Draw All Stereoisomers Of 3 Chloro 1 Butene
Have you ever looked at a molecule and wondered, "Is that the only way it can be arranged?" In the fascinating world of chemistry, the answer is often a resounding "Nope!" Today, we're diving into a fun puzzle: drawing all the different ways a molecule called 3-chloro-1-butene can exist. It might sound a bit niche, but understanding these "molecular siblings" is super important in so many fields, from developing life-saving medicines to creating new materials. Think of it like this: if you have a Lego set, there are a million ways to put those bricks together, right? Molecules are similar, but with very specific rules for how their "bricks" (atoms) can connect and orient themselves in space. This ability to see and draw these different arrangements, called stereoisomers, is a core skill for any budding chemist and even helps us understand the world around us better.
So, what's the big deal with stereoisomers? Imagine you have a pair of gloves. They're both gloves, made of the same material, same number of stitches, but you can't wear the left glove on your right hand and vice-versa, right? They are mirror images but not identical. This is the essence of chirality, a key concept when we talk about stereoisomers. Molecules can also be "left-handed" or "right-handed," and this difference can have a huge impact on how they interact with other molecules. This is particularly critical in biology. For example, one stereoisomer of a drug might be incredibly effective at treating a disease, while its mirror image might be completely inactive or even harmful! This is why pharmaceutical companies spend a lot of time and effort ensuring they produce the correct stereoisomer for their medications. Beyond medicine, understanding stereoisomers helps us design catalysts for chemical reactions, develop new flavors and fragrances (many of which are chiral and have distinct smells!), and even understand how our senses perceive the world.
Unraveling 3-Chloro-1-Butene: Our Molecular Star
Let's bring our attention to our star for today: 3-chloro-1-butene. This molecule has the chemical formula C4H7Cl. The name itself gives us clues: a four-carbon chain (butene), a double bond starting at the first carbon (1-butene), and a chlorine atom attached to the third carbon (3-chloro).
Must Read
The IUPAC name (International Union of Pure and Applied Chemistry) is a chemist's roadmap!
When we draw molecules, we often use different representations to show their structure. The most common ones are structural formulas, which show how atoms are connected, and spatial representations, which try to depict the 3D arrangement of atoms. For stereoisomers, the 3D representation is where things get interesting.
The first step in drawing any molecule is to get its basic connectivity right. For 3-chloro-1-butene, we have a four-carbon chain. Carbon 1 and 2 are connected by a double bond. Carbon 3 is attached to Carbon 2 and Carbon 4, and it also has a chlorine atom attached to it. Carbon 4 is attached to Carbon 3. We then fill in the remaining bonds with hydrogen atoms to satisfy the valency of each carbon (which is typically four bonds). So, we have CH2=CH-CHCl-CH3.

Spotting the Possibilities: Where the Fun Begins!
Now, for the stereoisomers! The key to identifying potential stereoisomers is to look for chiral centers. A chiral center is typically a carbon atom that is bonded to four different groups. Let's examine our 3-chloro-1-butene molecule. Looking at the structure CH2=CH-CHCl-CH3, let's focus on the carbon atoms:
- Carbon 1: It's part of a double bond and is bonded to two hydrogens. Not chiral.
- Carbon 2: It's part of a double bond and is bonded to one hydrogen and Carbon 1. Not chiral.
- Carbon 3: This carbon is bonded to Carbon 2, Carbon 4, a hydrogen atom, and a chlorine atom. Let's break down the groups attached:
- -CH=CH2 (from Carbon 2 and 1)
- -CH3 (from Carbon 4)
- -H (a hydrogen atom)
- -Cl (a chlorine atom)
- Carbon 4: This is a methyl group (-CH3) and is bonded to three hydrogens and Carbon 3. Not chiral.
Because we have one chiral center at Carbon 3, we know that 3-chloro-1-butene will exist as a pair of enantiomers. Enantiomers are stereoisomers that are non-superimposable mirror images of each other. Think back to our gloves – they are enantiomers.
Drawing the Enantiomers: A Tale of Two Hands
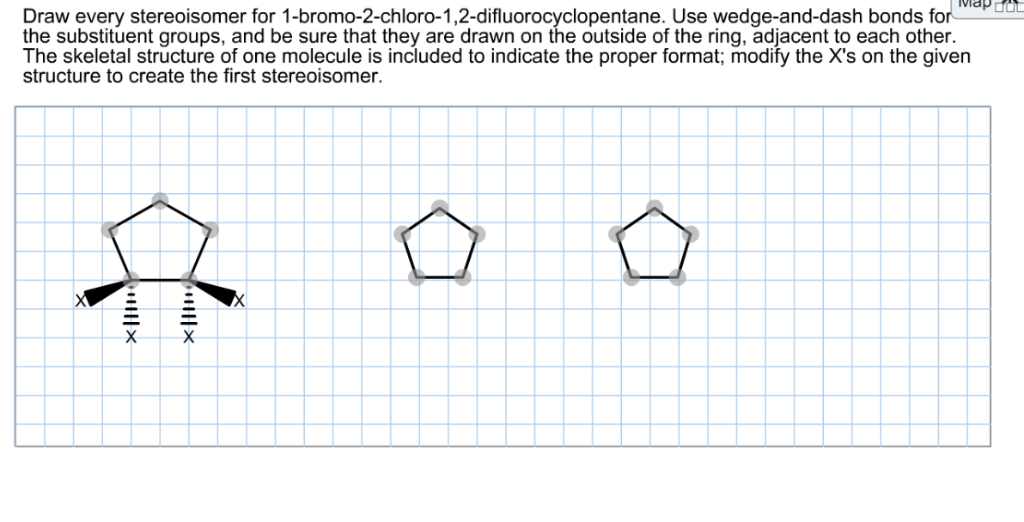
To draw these, we'll often use wedge-and-dash notation, which helps us visualize the 3D arrangement of atoms around the chiral center. A wedge represents an atom coming out of the plane of the paper towards you, and a dash represents an atom going back into the plane away from you.
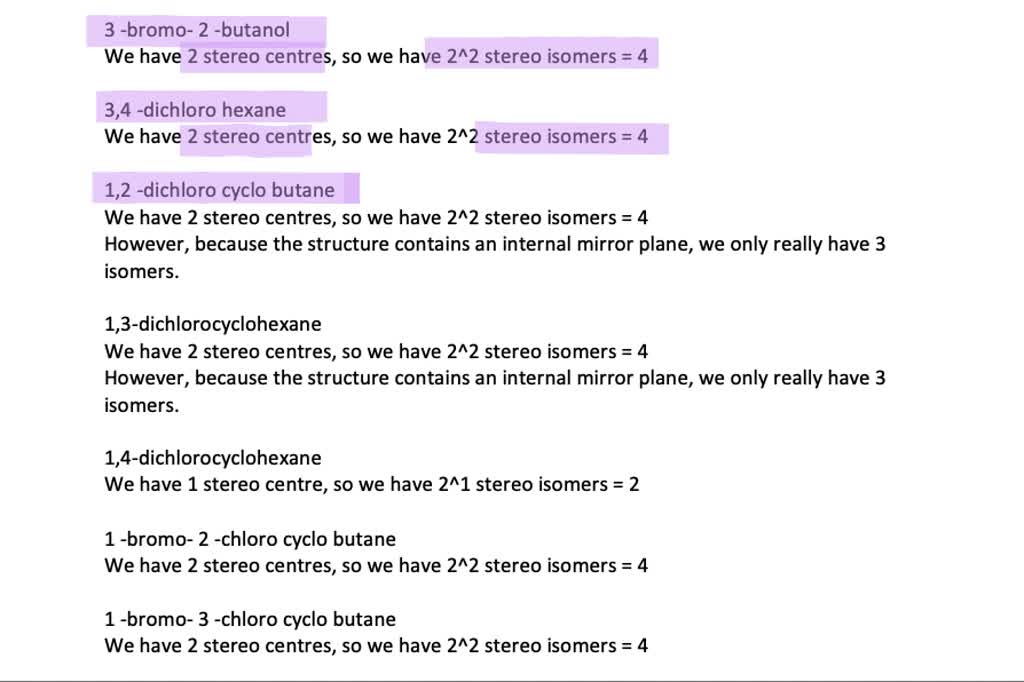
Let's draw the structure with Carbon 3 as our focus:
Enantiomer 1 (Let's call it the 'R' configuration for now, though we won't delve into R/S naming rules here):
- Imagine Carbon 3 at the center.
- Let's place the -CH3 group pointing down (solid line).
- The -Cl atom can be coming out towards us (wedge).
- The -H atom can be going back away from us (dash).
- And the -CH=CH2 group will be attached to Carbon 3, extending to the left.
So, you'd draw Carbon 3 with CH3 below, Cl in front, H behind, and the double bond group extending to the side.

Enantiomer 2 (The mirror image):
To draw the mirror image, we simply swap the positions of the groups that were on wedges and dashes, or we reflect the entire arrangement. If in Enantiomer 1, -Cl was a wedge, in Enantiomer 2, it will be a dash, and vice-versa for the hydrogen. The groups attached to the single bonds will maintain their relative positions.
- Carbon 3 at the center.
- -CH3 group pointing down (solid line).
- The -H atom can be coming out towards us (wedge).
- The -Cl atom can be going back into the plane away from us (dash).
- And the -CH=CH2 group extending to the left.
These two drawings will be perfect mirror images of each other, and you won't be able to overlay them perfectly, no matter how you rotate them.

Beyond Enantiomers: Diastereomers and Geometric Isomers
Now, you might be thinking, "Are there any other possibilities?" In this specific case of 3-chloro-1-butene, we only have one chiral center. This means we only have enantiomers. However, it's important to know that molecules with more than one chiral center, or molecules with double bonds in certain positions, can lead to other types of stereoisomers, like diastereomers and geometric isomers (also known as cis-trans isomers).
Geometric isomers arise when there's restricted rotation around a bond, typically a double bond, and there are different groups attached to the carbons of that double bond. In 1-butene, the double bond is between C1 and C2. C1 has two hydrogens, and C2 has one hydrogen and the -CHCl-CH3 group. Because C1 has two identical groups (the hydrogens), it cannot form geometric isomers.
Diastereomers occur when you have two or more chiral centers, and the stereoisomers are not mirror images of each other. They have different physical properties (like melting point and boiling point), unlike enantiomers which often have identical physical properties except for their interaction with polarized light.
So, for 3-chloro-1-butene, we have two stereoisomers, and they are enantiomers. Drawing them correctly requires careful attention to the 3D arrangement around the chiral carbon. It's a fantastic exercise that sharpens your spatial reasoning and lays the foundation for understanding the complex and fascinating world of molecular interactions.