Draw The Lewis Structure For The Pcl 4 Ion

Hey there, future Lewis structure superstar! Ever find yourself staring at chemical formulas and thinking, "What in the world is going on here?" You're not alone! Today, we're diving into a super fun puzzle: drawing the Lewis structure for the PCl4+ ion. Sounds fancy, right? But trust me, it’s more like building with LEGOs, but with atoms and electrons!
So, what's the big deal about Lewis structures? Think of them as a tiny blueprint. They show us how atoms link up in a molecule or ion. It’s like seeing the secret handshake of atoms! And the PCl4+ ion? It’s got a bit of a story to tell. It’s not your everyday molecule. It's an ion, which means it has a charge. And that charge adds a little extra spice to our drawing fun.
Let's Meet Our Players: Phosphorus and Chlorine!
First up, we have phosphorus (P). It's in Group 15 of the periodic table. That means it’s got five valence electrons. Think of valence electrons as the "party animals" of an atom, the ones that actually get to mingle and bond. Phosphorus is like the host of the party, ready to share!
Must Read
Then we have chlorine (Cl). Chlorine is in Group 17, a.k.a. the halogens. Halogens are famous for being a bit greedy with electrons. Each chlorine atom brings seven valence electrons to the table. They’re like the guests who show up with a plus-one and a really nice gift!
Counting Up Our Electrons: The Grand Total!
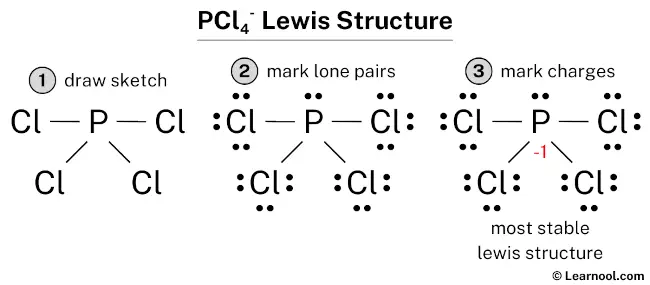
Now, for the super important step: counting all the electrons we have to work with. We have one phosphorus atom and four chlorine atoms. So, that's 5 electrons (from P) + (4 * 7 electrons from Cl). That gives us 5 + 28 = 33 electrons. Easy peasy, right?
But wait! Remember that little “+” sign next to PCl4? That’s a positive charge. A positive charge means the ion has lost an electron. So, we need to subtract one electron from our total. 33 - 1 = 32 valence electrons. This is our magic number! This is the total number of electrons we need to arrange perfectly in our Lewis structure.
Finding the Central Atom: Who's the Boss?
In any Lewis structure, there's usually a central atom. This is the atom that all the others will connect to. How do we find it? Usually, it’s the atom that’s the least electronegative (least "electron-hogging"). In our case, phosphorus is less electronegative than chlorine. So, phosphorus (P) is our star player, our central atom!
Imagine phosphorus sitting in the middle, like a proud conductor, ready to orchestrate the dance of the chlorine atoms. The four chlorine atoms will surround it, like eager dancers waiting for their cue.

Connecting the Dots: The First Bonds!
Okay, let's start building! We’ll place the phosphorus atom in the center. Then, we’ll draw single bonds connecting each of the four chlorine atoms to the central phosphorus. A single bond is made of two electrons. So, we've used 4 bonds * 2 electrons/bond = 8 electrons so far. We're making progress!
It’s like drawing lines on a piece of paper, connecting the dots. P is in the middle, and Cls are all around it, holding hands (or, you know, sharing electrons) with P.
Completing the Octets: Giving Everyone a Full Hand
Now, we need to make sure every atom is happy. In chemistry, "happy" often means having a full outer shell of electrons, usually eight (an octet). We’ve used 8 electrons for the bonds. We started with 32, so we have 32 - 8 = 24 electrons left. Plenty to go around!
Let’s give each of the surrounding chlorine atoms its fair share. Each chlorine atom already has 2 electrons in its bond with phosphorus. To complete its octet, it needs 6 more electrons. We can add these as lone pairs of electrons (dots) around each chlorine. So, 3 lone pairs * 2 electrons/pair = 6 electrons per chlorine. We have 4 chlorine atoms, so that’s 4 * 6 = 24 electrons. Voila! We've used up all our remaining electrons.
So, each chlorine atom now has 2 electrons from the bond + 6 electrons from lone pairs = 8 electrons. They’re all looking pretty content with their octets.
Checking the Central Atom: Is Phosphorus Happy Too?
But what about our central phosphorus atom? It has 4 single bonds connecting it to the chlorine atoms. Each bond has 2 electrons. So, phosphorus currently has 4 bonds * 2 electrons/bond = 8 electrons. That’s a full octet for phosphorus too! It’s like everyone at the party finally got their favorite snack!

The Crucial Step: The Formal Charge Dance
Now, here’s where things get a little more nuanced, and honestly, a lot more fun. We need to consider the formal charge. This is a way to see where the electrons "belong" in an ideal scenario. It helps us figure out the best Lewis structure, especially for ions.
The formula for formal charge is: (number of valence electrons in the free atom) - (number of non-bonding electrons) - (1/2 * number of bonding electrons).
Let's calculate it for each atom in our current structure:
- For each Chlorine (Cl) atom:
- For the Phosphorus (P) atom:
Valence electrons in free Cl = 7
Non-bonding electrons (lone pairs) = 6
Bonding electrons = 2 (from the single bond)
Formal charge = 7 - 6 - (1/2 * 2) = 7 - 6 - 1 = 0. Nice! All the chlorine atoms have a formal charge of zero.
Valence electrons in free P = 5
Non-bonding electrons = 0 (no lone pairs on P in this structure)
Bonding electrons = 8 (4 single bonds * 2 electrons/bond)
Formal charge = 5 - 0 - (1/2 * 8) = 5 - 0 - 4 = +1.
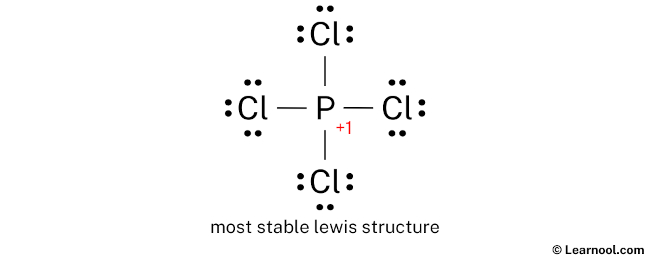
Look at that! The formal charge on phosphorus is +1. And guess what? The overall charge of the ion is also +1! This is a really good sign. It means our structure is likely the correct and most stable one. We've successfully placed our positive charge right where it belongs – on the phosphorus atom!

The Final Picture: PCl4+ Unveiled!
So, the Lewis structure for the PCl4+ ion looks like this: a central phosphorus atom single-bonded to four chlorine atoms. Each chlorine atom has three lone pairs of electrons. The phosphorus atom has no lone pairs. And the whole thing is enclosed in brackets with a superscript “+” sign. Ta-da!
This structure shows us that phosphorus, even though it’s in Group 15, can expand its octet when bonded to highly electronegative atoms like chlorine. And the positive charge? It’s a little sticker that tells us about the ion’s history – it lost an electron somewhere along the line.
Why is this Fun? The Quirky Side!
Why is this even fun, you ask? Well, for starters, it’s like being a tiny detective! You’re piecing together clues (electron counts, electronegativity) to solve a chemical mystery. Plus, visualizing these invisible structures is like having a superpower. You’re seeing the world of atoms in a whole new way.
And let's not forget the terminology! "Valence electrons," "octet rule," "formal charge" – they sound like words from a secret agent movie. But you just used them like a pro! It’s a bit like learning a new language, but this language explains how everything around you is put together.
Plus, drawing these structures is surprisingly therapeutic. It’s a calm, focused activity. You get to doodle, but with scientific meaning. Who knew that chemistry could be so… artistic? And the fact that we can accurately predict how atoms will behave and connect? That’s pretty darn cool, if you ask me!
So, next time you see PCl4+, you can smirk and think, "I know exactly what you look like!" You’ve just unlocked another level in the amazing game of chemistry. Keep exploring, keep drawing, and most importantly, keep having fun with it!