Explain Why Potassium Is More Reactive Than Sodium

Ever wondered why some things are just… more energetic than others? It’s a bit like comparing a sleepy cat to a puppy on a sugar rush. When it comes to the elements, those building blocks of everything around us, some are definitely more prone to getting excited. Today, we're going to talk about two such characters: sodium and potassium. They’re both in the same family, you see, kind of like cousins. But one of them, potassium, is famously a lot more of a firecracker than the other, sodium.
Think of them as two buddies who love to make friends. The way they make friends is by giving away one of their outer electrons. It’s like they have a little toy they’re really happy to share to make a new connection. But the urge to share that toy is where the difference lies. Potassium just can't wait to get rid of its electron, while sodium is a little more, shall we say, reserved about it.
It’s all about how badly they want to be complete. Like us, atoms like to feel a sense of stability. They achieve this by having a full outer shell of electrons. For sodium and potassium, that means getting rid of that single electron hanging out on the edge.
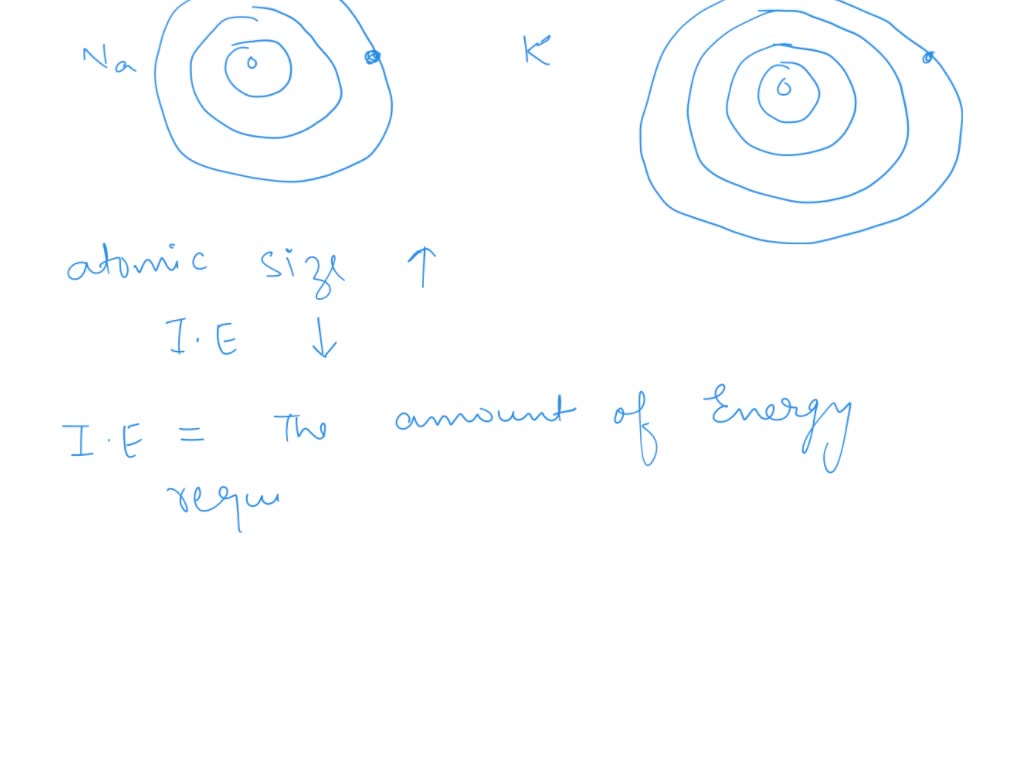
So, why is potassium so much more eager? It’s all about its size and the distance of that outermost electron from the center of the atom, the nucleus. Imagine the nucleus as a tiny, powerful magnet. The electrons are like little metal filings swirling around it. The further away a filing is from the magnet, the weaker the pull. Potassium atoms are bigger than sodium atoms. They have more layers of electrons tucked away inside, like those Russian nesting dolls.
This extra bulk means that potassium's outermost electron is much further away from the nucleus’s attractive grip. It’s like that metal filing is on the very edge of the room, while sodium’s electron is closer to the magnet. Because of this greater distance, the nucleus has a much weaker hold on potassium's lonely outer electron. It’s just not as strongly attached. This makes it super easy for potassium to say "Peace out!" to that electron and go off to find a new friend.

Sodium, on the other hand, is a bit smaller. Its outer electron is closer to its nucleus. The magnetic pull is stronger, so it holds onto that electron a little more tightly. It’s still willing to share, don’t get me wrong! Sodium is very reactive. But potassium? It’s on a whole other level of enthusiasm.
This difference in how tightly they hold onto their electrons is what makes potassium more reactive. It’s like a race. Potassium is already at the starting line, practically vibrating with excitement, ready to bolt. Sodium is there too, but it needs a little more of a nudge to get going.
When these elements meet other things, especially things that want electrons (like oxygen or chlorine), this eagerness really shows. Potassium is practically leaping into action, ready to hand over its electron and form a new compound. It reacts much faster and more vigorously.
This is why you often see demonstrations where sodium is dropped into water, and it fizzes and melts, sometimes even igniting. It’s quite a show! But if you do the same with potassium, it’s a whole different story. Potassium reacts explosively with water. It’s a much more dramatic, fiery display. It’s like the difference between a pop and a bang!
Think about it: sodium is the stuff in table salt, which is pretty stable and safe. We eat it every day! Potassium is also essential for our bodies, but in its pure elemental form, it's something you’d only encounter in a controlled science lab because it's just so eager to react.

The reactivity of these elements is a fundamental concept in chemistry, and it's a fantastic example of how subtle differences in atomic structure can lead to big differences in behavior. It’s the reason why we have different materials with vastly different properties. One atom being just a smidge larger can mean a world of difference in how it interacts with the world.
It’s this inherent drive, this almost impatient desire to connect and form new bonds, that makes potassium such a fascinating element to study. It’s a little rebel, a tiny powerhouse that’s always ready for action. And that, my friends, is why potassium is more reactive than its slightly more laid-back cousin, sodium. It’s a tale of atomic size, electron distance, and a whole lot of chemical enthusiasm!

![[ANSWERED] Sodium and potassium react with water much more vigorously](https://media.kunduz.com/media/sug-question-candidate/20210419084410741970-2943061.jpg?h=512)
