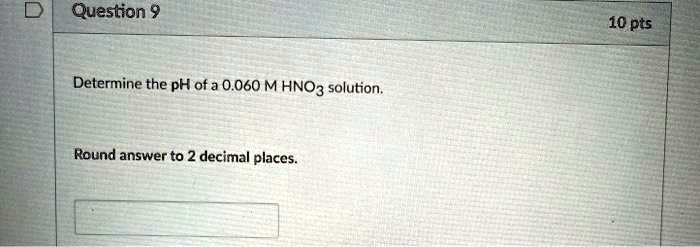
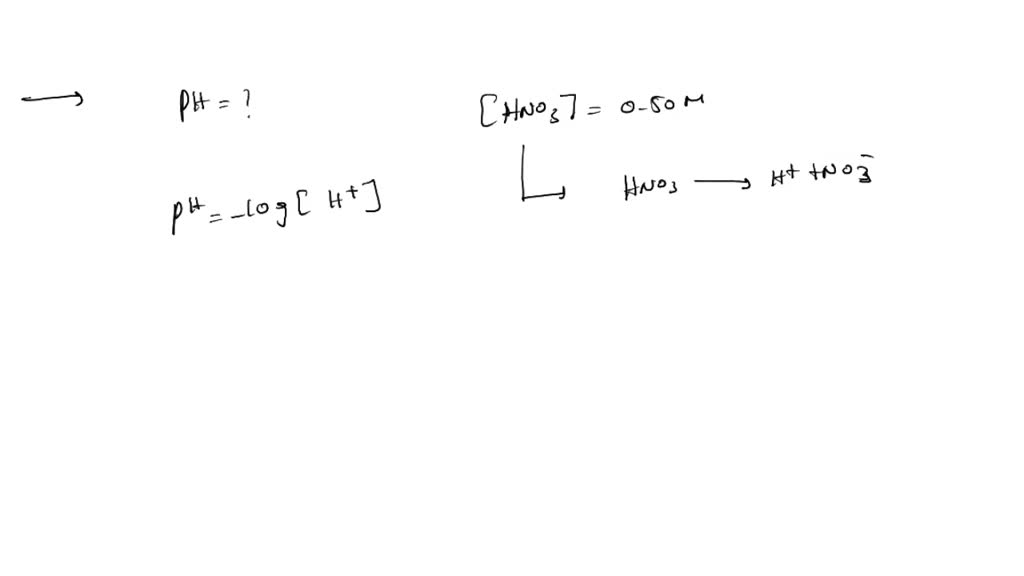Determine The Ph Of A 0.010 M Hno3 Solution

Ever stared at a blank canvas, a lump of clay, or a string of code and felt that spark of possibility? The world of creation is vast and wonderfully accessible, and sometimes, the most profound inspiration comes from the most unexpected places. Take, for instance, the seemingly simple act of understanding the pH of a 0.010 M HNO₃ solution. While it might sound like something straight out of a science lab manual, there's a surprising amount of creative juice to be squeezed from this very concept!
For artists and hobbyists, diving into the world of chemistry, even with a basic solution like nitric acid, can be an incredibly enriching experience. Imagine the vibrant hues you could achieve by exploring acid-base reactions in your paint pigments! Or perhaps you're a digital artist looking for unique visual metaphors; the idea of concentration and balance, inherent in pH calculations, can translate into compelling graphic designs. For casual learners, it's a gateway to understanding the fundamental building blocks of our world, demystifying the science that surrounds us every day. It’s about empowering yourself with knowledge, one fascinating concept at a time.
Think about the possibilities! An artist might experiment with creating aged paper effects using controlled acid solutions, achieving a vintage aesthetic. A sculptor could explore how different chemical concentrations affect the curing of certain materials, leading to entirely new textural variations. Even a writer could find inspiration in the precise language of chemistry, weaving scientific concepts into fantastical narratives. The beauty lies in the adaptability of the idea. It’s not just about the specific solution; it’s about the principle of measurement, concentration, and reaction.
Must Read
Ready to dip your toes into this fascinating realm? Trying this at home, with the right safety precautions of course, is more straightforward than you might think. For understanding the pH of a 0.010 M HNO₃ solution, you'll need a calculator and the basic formula for calculating pH from hydrogen ion concentration: pH = -log[H⁺]. Since HNO₃ is a strong acid, it dissociates completely, meaning the concentration of H⁺ ions is equal to the initial concentration of HNO₃. So, in this case, [H⁺] = 0.010 M. Plug that into your calculator: pH = -log(0.010). You'll find it's a nice, round number! Always remember to prioritize safety. If you're working with any acids, even dilute ones, wear appropriate safety glasses and gloves, and ensure good ventilation. Start with extremely dilute solutions and understand the properties of the chemicals you are using.
Ultimately, what makes exploring concepts like the pH of a 0.010 M HNO₃ solution so enjoyable is the sense of discovery. It’s about connecting seemingly disparate fields and realizing that science, art, and even everyday curiosity are all interwoven. It’s a chance to unleash your inner investigator and find joy in the elegant logic of the universe. So go forth, explore, and let the spark of understanding ignite your creativity!