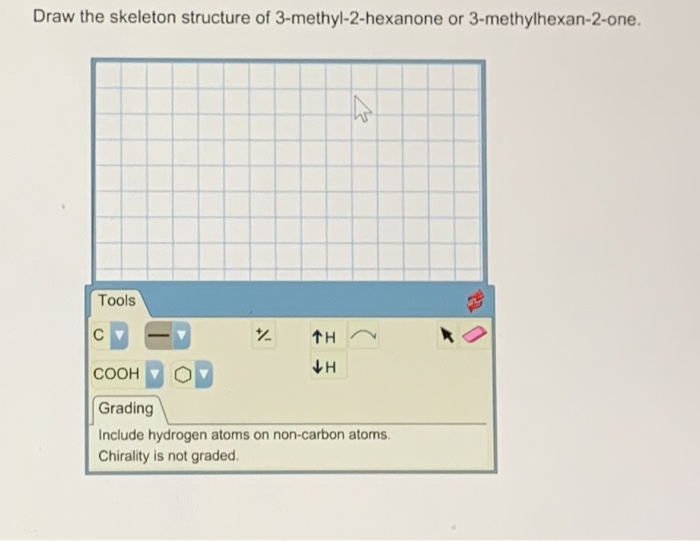
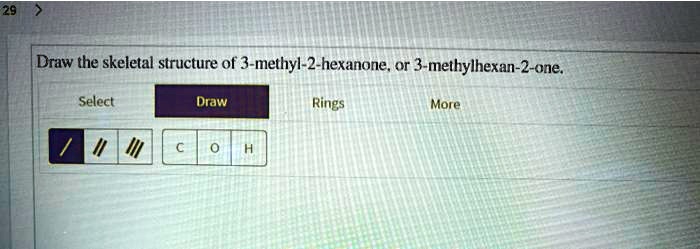
Draw The Structure Of 3 Methyl 2 Hexanone
Ever found yourself staring at a recipe, a skincare ingredient list, or even a cool chemistry experiment explained online, and suddenly, BAM! There it is: a name that sounds like it belongs in a sci-fi flick or a forgotten alchemist's notebook. Today, we're diving into one such intriguing molecule: 3-methyl-2-hexanone. Don't let the scientific-sounding name throw you off; think of it less as a tongue-twister and more as a tiny architectural blueprint for something that might be hiding in plain sight, adding a little je ne sais quoi to our everyday world.
So, what's the deal with this 3-methyl-2-hexanone? At its core, it's a ketone. Now, ketones are a whole family of organic compounds, and they’re pretty common. Think of them as the cool cousins of alcohols. They have a distinctive carbonyl group (that's a carbon atom double-bonded to an oxygen atom, C=O) that gives them their unique character. You'll find ketones in everything from nail polish remover (acetone, anyone?) to natural flavors and fragrances. They're the workhorses that add those zesty, sweet, or even slightly pungent notes to our sensory experiences.
Let's break down the name itself, because it's actually a clever little clue, like a treasure map for chemists. “Hexanone” tells us the main skeleton of our molecule. “Hexa-” means six, so we're looking at a chain of six carbon atoms. “-one” is the giveaway for the ketone functional group, meaning that carbonyl group is part of that six-carbon chain. But where is it? The “2-” before hexanone tells us the carbonyl group is hanging out on the second carbon atom of that chain. Imagine our six carbons lined up like dominoes; the ketone funkiness is on domino number two.
Must Read
Painting the Picture: The Structure Unveiled
Now for the fun part: drawing it! Don't worry, this isn't about becoming a molecular origami master overnight. It's more about visualizing the building blocks. We’ll start with that six-carbon chain. You can draw it as a straight line, or if you want to be fancy, a zig-zag line. The zig-zag is actually more accurate because carbon chains tend to bend in a tetrahedral shape, but for simplicity, a straight line works perfectly well to get the idea across. So, six Cs in a row.
Let’s number them from one end to the other: 1, 2, 3, 4, 5, 6. Easy peasy. Now, remember that “2-” in hexanone? That means our carbonyl group (C=O) attaches to the second carbon. So, where carbon number 2 is, we'll draw a double bond to an oxygen atom. This is what makes it a ketone!
But wait, there's more! The “3-methyl” part of the name is our final piece of the puzzle. “Methyl” refers to a methyl group, which is simply a carbon atom attached to three hydrogen atoms (CH3). The “3-” tells us where this little methyl group is attached. It's glued onto the third carbon atom in our main six-carbon chain.
So, to recap: you have your six-carbon chain. On the second carbon, you have your double-bonded oxygen. And on the third carbon, you have your little CH3 buddy hanging out. The remaining bonds on each carbon atom are filled with hydrogen atoms. Carbon likes to make four bonds, so you’ll see carbons with one or two hydrogens attached to make sure they’re all happy and bonded up.

Visualizing the Vibe: From Line Drawings to the Real Deal
When chemists draw these molecules, they often use what's called a skeletal structure. This is where the magic of simplification happens. You don't draw every single carbon and hydrogen. Instead, each point where lines meet, and each open end of a line, represents a carbon atom. Hydrogens are assumed to be there, filling up the valency of the carbon. So, 3-methyl-2-hexanone in skeletal form would look like a zig-zag line of six points (representing carbons), with a double bond to an oxygen off the second point, and a short little single line (representing the methyl group) sticking out from the third point.
Think of it like minimalist art for molecules. It’s clean, efficient, and gets the point across without the clutter. It’s the difference between a detailed architectural rendering and a stylish floor plan – both convey the information, but one has a certain aesthetic. And in the world of chemistry, this aesthetic is crucial for quick understanding and clear communication.
Where Do These Molecules Hang Out?
So, why should we care about drawing 3-methyl-2-hexanone? Well, molecules like this are the unsung heroes of many things we encounter daily. Ketones, in general, are prevalent in nature. They contribute to the aromas of fruits, flowers, and even certain cheeses. While 3-methyl-2-hexanone itself might not be a household name like vanilla or cinnamon, it’s a building block in the vast symphony of organic chemistry.
It’s possible that this specific ketone, or molecules very similar to it, could be found in natural flavors and fragrances, contributing to complex scents. Imagine a rare orchid with a unique perfume, or a tropical fruit with a surprisingly spicy undertone. Such nuances are often due to the intricate interplay of different organic molecules, and 3-methyl-2-hexanone could be a tiny, yet significant, player in that fragrant orchestra.
Beyond the olfactory, ketones are important in the industrial world too. They can be used as solvents, meaning they’re great at dissolving other substances. This makes them useful in cleaning products, paints, and even in the manufacturing processes of plastics and textiles. So, while you might not be directly interacting with pure 3-methyl-2-hexanone, the products you use, or the way those products are made, might involve its molecular cousins or even itself.
A Dash of Fun: The Art of Naming Molecules
The way chemists name compounds is a fascinating art form in itself, governed by the International Union of Pure and Applied Chemistry (IUPAC). It’s like a universal language, ensuring that a scientist in Tokyo and a scientist in Timbuktu can refer to the exact same molecule without any ambiguity. The system is based on the longest carbon chain, the type of functional groups present, and their positions on the chain. It’s a logical, hierarchical system that, once you get the hang of it, is incredibly powerful.
Think of it like giving a person a full name: a first name, a last name, and perhaps a middle name. "Hexanone" is the family name, the core identity. "2-" and "3-methyl" are the distinguishing details, the specific characteristics that make this molecule unique within its family. It's a system that prioritizes clarity and precision, a hallmark of good scientific practice.
And sometimes, these names even spark curiosity. The very act of decoding "3-methyl-2-hexanone" encourages us to look deeper, to understand the underlying structure and its potential roles. It's like finding a cryptic crossword clue that, once solved, reveals a hidden image or a witty phrase.

Practical Tips for the Curious Mind
If you're feeling inspired to doodle some molecules yourself, here are a few pointers:
- Start with the skeleton: Identify the longest carbon chain. This will give you the base name (e.g., hexane, pentane).
- Locate the funk: Find the main functional group (like the ketone -one, or alcohol -ol) and determine its position on the chain using the lowest possible number.
- Add the extras: Note any substituent groups (like methyl, ethyl) and their positions on the main chain.
- Use skeletal structures: Once you're comfortable, try drawing in skeletal form. It’s quicker and cleaner! Remember, each vertex and end point is a carbon, and hydrogens are implied.
- Practice makes perfect: Grab a textbook, a chemistry website, or even just a pen and paper. Draw simple molecules first, then build up to more complex ones. You’ll be surprised at how quickly it becomes second nature.
Don't be afraid to make mistakes! Even experienced chemists sometimes have to redraw their structures. The goal is understanding, not immediate perfection. Think of it as a creative exercise, a way to visualize the unseen world of molecules.
A Little Bit of Culture: Molecules in Art and Media
While 3-methyl-2-hexanone might not be a pop culture icon, the visual representation of molecules has found its way into art, design, and even popular media. Think of stylized molecular graphics in sci-fi films, or abstract art inspired by chemical structures. The elegance and complexity of molecular arrangements can be incredibly beautiful and thought-provoking.
Even something as seemingly mundane as a chemical formula can inspire a logo, a pattern, or a piece of jewelry. The inherent order and symmetry in many molecules lend themselves well to visual interpretation. It's a testament to the fact that even the most technical fields can spark artistic inspiration.
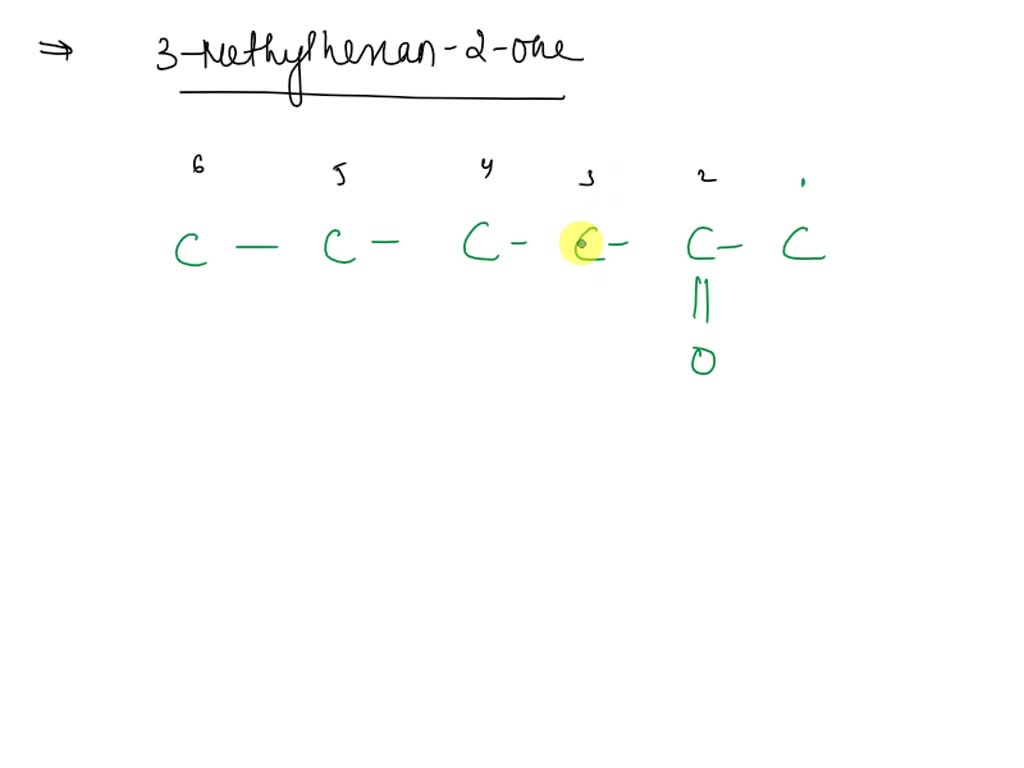
Consider the famous double helix of DNA – it's instantly recognizable and has become a symbol of life, science, and innovation. While our humble 3-methyl-2-hexanone is far simpler, the principle of a unique structure dictating function and identity is universal across the molecular world.
The Bigger Picture: Connecting to Daily Life
So, how does drawing a molecule like 3-methyl-2-hexanone connect to our daily lives? It’s about appreciating the intricate world around us. Every scent we inhale, every flavor we taste, every material we touch – it's all made up of molecules. Understanding the basics of their structure allows us to peek behind the curtain, to appreciate the complexity and ingenuity of the natural and chemical world.
It’s like learning to read the ingredients list on a food product. Initially, it might seem like a foreign language. But as you learn what each ingredient is, you gain a better understanding of what you’re consuming. Similarly, with molecules, understanding their structures gives us a deeper appreciation for the substances that form our world.
Furthermore, the process of drawing and understanding molecular structures cultivates a sense of curiosity and problem-solving. It encourages us to think logically, to break down complex problems into smaller parts, and to visualize abstract concepts. These are skills that are invaluable, whether you're navigating a tricky DIY project, planning your next vacation, or even just trying to assemble a piece of IKEA furniture.
Ultimately, the exercise of drawing 3-methyl-2-hexanone is a small step into a vast and fascinating universe. It’s a reminder that even the most complex phenomena can be understood by breaking them down into their fundamental components. And in doing so, we gain a richer, more informed perspective on the world we inhabit, one molecule at a time.