something totally edible and, dare I say, delicious. It’s like the universe saying, “See? Even the most unlikely pairings can create something wonderful!”

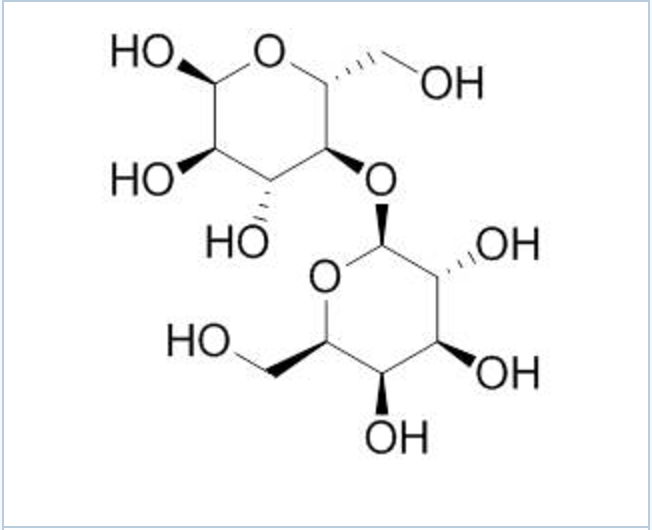
And don't even get me started on carbon. This guy is the rockstar of the periodic table. He can form four bonds, which is like being able to juggle four flaming torches while riding a unicycle. This ability allows carbon to create an almost infinite number of complex molecules. Think of all the amazing things it builds: the DNA in your cells, the sugar in your coffee, the plastic in your phone, and, yes, the aforementioned donut. The formula for glucose, a type of sugar, is C6H12O6. That's 6 carbons, 12 hydrogens, and 6 oxygens, all chained together in a beautiful, energy-giving structure. It’s like a microscopic LEGO set, but instead of plastic bricks, you’ve got atoms snapping into place.
When Formulas Get Fancy: It's Not Always Just Simple Families
Sometimes, the formulas get a little more complicated, and this is where things get really interesting. You'll see numbers in parentheses, like in Calcium Hydroxide: Ca(OH)2. What's going on here? It's like a mini-family within the larger family. The parentheses mean that whatever is inside is grouped together, and the subscript outside applies to the entire group. So, in Ca(OH)2, you have one calcium atom (Ca) and two hydroxide groups (OH). Each hydroxide group consists of one oxygen (O) and one hydrogen (H). So, in total, you’ve got one Ca, two Os, and two Hs. It’s a bit like a recipe: you need two scoops of the "hydroxide" mixture for every one scoop of calcium. It’s all about organization!
And then there are the acids and bases. Ever heard of hydrochloric acid (HCl)? That's just one hydrogen and one chlorine. Strong stuff. Then there's sulfuric acid, H2SO4. That's two hydrogens, one sulfur, and four oxygens. It's a bit more complex, and let me tell you, it means business. These formulas are crucial for understanding how things react. They're the blueprints for all the chemical reactions happening around us, from your body digesting lunch to a volcano erupting (which, by the way, involves a whole lot of fancy chemistry!).
The amazing thing is, these formulas are universal. A chemist in Tokyo and a chemist in Timbuktu will look at H2O and understand exactly the same thing. It's a language that transcends borders and cultures. It’s the universal instruction manual for matter. So, the next time you see a chemical formula, don't run for the hills. Take a moment, appreciate the elegant simplicity, and remember that you're looking at a tiny, perfectly crafted snapshot of the world's most fundamental building blocks. They're not just letters and numbers; they're the whispers of the universe, telling us the stories of everything.