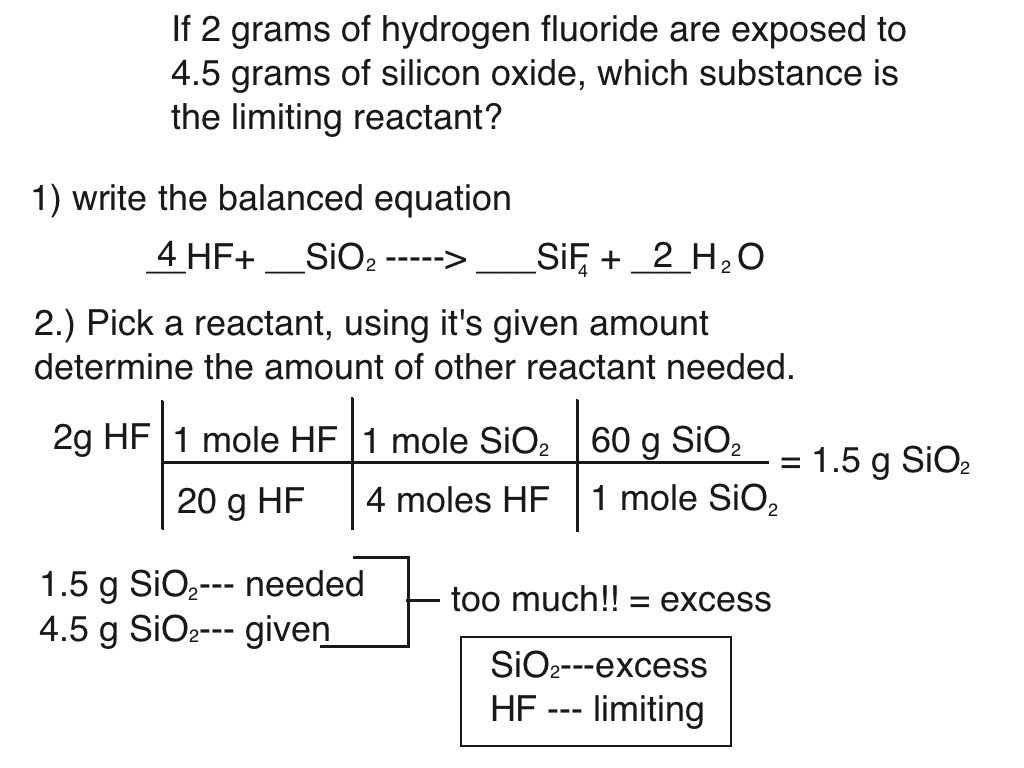
Experiment 8 Limiting Reactant Lab Report

Hey there, science curious cats! Ever feel like you're juggling way too many ingredients when you're trying to bake the perfect cookie? You know, you've got your flour, your sugar, your chocolate chips... and then BAM! You realize you’re totally out of eggs. Suddenly, your cookie dreams are dashed. Well, guess what? Chemistry has a super similar, and honestly, way more exciting, problem. It’s called the limiting reactant. And we totally tackled it in our latest lab, Experiment 8. Buckle up, buttercups, because this is gonna be fun!
So, what's a limiting reactant? Think of it as the "not enough stuff" ingredient. It's the thing that runs out first in a chemical reaction, like our missing eggs in the cookie dough. Once the limiting reactant is gone, poof! The reaction stops. No more product. Bummer, right? But also, super useful to know!
Our lab was all about figuring out which ingredient was gonna be our little chemistry party pooper. We weren't making cookies, sadly. We were doing something way cooler: making a precipitate. Imagine tiny, solid particles forming out of thin air, or rather, out of two clear liquids. That’s a precipitate! It’s like magic, but with science.
Must Read
We mixed two solutions together. One had calcium chloride in it. Think of it as the building blocks for our solid stuff. The other solution had sodium carbonate. More building blocks! When you mix them, they decided to get together and form calcium carbonate. That's the solid stuff, the precipitate. It looks like a cloudy haze, or sometimes even like fine white dust settling at the bottom. Pretty neat, huh?
The Great Recipe Reveal (of sorts!)
The tricky part, and where our limiting reactant detective work came in, was that we didn't know exactly how much of each thing would react. We had specific amounts of calcium chloride and sodium carbonate. We added them, stirred them up, and watched the magic happen. But was there too much calcium chloride? Or was the sodium carbonate the real star of the show, the one that would disappear first?
Our mission, should we choose to accept it (and we totally did, armed with pipettes and safety goggles!), was to figure out which one was the limiting reactant. It's like being a culinary detective. You look at your recipe, you look at your ingredients, and you figure out what's going to run out first. Is it the sprinkles? The frosting? The actual cake mix?

The fun part is that the limiting reactant dictates how much product we can actually make. If you only have enough flour for two cookies, it doesn't matter if you have a whole vat of chocolate chips. You're only making two cookies. Get it? The limiting reactant is the ultimate cookie-count controller.
The Ominous "Excess Reactant"
And what about the stuff that's left over? The ingredients that didn't get used up because their partner ingredient ran out? Those are called excess reactants. Imagine you have enough eggs for 50 cookies, but only enough flour for 10. The flour is your limiting reactant. The eggs? They're your excess reactants. They're just chilling, waiting for more flour that never arrives.
In our lab, we were looking for evidence of these leftover guys. Did we have calcium chloride floating around, looking sad and unused? Or was it the sodium carbonate that was left behind? Finding the excess reactant is a HUGE clue to identifying the limiting one. It's like finding crumbs on the floor after a cookie heist – you know something was there.
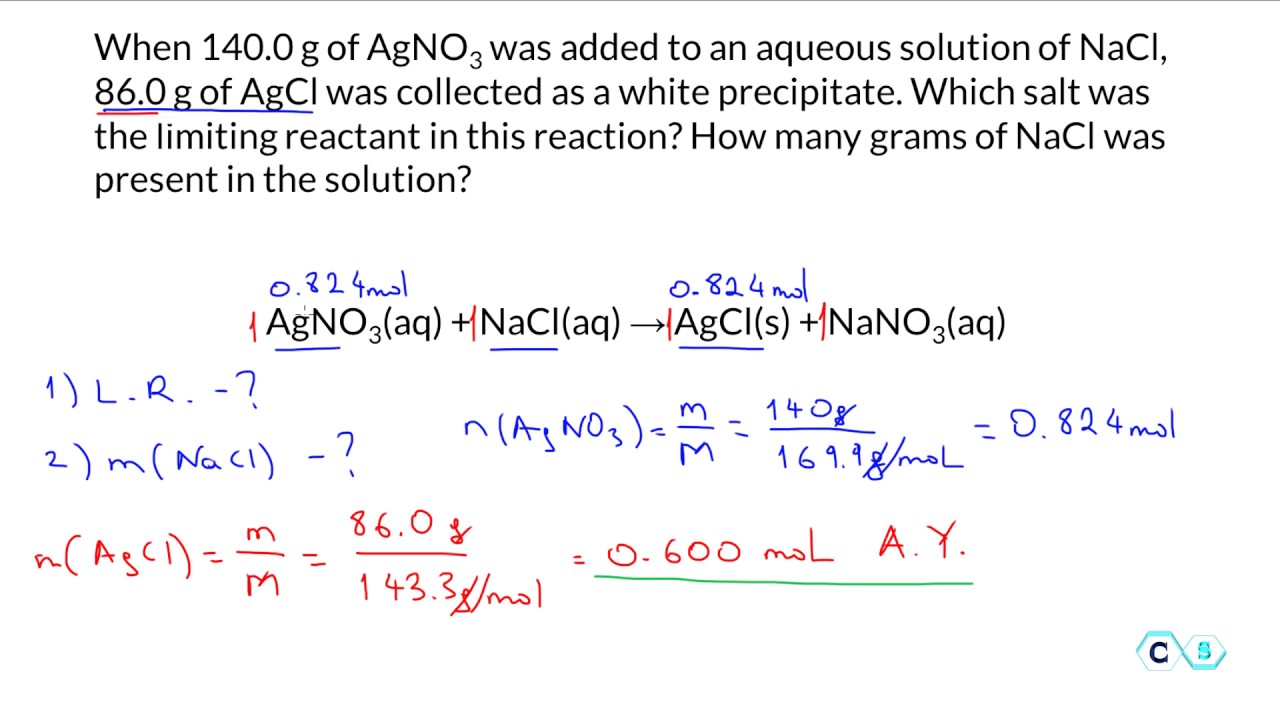
Our lab report was our chance to show off our detective skills. We had to do calculations. Lots of them. It involved something called stoichiometry. Don't let the fancy word scare you! It's just the science of how much stuff reacts with how much other stuff. Think of it as the chemical rulebook for proportions. It tells you the perfect ratio for your ingredients.
We calculated the theoretical yield. That's the maximum amount of product we should have made if everything went perfectly and we used up all of our limiting reactant. Then, we actually weighed the precipitate we collected. This was our actual yield. Comparing the two is where the real fun begins.
The Percent Yield Shenanigans
The difference between theoretical and actual yield is a big deal in chemistry. Why don't we always get 100%? Well, in the real world (and in the lab!), things aren't always perfect. Some of the precipitate might have stuck to the beaker. Some might have dissolved back into the solution (gasp!). Or maybe, just maybe, our measurements weren't exactly spot on. These little imperfections are why we calculate the percent yield. It's basically saying, "Okay, here's how much we should have made, and here's how much we actually made. What percentage of the dream did we achieve?"

This is where the quirky facts and funny details really come in. Sometimes, you get a surprisingly high percent yield. You're like, "Whoa! Did we accidentally discover a new super-efficient reaction?!" Other times, you get a ridiculously low one, and you're scratching your head, wondering if a tiny chemical gremlin snuck into your beaker. It’s all part of the adventure!
For Experiment 8, we had to meticulously record our data. The mass of the reactants we started with. The mass of the precipitate we collected. And then, the glorious calculations to figure out our limiting and excess reactants. It’s like putting together a puzzle. Each piece of data is a clue, and when you put them all together, you get the answer!
One of the funniest things about these labs is how seriously we take them, and then how we inevitably spill something or forget a crucial step and have to start all over. But that’s the beauty of science, right? It’s messy, it’s experimental, and it’s always teaching us something new. Even if that something new is that calcium chloride and sodium carbonate make a fantastic white, cloudy mess.

So, why is this limiting reactant thing so fun to talk about? Because it’s a real-world concept dressed up in science lab clothes! It applies to baking, to cooking, to manufacturing – basically, to anything where you have ingredients or resources that are combined to make something. Understanding limiting reactants helps us be more efficient, more precise, and honestly, less likely to run out of eggs when we’re really craving cookies.
Our lab report was our final say on the matter. We declared, with scientific certainty (and a few shaky calculations), which reactant was the boss of the reaction. We showed our work, explaining how we got there. It was a testament to our understanding of how chemicals play by the rules of proportion. It’s not just about mixing stuff; it’s about understanding the why and the how much.
And the satisfaction of figuring it all out? Priceless. It’s like solving a riddle, but with chemicals. Plus, it makes you appreciate all the careful planning that goes into making things we use every day. So next time you're baking, or even just eating a well-made meal, give a little nod to the limiting reactant. It’s the unsung hero (or villain!) of every chemical creation.