Conversion Of Cis 2-butene To Trans 2-butene

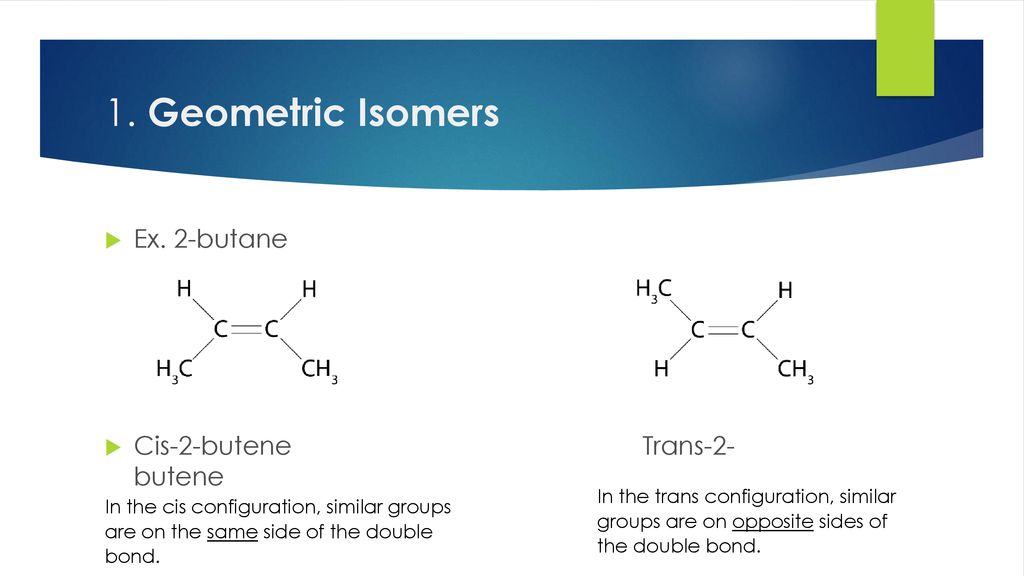
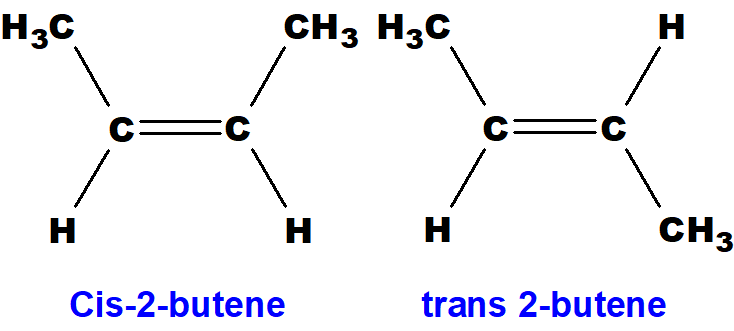
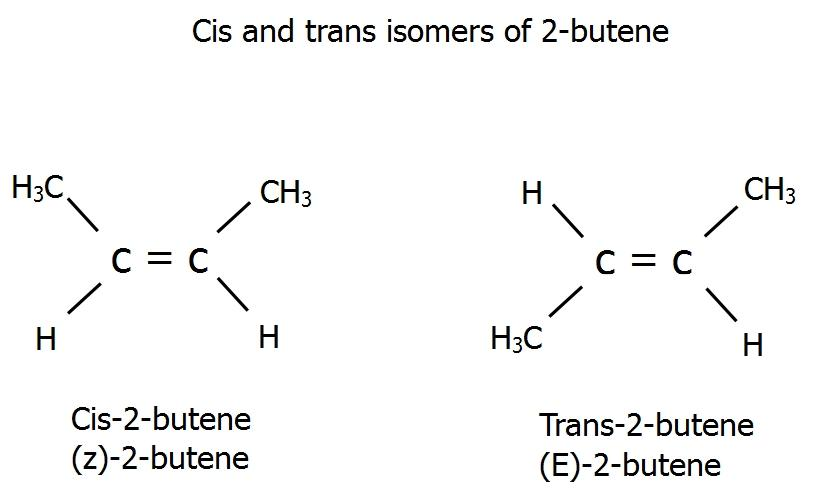
Hey there! Grab a cuppa, you’re gonna want to hear this. We’re gonna dive into a little chemical shapeshifting, a real molecular makeover, if you will. Imagine you’ve got this molecule, cis-2-butene. Sounds fancy, right? It’s basically a four-carbon chain with a double bond smack dab in the middle. The cool thing about cis is that the two little hydrogen atoms sticking off the double bond are on the same side. Think of them like best buds, always hanging out together. They’re cozy, they’re close, and honestly, a little bit crowded.
Now, its buddy, trans-2-butene, is the exact same molecule, same atoms, same everything, but with a crucial difference. Those two hydrogen atoms? They’re on opposite sides of the double bond. They’re like, “Nah, we need our personal space, man.” This subtle switcheroo might seem small, but oh boy, it changes things. It’s like switching from wearing sweatpants to a fancy suit. Same person, totally different vibe, you know?
So, why would we even want to mess with this? Why bother turning our cuddly cis isomer into the more aloof trans isomer? Well, sometimes the trans version is just… better. Maybe it’s more stable, like a person who’s had a good night’s sleep versus someone who’s been up all night. Or maybe it fits into a certain reaction pathway more perfectly, like a puzzle piece snapping into place. It’s all about that chemical efficiency, baby! We’re always looking for the most streamlined way to do things, aren't we? Especially in the lab, where time is money, and frankly, so is patience.
Must Read
The most common way we whip cis-2-butene into trans-2-butene is through a little something called isomerization. Think of it as a chemical spa treatment. We’re not adding or removing anything, just rearranging the existing bits. And how do we achieve this magical rearrangement? Usually, we throw in a catalyst. These are like the fairy godmothers of chemistry, speeding up reactions without actually getting used up themselves. They’re the ultimate wingmen for molecules.
One of the star players in this isomerization game is often a metal catalyst. Think platinum, palladium, nickel – those shiny, precious metals we associate with fancy jewelry, but here they're busy making molecules do our bidding. They're like the bouncers at a club, creating the perfect environment for the molecules to mingle and change their positions. They provide a surface, a little dance floor if you will, where the cis molecule can get a little… uncomfortable.
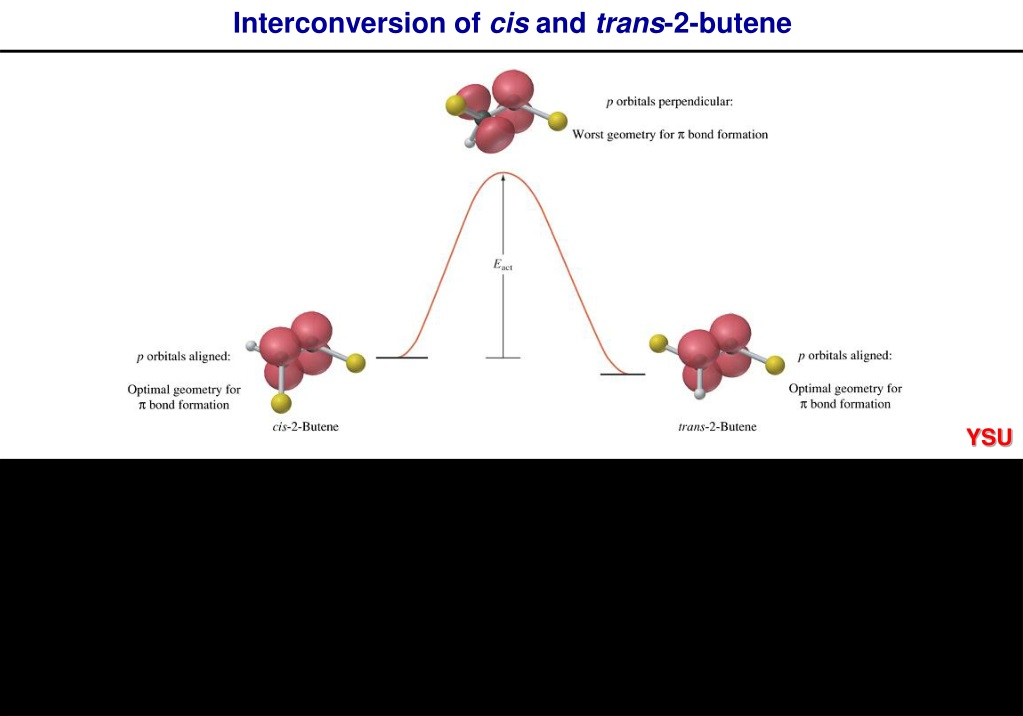
Here’s where it gets a bit more technical, but stick with me, it’s still pretty cool. The double bond in alkenes (that's what butene is) is a bit like a sticky situation. It’s got a strong sigma bond, which is pretty rigid, and a weaker pi bond above and below it. This pi bond is the key player here. It’s more accessible, more… available for interaction. So, when our metal catalyst comes along, it can interact with this pi bond. It's like it’s giving the double bond a little hug, or maybe a gentle nudge.
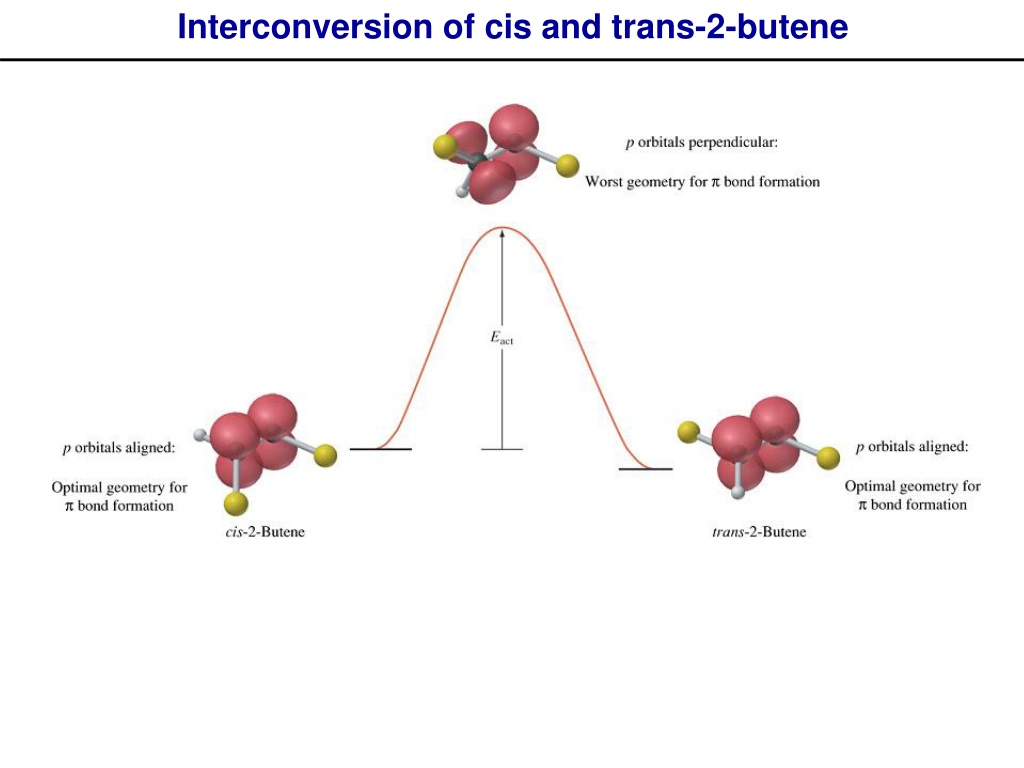
This interaction weakens the double bond just enough. Not enough to break it completely (we don't want a full meltdown!), but enough to allow for some rotation. Imagine you're trying to twist a pretzel. It’s tough at first, right? But if you could loosen the dough a little, it becomes much easier. That’s kind of what the catalyst is doing. It’s loosening up the double bond's grip.
Once that rotation can happen, the molecule can start to untwist. The two hydrogen atoms, which were previously bumping shoulders on the same side, can now swing around. They can go from being huddled together to being on opposite sides of the molecule. It’s like they’ve decided to go their separate ways, at least for now. And voilà! You’ve just converted your cis isomer into the more spread-out trans isomer. Pretty neat, huh?
Now, this reaction isn't always a simple one-and-done deal. Sometimes, it happens in the presence of hydrogen gas. Why hydrogen? Well, it’s a bit like a sacrificial lamb, or perhaps a helpful friend. The hydrogen can add to the double bond, breaking it and forming a single bond. Then, one of the hydrogen atoms can leave, and as it does, the molecule can re-form a double bond, but this time, in the more stable trans configuration. It’s like a quick detour to break things down and then build them back up, but with a different structure.
This process, where hydrogen is involved, is often called catalytic hydrogenation, followed by a little dehydrogenation to get that double bond back. It’s a bit of a dance – add, then remove. But the end goal is always that trans product. Think of it as a temporary disassembly to achieve a more favorable reassembly. It's like taking apart a Lego castle and rebuilding it to be sturdier and more aesthetically pleasing.
Another interesting way to coax cis-2-butene into its trans cousin is by using a bit of heat. Yep, sometimes a good ol’ fashioned temperature increase is all it takes. This is known as thermal isomerization. At higher temperatures, molecules have more kinetic energy, meaning they’re jiggling and wiggling around with more gusto. This extra energy can be enough to overcome the rotational barrier of the double bond, allowing those hydrogens to switch sides.
However, thermal isomerization can be a bit of a blunt instrument. It’s like turning up the heat on a pot of water – it gets things moving, but you might end up boiling over if you’re not careful. High temperatures can sometimes lead to side reactions, where the molecule decides to do something completely different and unwanted, like breaking down into smaller pieces or forming even more complex structures. So, while it can work, it’s not always the cleanest or most efficient method. We usually prefer our catalysts for a more controlled transformation.
And what about light? Could we use a flashlight to flip our molecules? Well, yes and no. Sometimes, certain types of light, specifically ultraviolet (UV) light, can provide the energy needed to promote a molecule from its ground state to an excited state. In this excited state, the molecule is more reactive, and the double bond can twist. Think of it like giving the molecule a little zap of energy that makes it more flexible.

When a molecule like cis-2-butene absorbs UV light, it can enter an excited state where the pi bond is more easily broken, allowing for rotation. Once it relaxes back to its ground state, it can end up as either the cis or the trans isomer. It’s kind of a bit of a gamble, this photoisomerization. You might get more of the desired trans isomer, or you might end up with a mixture of both. It’s like flipping a coin, but with molecules! This method is often called photoisomerization, and while it’s a fascinating concept, it’s not always the most practical for large-scale production.
So, we’ve got our catalysts, our hydrogen, our heat, and even our light. Which one is the best? Well, that depends on what you’re trying to achieve! For pure efficiency and control, the metal-catalyzed isomerization is usually the way to go. It’s precise, it’s predictable, and it generally gives you a good yield of the trans product without a whole lot of fuss. It’s the reliable workhorse of the cis-to-trans conversion world.
The choice of catalyst is also super important. Different metals have different affinities for the alkene. Some might be more aggressive, while others are gentler. It’s like picking the right tool for the job. You wouldn’t use a sledgehammer to crack a nut, would you? Similarly, you wouldn’t use a super-reactive catalyst if you just wanted a subtle tweak. We’re talking about subtle adjustments, not molecular demolition!
And let’s not forget the solvent! The medium in which the reaction takes place can also play a role. Sometimes, a polar solvent can help stabilize certain intermediates, while a non-polar solvent might favor others. It’s like choosing the right social setting for your molecules. Do they need a lively atmosphere, or a more calm and collected environment to do their thing? It all matters!
Think about the implications of this. This isn’t just some abstract chemistry problem. These kinds of isomerizations are happening all the time in the industrial world. They’re used to make all sorts of useful chemicals, from plastics to pharmaceuticals. Imagine the sheer volume of cis-2-butene that gets nudged into becoming trans-2-butene every single day. It’s mind-boggling when you stop and think about it. We’re literally reshaping the building blocks of modern life with these subtle molecular shifts.
It’s also a great example of how even small changes in molecular structure can have big consequences. The difference in stability between cis and trans isomers is often significant. The trans isomer, with its more spread-out groups, generally has less steric hindrance. That’s a fancy way of saying there’s less crowding between atoms. Less crowding means less repulsion, and less repulsion means a more stable molecule. It’s like the difference between trying to fit into a crowded elevator versus having your own personal space. The latter is always going to feel more comfortable and less stressful, right?
So, next time you see a plastic bottle or a pill, just remember that somewhere along the line, a molecule might have gone through a little makeover, a subtle shift from a crowded ‘cis’ existence to a more spacious ‘trans’ one, all thanks to the magic of chemistry and a good catalyst. It’s the unsung heroes of the molecular world, doing their important work behind the scenes. Who knew such tiny changes could make such a big difference? It’s pretty inspiring, if you ask me.
And there you have it! A little peek into the fascinating world of cis-2-butene and its transformation into trans-2-butene. It’s a testament to the elegance and power of chemistry, and how we can harness these principles to create the materials and medicines we rely on every day. Who’s ready for another cuppa? We’ve barely scratched the surface of what these molecules can do!