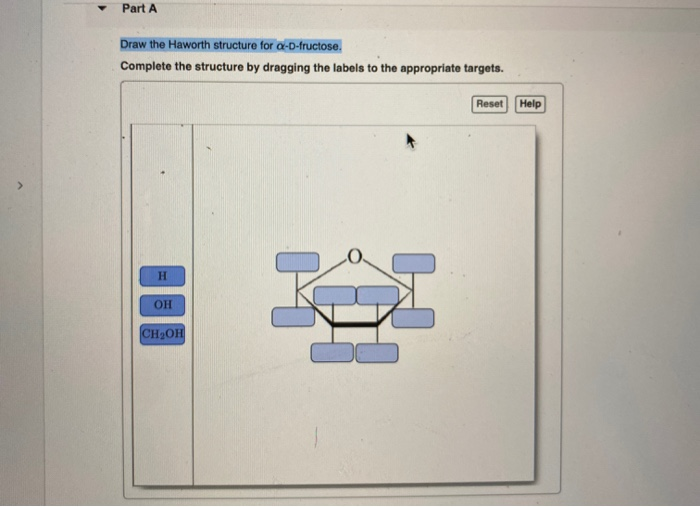
Draw The Haworth Structure For α-d-fructose
Hey there, fellow explorers of the sweet side of life! Ever find yourself staring at a sugar cube and wondering, "What's really going on in there?" Well, buckle up, because today we're taking a little detour into the fascinating world of molecular structures, specifically, the darling of the fruit bowl: α-d-fructose. Forget stuffy textbooks and intimidating diagrams; we're going to draw this beauty like we're sketching a cool new logo or planning a chill weekend getaway. Think of it as molecular doodling for the soul.
Now, you might be thinking, "Haworth structure? Sounds intense!" But honestly, it's just a fancy way of drawing a molecule in a ring shape, making it easier to visualize how those little atoms are hanging out. And fructose, oh fructose! It's that irresistible sweetness that makes fruits sing and desserts divine. It’s the sugar that powers your morning smoothie, the subtle magic in your afternoon tea, and the star of many a baked treat. We encounter it every single day, and understanding its structure is like getting a backstage pass to its deliciousness.
So, why α-d-fructose? Let's break it down. The "d" part tells us about its handedness, a concept in chemistry that’s a bit like looking in a mirror – some molecules are mirror images of each other. "Fructose" is the name of the sugar itself, that wonderfully sweet monosaccharide. And the "α" (alpha) is a special designation that refers to the orientation of a particular group when the molecule decides to get cozy and form a ring. Don't worry if that sounds a tad technical; we'll get a feel for it as we go.
Must Read
Unpacking the Building Blocks: What We Need
Before we grab our virtual pencils, let's get acquainted with the players. Fructose is a six-carbon sugar. Yep, six little carbon atoms, the backbone of so much organic life. In its open-chain form, it has a ketone group (a carbon double-bonded to oxygen and single-bonded to two other carbons) on the second carbon, which is a bit different from its cousin, glucose, which has an aldehyde group. This ketone group is key to fructose’s unique behavior, especially when it decides to ring up.
We’ll also need to remember the hydroxyl groups (-OH) attached to the other carbons. These are the water-loving buddies that make sugars soluble and are super important for their chemical reactions. And of course, the oxygen atom that will bridge the ring structure. Think of it as the glue that holds our molecular party together.
So, we have our carbon backbone, our hydroxyl groups, our oxygen, and the special ketone group that sets fructose apart. Ready to bring them to life?
The Art of the Ring: Drawing the Haworth Structure
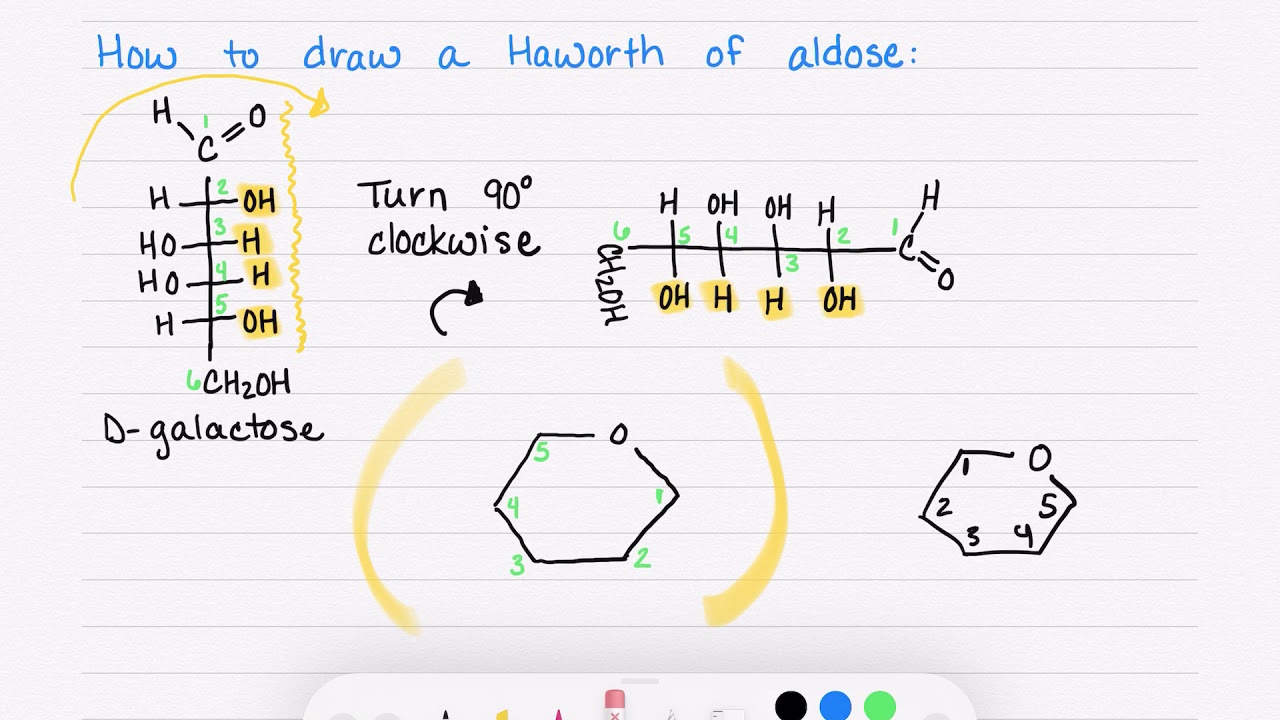
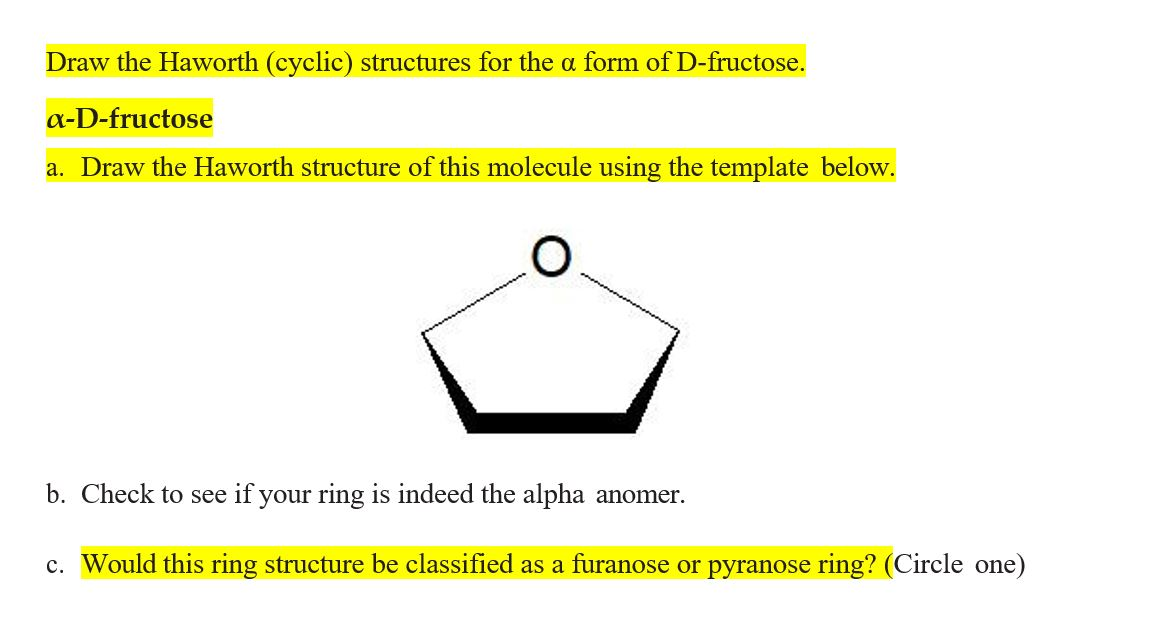
The Haworth structure is all about perspective. Imagine you're looking at the ring of fructose from a specific angle. We'll draw it as a slightly tilted, almost hexagonal shape. But wait, fructose is a five-membered ring when it cyclizes! This is one of its signature moves, a little twist that makes it a furanose, unlike glucose which forms a six-membered pyranose ring. So, our Haworth structure for fructose will actually be a five-membered ring with one oxygen atom as part of the ring, and the sixth carbon atom hanging out as a substituent.
First, let’s draw our basic five-membered ring. It looks a bit like a pentagon, but one vertex is an oxygen atom. We’ll place this oxygen atom on the right side of our drawing, pointing upwards. This is going to be our anchor.
Now, let’s place our carbon atoms. The carbon that was C1 in the open-chain form will be the carbon adjacent to the ring oxygen, on the top. We’ll call this C2. Then, moving clockwise, we have C3, C4, and C5. C5 is the carbon that will connect back to the ring oxygen, closing the loop with C2. C6, the last carbon, will be attached to C5 as a substituent group, usually as a -CH2OH.
Placing the Substituents: Up, Down, and All Around
This is where the "α" and "d" come into play, determining the orientation of our hydroxyl groups. For the Haworth projection, we imagine the ring lying flat (though it's not truly flat in 3D, this is a simplification!). Groups pointing up are above the plane of the ring, and groups pointing down are below.
Let's start with the "d" configuration. This generally means that the hydroxyl group on the highest numbered chiral center (the carbon that has four different groups attached) will be on the right in the Fischer projection, which translates to being down in the Haworth projection for d-sugars. For fructose, our highest numbered chiral center (if we consider the open-chain form) is C5. In the ring structure, we need to be mindful of the orientation of the groups on C2, C3, and C4.
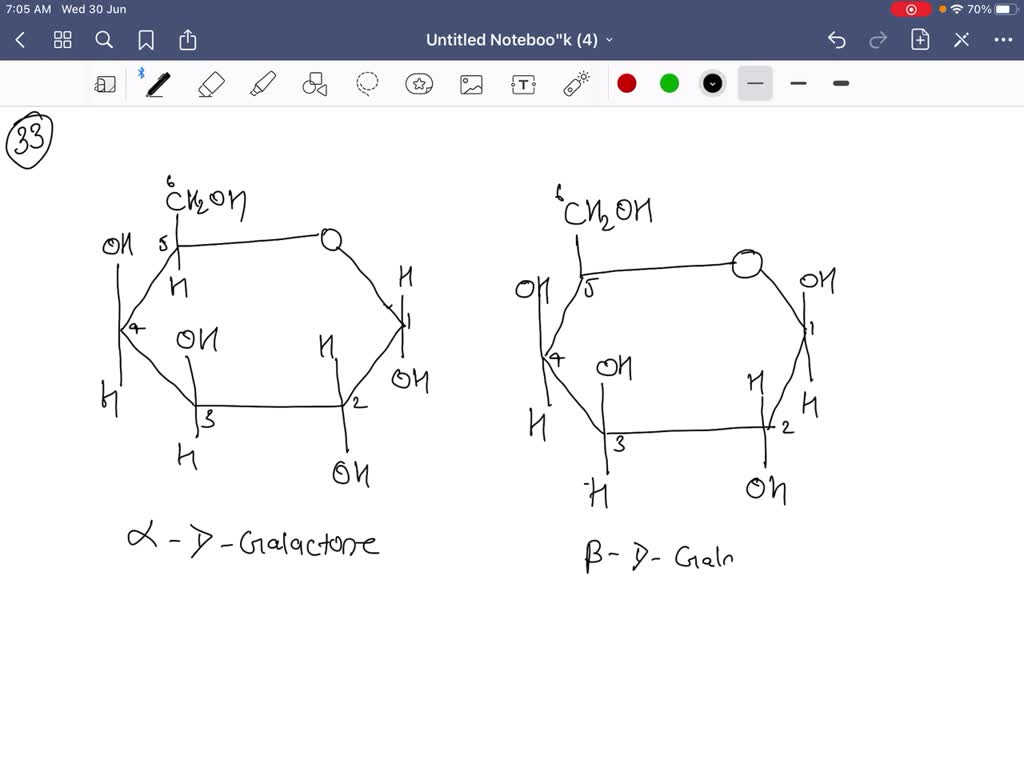
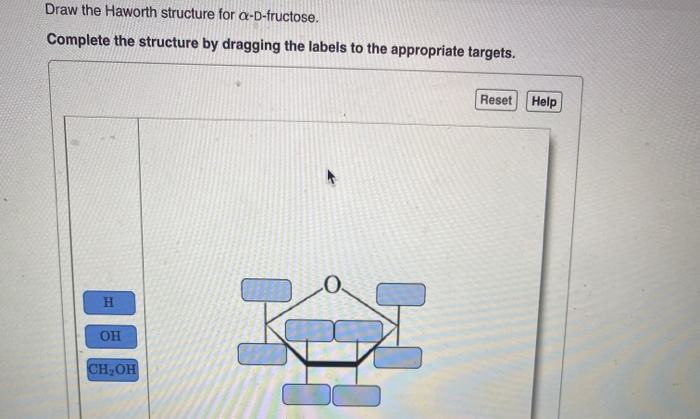
Now, for the crucial "α" designation in α-d-fructose. This tells us the orientation of the hydroxyl group on the anomeric carbon. The anomeric carbon is the carbon that was originally the carbonyl carbon (C1 in the open-chain form) and is now attached to two oxygen atoms – one in the ring and one in the hydroxyl group that formed the ring. In fructose, the anomeric carbon is C2. For the α-anomer, the hydroxyl group on C2 points down. This is a key distinguishing feature of α-fructose.
So, let's place our hydroxyl groups:
- C2 (anomeric carbon): The -OH group points down.
- C3: The -OH group points up.
- C4: The -OH group points down.
- C5: This carbon is part of the ring and is bonded to C4 and the ring oxygen. The -CH2OH group (which is C6) is attached to C5. In the Haworth structure, this -CH2OH group typically points up.
It's like a little dance of up and down. You can remember it this way: for d-fructose in its ring form, C2-OH is down (alpha), C3-OH is up, and C4-OH is down. The C6 group (-CH2OH) is usually pointing up.
A Fun Little Fact: The Furan vs. Pyran Debate
Did you know that fructose can also form a six-membered ring, a pyranose structure, just like glucose? However, it much prefers to form the five-membered furanose ring, especially in solution. This is why the Haworth structure we're drawing is a furanose. It's like fructose saying, "You know what? I like a cozy little five-member club better!" This preference for the furanose form is a big reason why fructose has slightly different chemical properties compared to glucose.
Putting It All Together: Your α-d-Fructose Masterpiece
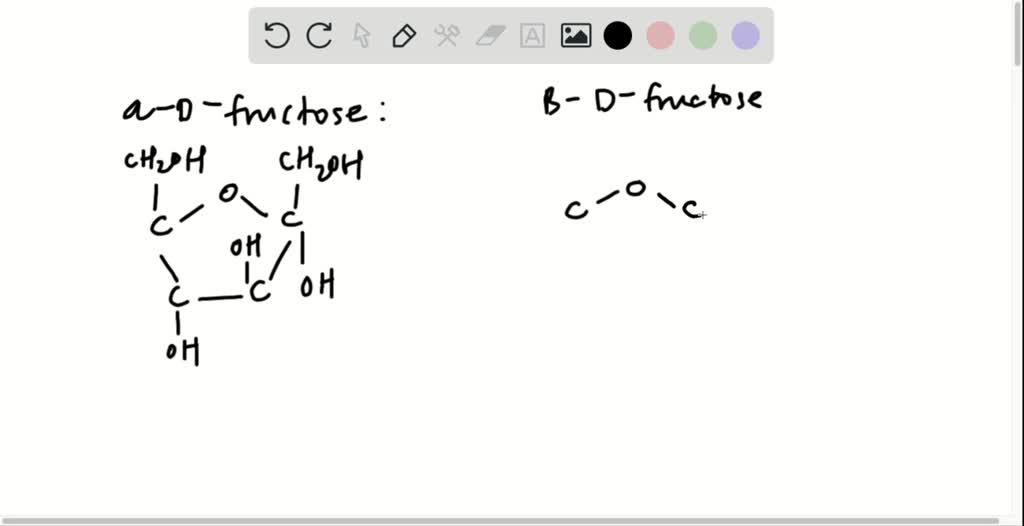
Let’s visualize the final drawing:
Imagine a slightly tilted pentagon. The top-right vertex is an oxygen atom. Moving clockwise from there:
- The next vertex is C2. Attached to C2, pointing downwards, is an -OH group.
- The next vertex is C3. Attached to C3, pointing upwards, is an -OH group.
- The next vertex is C4. Attached to C4, pointing downwards, is an -OH group.
- The last vertex in the ring is C5. Attached to C5, pointing upwards, is a -CH2OH group. The C5 carbon is also bonded to the ring oxygen, completing the furanose ring.
And there you have it! Your very own Haworth structure of α-d-fructose. It's a visual representation of how these atoms arrange themselves to create that wonderful sweetness we love. You can even draw it on a sticky note and stick it on your fridge – a little reminder of the molecular marvels that fuel our bodies.
Tips for Your Molecular Doodling
- Start with the ring: Always sketch out the ring shape first. For fructose, it’s a five-membered ring with an oxygen.
- Place the anomeric carbon: Identify your anomeric carbon (C2 for fructose) and remember the alpha/beta distinction dictates its -OH group's orientation.
- Memorize the patterns: While understanding is key, there are patterns for common sugars. For d-fructose furanose, think Down-Up-Down for the OH groups on C2, C3, C4, with C6-CH2OH usually Up.
- Practice makes perfect: The more you draw them, the more intuitive it becomes. Grab some paper and sketch it out while you’re enjoying a piece of fruit!
- Color-coding can help: If you're feeling artistic, use different colors for carbon, oxygen, and hydrogen to make it even clearer.
Think of this as your secret code to understanding the sweetness. It's not just random atoms; it's a carefully arranged structure that interacts with our taste buds in a specific way.
A Sweet Connection to Our Daily Lives
Why does this matter, beyond a fun doodle? Understanding these structures helps us appreciate the science behind what we consume. It’s how scientists develop new sweeteners, understand how our bodies metabolize sugars, and even how certain fruits maintain their unique flavors. When you bite into a juicy apple, you're experiencing the culmination of intricate molecular design.
Fructose, in its natural form within fruits, comes bundled with fiber, vitamins, and minerals. This complex package means our bodies process it differently than the highly processed high-fructose corn syrup you might find in some sodas. It's a subtle, yet significant, difference that knowing about molecular structure can help illuminate.
So, the next time you're enjoying a sweet treat, take a moment. Close your eyes and picture that little five-membered ring, the dance of hydroxyl groups, and the sheer elegance of α-d-fructose. It’s a reminder that even in the simplest pleasures, there’s a world of fascinating science at play, waiting to be explored, one delicious molecule at a time.
Keep on exploring, keep on tasting, and keep on drawing! Your journey into the sweet science of life is just beginning.