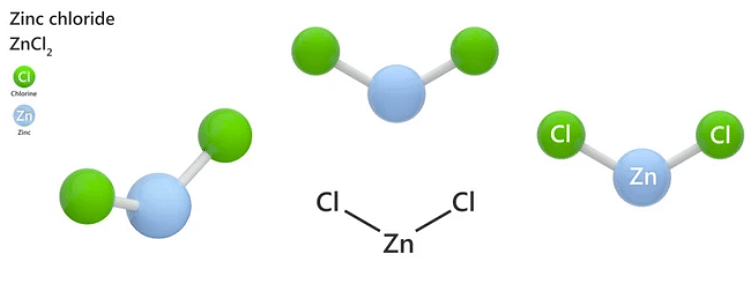
Empirical Formula Zinc Chloride Lab Answers

Hey there, budding chemists and lab wizards! Ever stared at a pile of data from your zinc chloride empirical formula lab and thought, "What in the actual periodic table am I supposed to do with all this?" Don't sweat it! We've all been there, squinting at numbers and wondering if we accidentally stumbled into a secret code. But guess what? Figuring out the empirical formula for zinc chloride is actually way less intimidating than it sounds, and dare I say, even a little bit fun! Think of it as a detective mission, and you're the super-smart sleuth cracking the case of the missing atoms.
So, what exactly is this "empirical formula" thing we're chasing? In simple terms, it's the simplest whole-number ratio of elements in a compound. Like, if you've got a cookie recipe that calls for 10 cups of flour and 5 cups of sugar, the empirical formula for your cookie ingredients would be 2 parts flour to 1 part sugar. See? Not so scary. For zinc chloride, we're trying to find that same basic, unadulterated ratio of zinc (Zn) to chlorine (Cl).
This lab usually involves heating zinc metal in the presence of chlorine gas (or sometimes a source that releases chlorine gas, like a strong acid and a chloride salt – wink wink). As you heat things up, the zinc and chlorine decide to get cozy and form a chemical bond. Your job, my dear scientist, is to meticulously measure the mass of the zinc before the reaction and the mass of the zinc chloride compound after the reaction. This difference in mass? That, my friends, is the mass of the chlorine that joined the party!
Must Read
Let's talk about the nitty-gritty of the numbers. You'll likely have a few key measurements: the initial mass of your zinc, the mass of your crucible (that little dish you heat stuff in), and the mass of your crucible with the final zinc chloride product. To get the mass of the zinc chloride itself, you'll subtract the mass of the empty crucible from the mass of the crucible plus the product. Easy peasy, lemon squeezy.
The mass of the chlorine is a bit of a detective trick. You know how much zinc you started with, right? And you know how much zinc chloride you ended up with. So, the mass of the chlorine that must have been added is simply the mass of the zinc chloride minus the mass of the original zinc. Ta-da! You've just isolated the chlorine's contribution. It’s like figuring out how much pizza dough was left after you added all the pepperoni. Clever, huh?
So, You've Got Your Masses. Now What?
Alright, you've got your trusty numbers: mass of zinc and mass of chlorine. But how do we get from grams to the simple ratio we're after? This is where the periodic table becomes your best friend. Remember those numbers at the bottom of each element's box? Those are the atomic masses, usually expressed in grams per mole (g/mol). Think of a mole as a ridiculously huge baker's dozen – it’s just a specific number of particles (like atoms or molecules).
To find out how many "moles" of zinc you have, you’ll divide the mass of your zinc by its atomic mass. So, if you had, say, 5 grams of zinc, and zinc's atomic mass is roughly 65.38 g/mol, you'd do 5 g / 65.38 g/mol. That gives you the number of moles of zinc. Keep those units handy – they help make sure you're doing the math right. It’s like checking your grocery list to make sure you didn’t accidentally pick up dog food instead of flour. Whoops!
We do the exact same thing for the chlorine! Find the atomic mass of chlorine on the periodic table (it's around 35.45 g/mol). Then, divide the mass of chlorine you calculated earlier by its atomic mass. This gives you the number of moles of chlorine. Now you're getting closer to that sweet, sweet ratio!
The Moment of Truth: Finding the Ratio
You've got moles of zinc and moles of chlorine. Now, the magic happens. To find the simplest whole-number ratio, we usually divide both of your mole values by the smaller of the two mole numbers. This normalizes everything, making one of the elements have a ratio of 1. It's like picking the shortest person in a group to measure everyone else against. See? It’s all about comparison!
For example, let’s say you ended up with 0.076 moles of zinc and 0.153 moles of chlorine. The smaller number is 0.076. So, you’d divide both by 0.076:
- Zinc: 0.076 moles / 0.076 moles = 1
- Chlorine: 0.153 moles / 0.076 moles = approximately 2
And there you have it! A ratio of approximately 1 to 2. This means for every 1 atom of zinc, there are about 2 atoms of chlorine. So, the empirical formula for zinc chloride would be ZnCl₂. Boom! You've cracked the code!
What if My Numbers Aren't So Pretty?
Okay, real talk for a sec. Your numbers probably won't be exactly 1 and 2. Science is messy, and labs have imperfections. You might get something like 1:1.95 or 1:2.08. Don't panic! If the numbers are close to a whole number (like within 0.1 or 0.2), it's generally acceptable to round them. Think of it as rounding to the nearest whole cookie. You wouldn’t split a cookie into 1.95 pieces, right? You'd just call it 2 cookies!
However, if you get something really weird, like 1:1.5 or 1:2.7, that might be a sign to double-check your calculations or think about potential sources of error in your experiment. Did you make sure all your measurements were accurate? Did you heat the zinc thoroughly to ensure it reacted completely? Did you allow it to cool properly before weighing? Little things can make a big difference, like forgetting to add the eggs to your cookie dough. Chef's kiss of disaster.
Sometimes, the heating process might not have been long enough for all the zinc to react. Or maybe some of the product evaporated a bit during heating. These are all common sources of error. If you're getting a ratio that’s a nice fraction, like 1.5, it might indicate a different empirical formula like Zn₂Cl₃. But for zinc chloride, we're generally aiming for that trusty ZnCl₂. Just remember that experimental error is a natural part of science! It’s not a failure; it’s a learning opportunity.
Another thing to consider is the stoichiometry of the reaction. For zinc chloride, the typical ionic compound formed is indeed ZnCl₂. If your lab setup wasn't perfect and you ended up with some unreacted zinc, your chlorine ratio might appear lower than expected. Conversely, if there was some loss of product, the ratio might skew differently. It’s like trying to count the number of friends who showed up to a party when a few decided to sneak in through the back door. A little mystery!
So, don't beat yourself up if your numbers aren't textbook-perfect. The goal is to understand the process and get a result that’s reasonably close. In the real world of science, we often have to deal with approximations and understanding the limitations of our measurements. It's what makes scientists so darn resourceful and, frankly, pretty cool.

Putting It All Together: The Lab Report
Now that you've conquered the calculations, it's time to show off your brilliance! Your lab report will likely ask for:
- The initial mass of zinc. (Your starting point, the star of the show!)
- The mass of the crucible. (The humble stage upon which the reaction occurs.)
- The mass of the crucible with the product. (The grand finale!)
- The calculated mass of the zinc chloride product. (The sweet reward!)
- The calculated mass of chlorine that reacted. (The detective's clue!)
- The moles of zinc. (The microscopic crowd of zinc atoms.)
- The moles of chlorine. (The equally microscopic, but highly reactive, crowd of chlorine atoms.)
- The mole ratio. (The grand unveiling of their relationship!)
- The empirical formula. (The final, simplified answer!)
Don't forget to include any observations you made during the experiment. Did the zinc change color? Did you see any fumes? These details add color and context to your findings. It's like adding a footnote to a great story, giving readers a little extra insight. Plus, your teacher will be impressed by your keen eye for detail. Who doesn't love a bit of impressed teacher energy? High fives all around!
Remember to clearly state your final empirical formula. If your ratio was 1:2, then your empirical formula is ZnCl₂. If, by some wild chance, your calculations led you to something like 1:1.5, you'd need to multiply both numbers by 2 to get a whole number ratio, resulting in Zn₂Cl₃. But again, for zinc chloride, ZnCl₂ is the standard. So, unless your lab was exceptionally unusual, you're probably aiming for that familiar compound.
And if you were asked to calculate the percent composition by mass of zinc and chlorine in your compound, that's just another layer of fun. You’d take the mass of zinc in your final product and divide it by the total mass of the zinc chloride, then multiply by 100. Do the same for chlorine. These percentages should add up to roughly 100% (again, within experimental error, because nothing is ever perfectly 100% in the messy world of chemistry!).
A Word on Precision and Accuracy
It’s worth mentioning the difference between precision and accuracy, as they are crucial in any lab experiment, especially when determining empirical formulas. Precision refers to how close your repeated measurements are to each other. If you weigh the same zinc sample five times and get 1.01g, 1.02g, 1.01g, 1.03g, 1.02g – that’s pretty precise! Accuracy, on the other hand, refers to how close your measurements are to the true, correct value. Getting a result that’s close to the theoretical empirical formula of ZnCl₂ shows good accuracy. Our goal in this lab is to achieve both!
Think of it like shooting arrows. If all your arrows hit the same spot on the wall, but that spot is way off the bullseye, you’re precise but not accurate. If your arrows are scattered all over the place, you're neither precise nor accurate. But if your arrows are clustered tightly around the bullseye? Bingo! You’re both precise and accurate. In the zinc chloride lab, we strive for that tight cluster around the true empirical formula. It’s a beautiful thing when it all comes together.
Sometimes, when looking at your final results, you might get a percentage of error. This is calculated by comparing your experimental result to the accepted theoretical value. The formula for percent error is: |Experimental Value - Theoretical Value| / Theoretical Value * 100%. Don’t be alarmed if you have a small percentage of error; it’s a testament to the fact that you’re engaging with the real-world challenges of scientific measurement. It’s like seeing a little smudge on a masterpiece painting – it doesn't detract from the overall brilliance.
And hey, if you’re really struggling with the numbers, don't hesitate to ask your teacher or a classmate for a little clarification. Collaboration is key in science! Sometimes, a fresh pair of eyes can spot a miscalculation or a misunderstanding that you might have missed. It’s like having a second opinion when you're trying to assemble IKEA furniture – essential!
So, there you have it! The mystery of the zinc chloride empirical formula, solved. You’ve transformed raw data into a fundamental chemical formula. You’ve navigated the treacherous waters of atomic masses and mole calculations. You’ve emerged victorious, armed with knowledge and a newfound appreciation for the elegant simplicity of chemical compounds.
Remember, every lab experiment, even the ones that feel a little confusing at first, is a step forward in your scientific journey. You’re not just following instructions; you’re actively participating in the process of discovery. So, give yourself a pat on the back, you brilliant scientist! You’ve tackled a classic chemistry challenge and come out on top. Keep that curiosity burning bright, and the world of science will always be an exciting adventure waiting for you. Now go forth and conquer the next lab, armed with your newfound empirical formula expertise and a smile! You’ve totally got this!