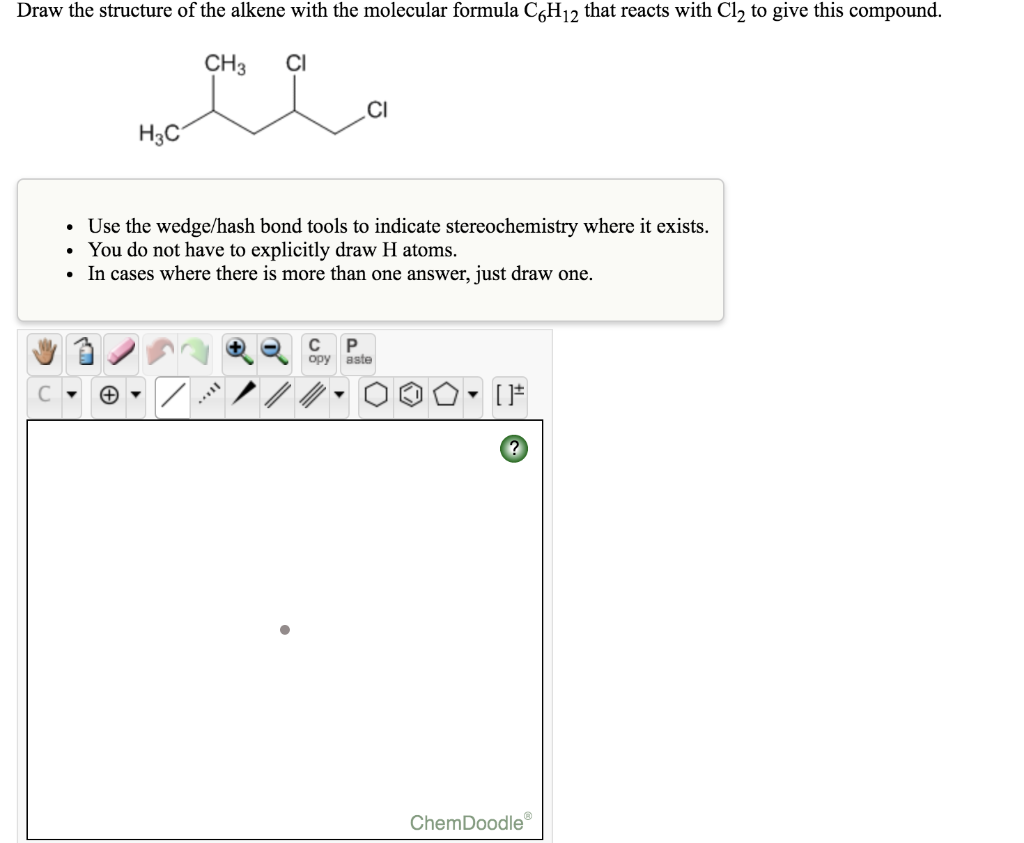
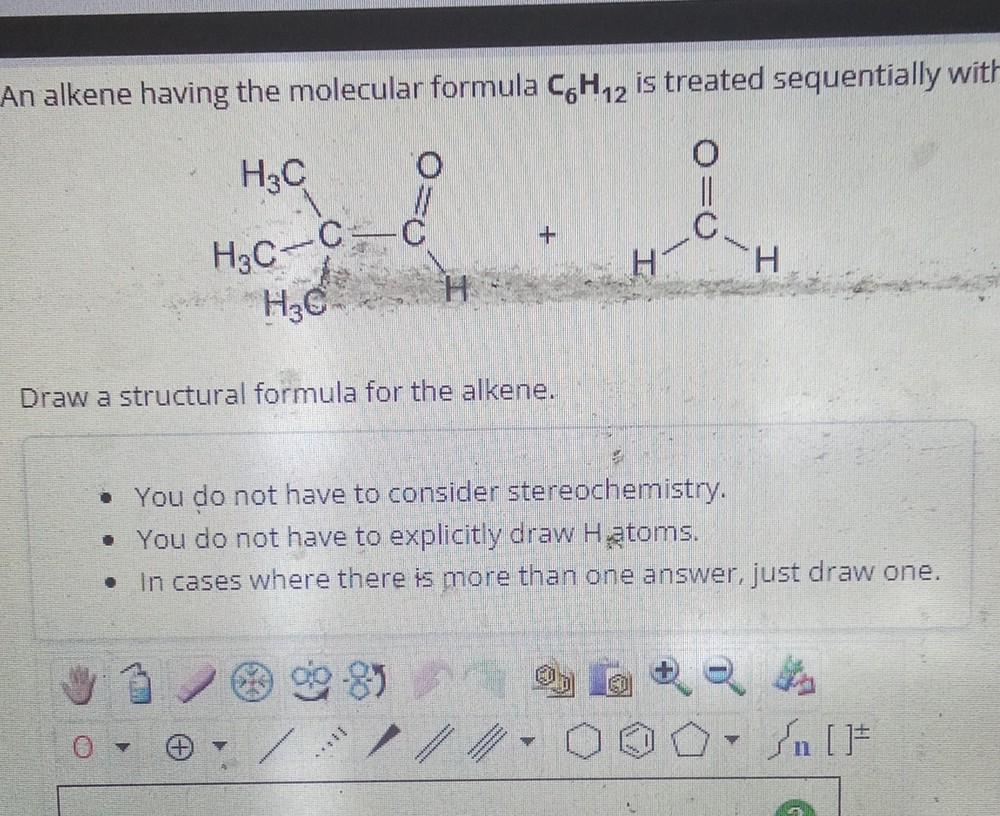
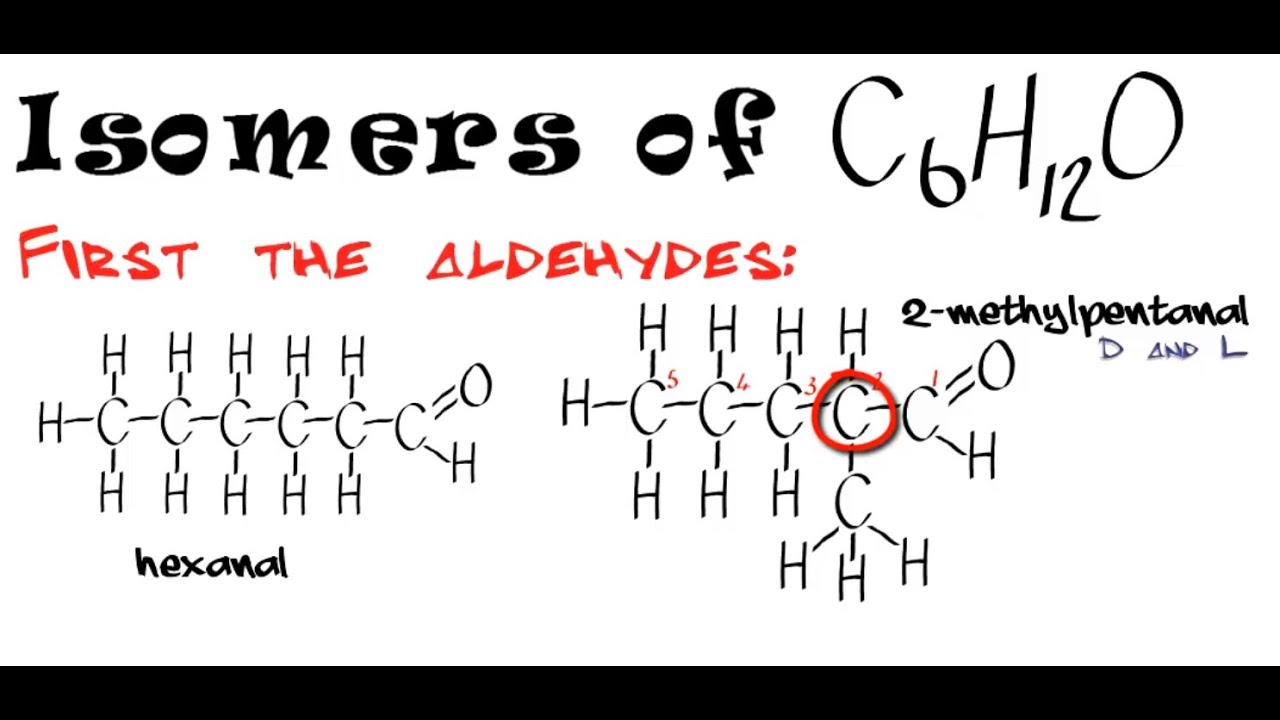
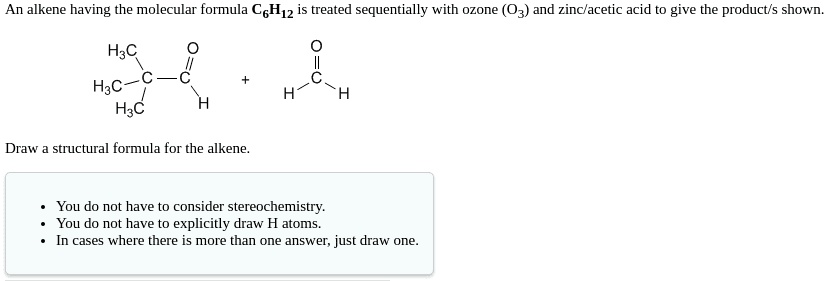
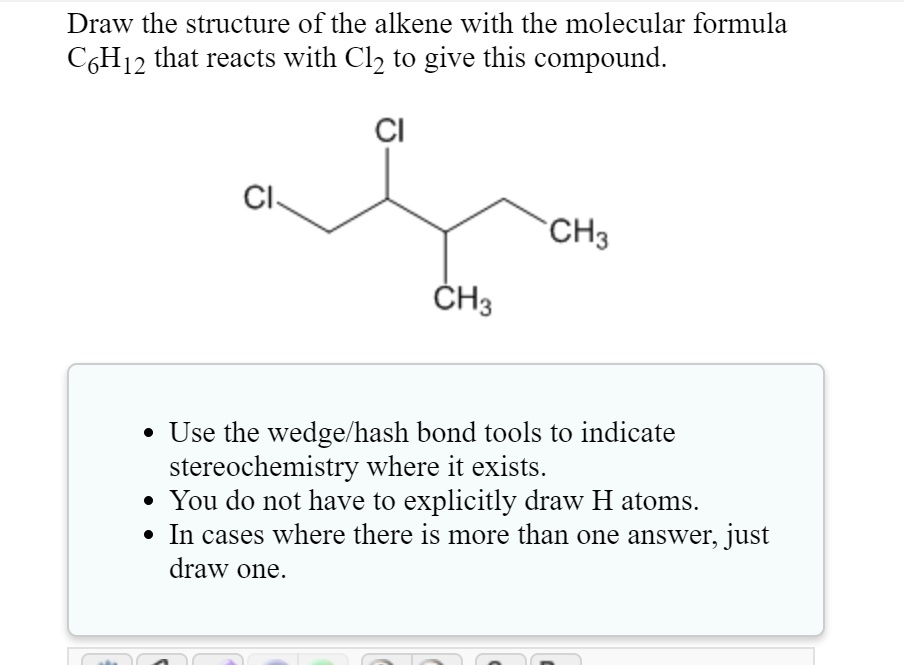
Draw A Chiral Alkene With The Formula C6h12

Ever played with LEGOs? You know how some pieces can fit together in only one way? Well, imagine a molecule that's a bit like that, but with a twist! We're going to explore a super cool molecule with the formula C6H12. That just means it has 6 carbon atoms and 12 hydrogen atoms. Easy peasy, right?
But here's where the fun begins. This C6H12 molecule can be drawn in a way that's a little bit like having a right hand and a left hand. Sounds weird, but stick with me! It's all about a special arrangement called chirality. Think of it like this: your hands are mirror images of each other, but you can't put your right glove on your left hand, can you? They just don't fit perfectly. Molecules can do the same thing!
The particular type of molecule we're talking about has a double bond between two carbon atoms. This is what makes it an alkene. The double bond is like a very strong handshake between two carbons. And when this special handshake happens in the right way, it opens up the door for this whole "handiness" thing.
Must Read
So, what makes drawing a chiral alkene with the formula C6H12 so entertaining? It's like solving a fun little puzzle. You have to think about how the atoms are arranged in space, not just in a flat drawing. It's like looking at a 3D sculpture versus a flat drawing of that sculpture. One gives you so much more information and allows for really interesting possibilities.
Imagine you're building with those LEGOs, but instead of just snapping them together, you have to consider which way they're pointing. For our C6H12 alkene, the double bond is the key. Around this double bond, the atoms have to be arranged in a specific order to make it chiral. If you swap just two of those atoms, you might end up with its mirror image, its "other hand."
Why is this special? Well, in the world of chemistry, these different "hands" can behave completely differently. Sometimes, one hand might be great for making a medicine that cures an illness, while its mirror image could be completely useless, or even harmful! It's like having a key that only fits a specific lock. The shape matters, and for these chiral molecules, the shape is determined by their "handedness."
Let's get a little more visual. For a chiral alkene with C6H12, we need to make sure that each of the two carbons involved in the double bond is attached to four different groups. This is the magic ingredient for chirality in alkenes. If any of those carbons have two of the same groups attached, then it can't be chiral. It's like trying to make a hand where two fingers are identical – it just doesn't work that way!
So, we're looking for a C6H12 molecule where there's a carbon-carbon double bond, and each of those carbons has four unique things sticking off of it. Think of those unique things as different colored LEGO bricks. You need four different colors on each of the double-bonded carbons. When you achieve this, you get two versions of the molecule: one that's like your right hand and one that's like your left hand. They're both C6H12, they both have that double bond, but their spatial arrangement is different.

It's a bit like looking in a funhouse mirror that shows you a slightly different, but still recognizable, version of yourself. These aren't just abstract drawings; they represent real molecules that scientists work with every day. Understanding how to draw and identify them is a fundamental skill in chemistry, and it all starts with recognizing that special arrangement around that double bond.
The beauty of it is that you can experiment with different ways to attach the groups. You can swap them around, and as long as you maintain that rule of "four different groups on each double-bonded carbon," you'll still have a chiral alkene. It's like rearranging the LEGO bricks to create slightly different but equally valid structures. The challenge and the fun come from visualizing these 3D arrangements and understanding their implications.
So, next time you hear about a molecule like C6H12, remember that it's not just a jumble of atoms. It can have a hidden complexity, a sense of "handedness" that makes it incredibly special. Drawing a chiral alkene with this formula is like uncovering a secret handshake in the molecular world. It's a little puzzle that reveals a lot about the intricate and fascinating nature of chemistry. Who knew playing with atoms could be so much like playing with hands or LEGOs?