Draw All Constitutional Isomers Of C4h9br

Hey there, science curious folks! Ever look at something simple, like a LEGO brick, and realize it can be put together in a whole bunch of different ways? That’s kind of what we’re diving into today, but with a tiny bit of chemistry. We’re going to explore something called "constitutional isomers" and how they apply to a molecule with four carbon atoms, nine hydrogen atoms, and one bromine atom – a rather friendly-sounding character we'll call C4H9Br.
Now, before you start imagining complicated lab coats and bubbling beakers, let’s take a deep breath. This isn't about becoming a chemistry whiz overnight. It's about understanding a cool concept that shows up more often than you think in the world around us. Think of it like recognizing different breeds of dogs. They're all dogs, right? They have four legs, a tail, they bark, but a Golden Retriever looks and acts quite differently from a tiny Chihuahua. Same "dogness," different arrangements!
So, what exactly are these "constitutional isomers"? Imagine you have a set of building blocks – let’s say four black blocks (our carbon atoms) and nine little white ones (our hydrogen atoms), plus one special red block (our bromine atom). Constitutional isomers are like building the exact same structure using those blocks, but connecting them in different orders. The number and type of blocks are the same, but the arrangement is unique.
Must Read
In our case, C4H9Br means we have four carbons, nine hydrogens, and one bromine. The "Br" is like our special connector piece, and it's only going to connect to one of the carbon atoms. The carbons will link up with each other, and the hydrogens will fill in the remaining spots to make everyone happy and stable.
Why should we care about this, you ask? Well, even though these molecules look identical on paper (same formula!), they can behave completely differently in the real world. It’s like having two identical-looking boxes, but one is filled with delicious cookies and the other with… well, let’s just say something less tasty. The outside is the same, but the inside experience is worlds apart!
This difference in behavior is super important in things like medicine. A drug molecule might have a specific shape and arrangement to fit perfectly into a target in your body, like a key in a lock. If you change that arrangement even a little bit, the "key" might not fit anymore, and the drug won't work, or worse, it could have harmful side effects. So, chemists need to be able to draw and understand all these possible arrangements to make sure they’re creating the right stuff.
Let's get our hands (virtually!) dirty and draw these possibilities for C4H9Br. We'll start by imagining our four carbon atoms as a chain, like a string of pearls.
The Straight and Narrow Chain
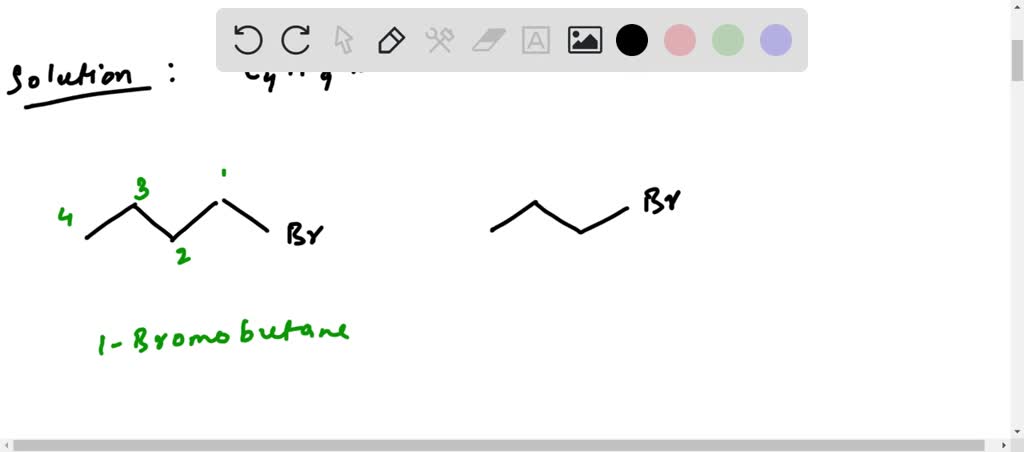
The simplest way to connect four carbon atoms is in a straight line. Think of it as a perfectly straight road. We'll call this the main chain. So, we have C-C-C-C. Now, we need to attach our nine hydrogens and our one bromine. The bromine can go on any of the carbons, but since this is a straight chain, the ends are identical, and the middle two are identical. Let’s put our bromine on one of the end carbons. We’ll have:
CH3-CH2-CH2-CH2-Br

This is one possibility, and it’s called 1-bromobutane. Think of it as the "straight-laced" isomer, very predictable.
What if we put the bromine on one of the inner carbons? Since the two ends are the same, and the two middle carbons are the same, putting it on either middle carbon will give us the same molecule. Let’s try it:
CH3-CH2-CH(Br)-CH3
This one is called 2-bromobutane. It’s like a slightly more interesting road, with a little detour. Notice how the bromine is now attached to a carbon that’s also attached to two other carbons. This is a key difference!
Branching Out!
But wait, can we arrange those four carbon atoms differently? Remember our LEGO analogy? We don’t have to make a straight line. We can make a branch! Imagine taking that straight chain and bending it a bit.
Let's think about a main chain of three carbon atoms. That leaves us with one extra carbon atom that needs to be attached somewhere. We can't just stick it on the end, because that would just make our straight chain again. We have to attach it as a branch. So, we have:

C-C-C
|&
C
This is our branched carbon skeleton. Now, we need to attach our nine hydrogens and our one bromine. Where can our bromine go?
Possibility 1: Bromine on the Main Chain
Let’s attach the bromine to one of the carbons on the main three-carbon chain. If we put it on either of the end carbons of the main chain, we get the same molecule. Let’s try that:
Br
|
CH3-CH-CH3
|
CH3
This molecule is called 2-bromo-2-methylpropane. This is a bit of a mouthful, so it often gets a fun nickname: tert-butyl bromide. Think of it like a little cluster of atoms, all huddled together. The bromine is attached to a carbon that’s connected to three other carbon atoms. This is quite different from our previous examples!
Possibility 2: Bromine on the Branch
What if we attach the bromine to that little single carbon that’s sticking out as a branch? Let’s see:
CH3
|
CH3-CH2-CH-Br
This molecule is called 1-bromo-2-methylpropane. It’s like the straight chain with a little side-shop. The bromine is on a carbon that’s connected to only one other carbon. Notice how the carbon skeleton here is essentially a three-carbon chain with a methyl group attached to the second carbon.
Are We Done Yet?
So, let's recap. We started with the formula C4H9Br and we've come up with:
- 1-bromobutane (straight chain, Br on the end)
- 2-bromobutane (straight chain, Br in the middle)
- 2-bromo-2-methylpropane (branched, Br on the central carbon of the main chain)
- 1-bromo-2-methylpropane (branched, Br on an outer carbon of the main chain)
It might seem like we could jiggle things around more, but if you try to create any other arrangement with four carbons, nine hydrogens, and one bromine attached to a carbon, you’ll find you just end up recreating one of these four. It’s like trying to build a different house with the same set of LEGOs – you can rearrange them, but you’ll always end up with the same fundamental structure if you stick to the rules.
Each of these molecules, while sharing the same ingredients (C4H9Br), has a unique blueprint. This difference in blueprint is what gives them their distinct personalities and makes them behave differently. This is why understanding constitutional isomers is so crucial, not just for scientists in labs, but for anyone who wants to understand how the world works at a fundamental level. It's the tiny details, the little twists and turns in molecular structure, that make all the difference!
So, the next time you hear about a molecule, remember that its formula is just the ingredient list. The real story is in how those ingredients are put together. And for C4H9Br, there are four fascinating ways to arrange those atoms, each with its own set of properties and potential!