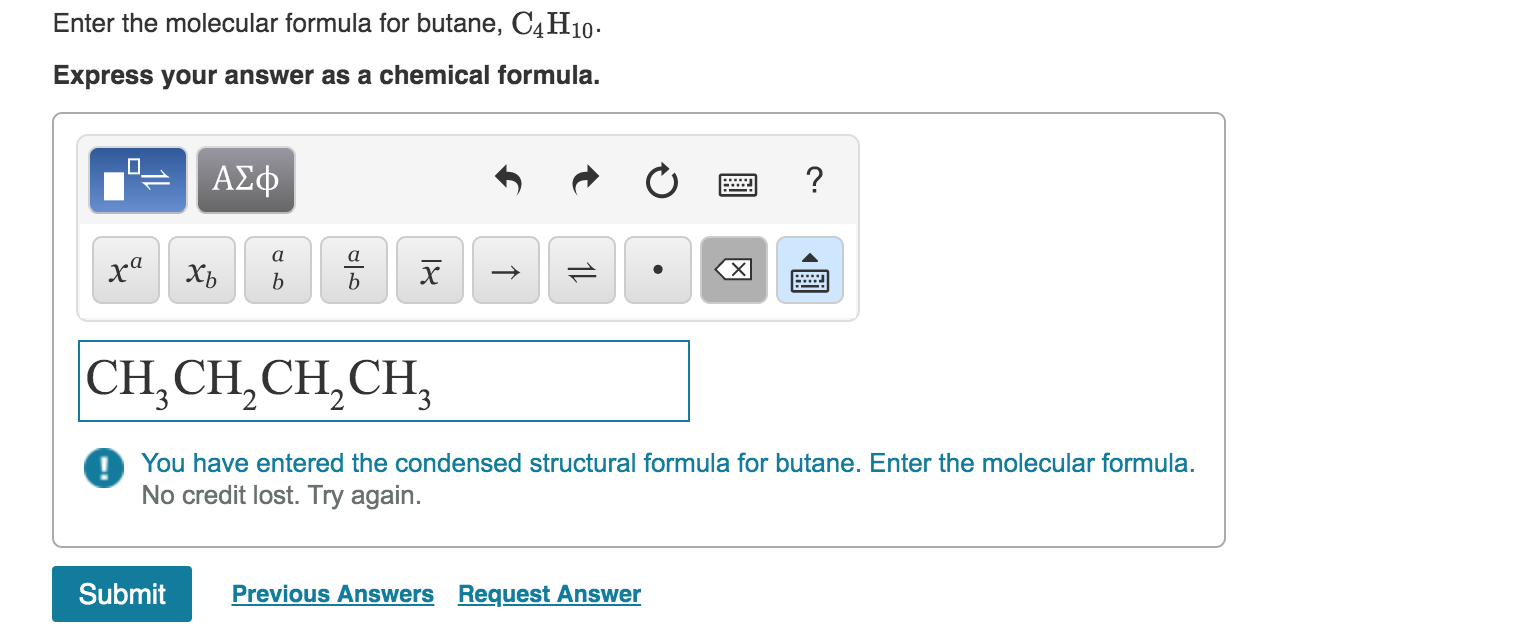
Enter The Molecular Formula For Butane C4h10
Get ready to meet a true superstar in the world of chemistry, folks! Today, we're diving headfirst into the fabulous and surprisingly familiar realm of butane. This isn't some stuffy, complicated science lesson; oh no, this is about uncovering a hidden hero that's probably been helping you out more than you realize. We're going to unlock its secret identity, and trust me, it's more exciting than a surprise party!
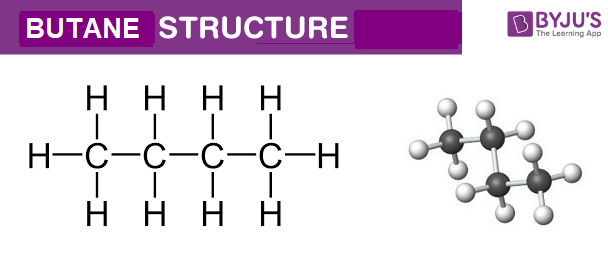
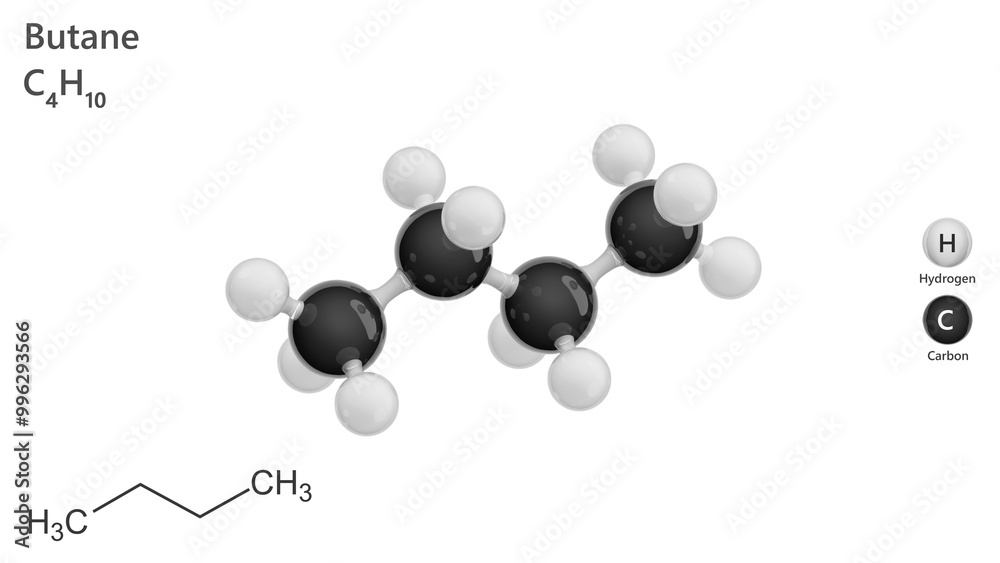
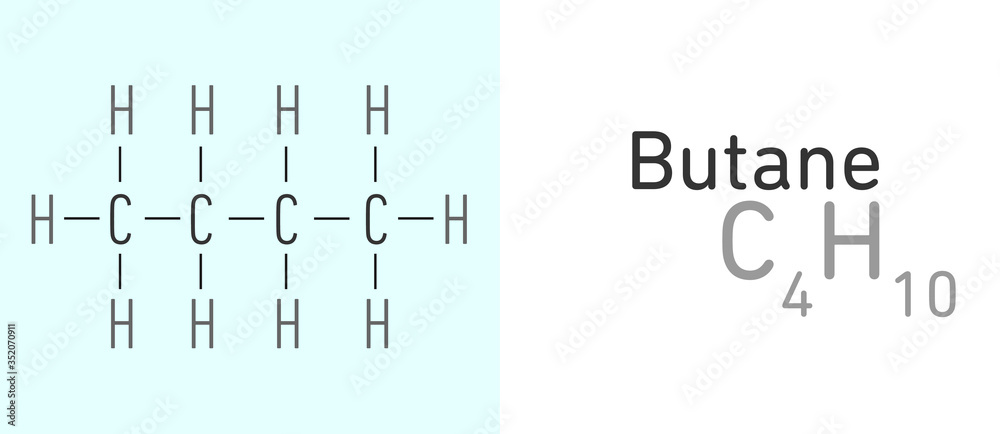
And the grand reveal for its official, super-secret molecular formula is none other than... wait for it... C4H10! Ta-da! It's like a tiny chemical code, a secret handshake that tells you exactly what this awesome molecule is made of. Think of it as its nutritional label, but way cooler and much more exciting. This little string of letters and numbers packs a punch!
So, what does this magical C4H10 actually mean? Well, it's pretty straightforward, even if it sounds a little like something from a sci-fi movie. The 'C' stands for carbon, the building block of all life as we know it. And the 'H'? That's for hydrogen, the most abundant element in the universe!
Must Read
Now, let's break down those numbers. The little '4' next to the 'C' tells us that in one molecule of butane, there are exactly four carbon atoms. Imagine them holding hands in a neat little chain. It's like a miniature carbon party, and everyone's invited!
And then we have the '10' next to the 'H'. This means for every four carbon atoms, there are a whopping ten hydrogen atoms. They're the energetic little sidekicks, hanging out with the carbon buddies, making everything stable and, well, useful! It’s a perfectly balanced partnership.
Think of it like this: if carbon atoms were comfy, sturdy little chairs, then butane would have four of them all lined up. And the hydrogen atoms? They're like the super-supportive cushions, making sure everything is just right. Ten cushions for four chairs – that's a lot of comfort and stability!
Now, you might be asking, "Where on earth do I encounter this butane in my everyday life?" Prepare to be amazed! This seemingly simple C4H10 molecule is a workhorse, a silent hero in so many of the things we take for granted. It’s practically everywhere, like a friendly ghost!
One of its most famous roles? It's the star of the show in your backyard barbecue! Yep, the fuel that ignites those glorious flames on your grill is often butane. So next time you're sizzling up some burgers, you can give a little nod to C4H10 for making it all possible. It's the unsung hero of summer cookouts!
And it doesn't stop at barbecue bliss. That handy-dandy lighter you use to ignite candles or start a campfire? Chances are, it's powered by butane! It’s the tiny engine that provides that instant spark of joy and warmth. So, thank butane for those perfectly lit birthday candles and cozy evening fires.
Ever used an aerosol can? Think hairspray, deodorant, or even spray paint. Inside those cans, butane often plays the role of a propellant. It's the invisible force that pushes the product out, making your hair fabulous or your walls look brand new. It’s the unsung push-power of convenience!
Butane is a type of alkane, which is a fancy word for a simple hydrocarbon. Hydrocarbons are molecules made up entirely of carbon and hydrogen atoms. They're like the foundational elements of the fuel world. And butane, with its specific C4H10 arrangement, is a particularly popular and useful one.
The beauty of butane lies in its versatility. It’s a gas at room temperature and pressure, which makes it incredibly convenient to store and transport. But when you put it under a little pressure, it easily becomes a liquid, packing more punch into a smaller space. It’s like magic, but it's just good ol' chemistry!
Think about how it’s stored in those small, portable gas canisters. That’s where the liquid state comes in handy, allowing for a lot of fuel to be condensed into a compact container. This makes it perfect for camping trips, portable stoves, and all sorts of on-the-go adventures. It's the ultimate travel-sized fuel!

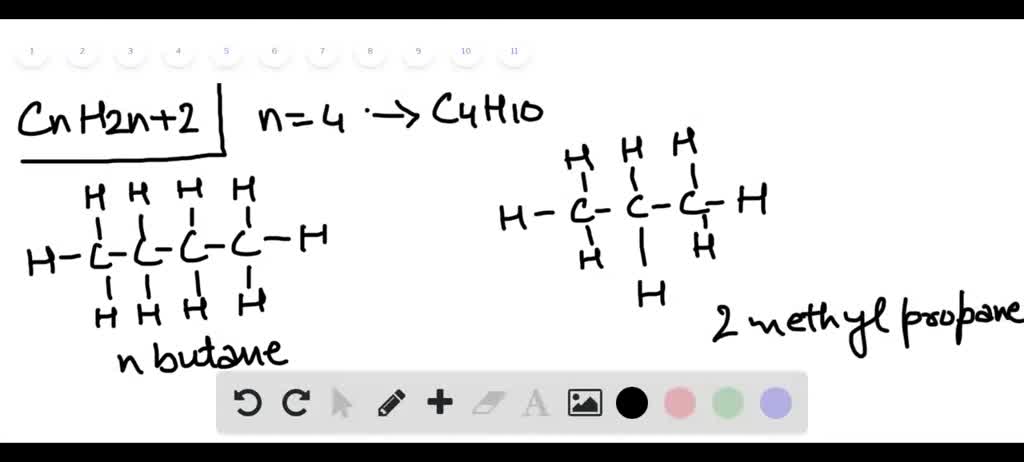
The two most common forms, or isomers, of butane are n-butane and isobutane. Don't let those names intimidate you! They're just different ways those four carbon atoms can link up with the ten hydrogen atoms. It’s like having different arrangements of furniture in the same room; the pieces are the same, but the layout changes things slightly.
n-butane has its carbon atoms all in a straight line, like a perfectly ordered row of soldiers. It's efficient and dependable. Then there's isobutane, where one of the carbon atoms branches off, creating a slightly more compact structure. It’s a little more creatively arranged, but equally useful.
Both versions of butane, C4H10, are incredibly important. They’re often found mixed together in various applications, depending on what properties are needed. It’s like a chef using different spices; the base is the same, but the blend creates a unique flavor. This flexibility is what makes butane so valuable.
When butane burns, it reacts with oxygen, and the result is energy, along with carbon dioxide and water. This energy release is what makes it such a fantastic fuel source. It’s a clean and efficient way to get heat and power. Think of it as a tiny, controlled explosion that gives us warmth and light.
The molecular formula C4H10 is more than just a collection of letters and numbers; it’s a gateway to understanding how things work. It tells us about the precise atomic makeup of this incredibly useful substance. It’s the blueprint for a molecule that powers so much of our modern world.
So, the next time you see a grill roaring to life, or light a candle with a flick of a lighter, take a moment to appreciate the silent, invisible work of butane and its humble yet mighty formula: C4H10. It’s a testament to the fact that even the simplest-looking things can have a huge impact. It’s a tiny molecule with a massive personality!
It’s truly amazing to think that just four carbon atoms and ten hydrogen atoms, arranged in a specific way, can provide so much convenience and utility. Butane is a reminder that chemistry isn't just about test tubes and beakers; it's about the fundamental building blocks of our world and how they contribute to our lives in countless ways. Let's give a cheer for C4H10!
So, there you have it! The dazzling molecular formula of butane, C4H10, revealed! It's a simple yet powerful combination of carbon and hydrogen that fuels our barbecues, lights our way, and makes our lives just a little bit easier, one molecule at a time. Isn't science just the coolest?