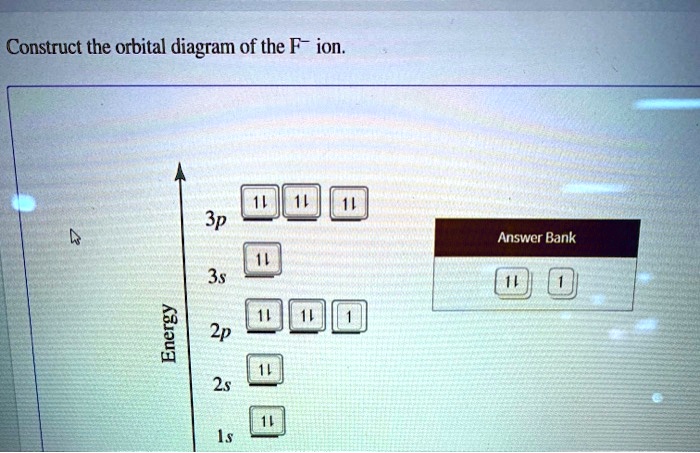
Construct The Orbital Diagram Of The F Ion

Hey there, science curious folks! Ever found yourself staring up at the night sky, wondering about all those tiny specks of light and the incredible stuff they're made of? Well, today, we're going to zoom in, way, way in, and talk about something super cool happening at the atomic level. We're going to be building an orbital diagram for the F- ion. Sounds a bit technical, right? But stick with me, it's actually like putting together a really neat puzzle, and the pieces are all about how electrons arrange themselves. Think of it like assigning seats to guests at a very exclusive party!
So, what exactly is an ion, you ask? Imagine an atom is like a perfectly balanced scale. It has protons (positive charges) and electrons (negative charges) that cancel each other out, making it neutral. An ion is just an atom that's decided to either gain or lose a little bit of that balance. In the case of our F- ion, the 'minus' sign tells us it's gained an electron. It’s like that atom decided it needed one more friend to feel complete, or maybe it just found an extra electron hanging around and thought, "Why not?"
Now, before we dive into the diagram, let's get a feel for the star of our show: Fluorine. Fluorine, or 'F', is element number 9 on the periodic table. This means a neutral Fluorine atom has 9 protons and, you guessed it, 9 electrons. These electrons aren't just floating around randomly; they have favorite spots to hang out in, kind of like how we have our favorite rooms in a house. These favorite spots are called orbitals.
Must Read
Think of orbitals as different types of rooms. We have the 's' rooms, which are nice, simple, spherical rooms, and the 'p' rooms, which are a bit more complex, shaped like dumbbells. Each orbital can hold a maximum of two electrons. It’s a strict rule, like only two people allowed in a small elevator! And just like in a house, there are different floors or energy levels. The first floor (n=1) is the lowest energy, and as you go up in floors (n=2, n=3, and so on), the energy levels increase.
So, in a neutral Fluorine atom, we’ve got 9 electrons to place. We fill the lowest energy levels first. The first floor (n=1) has one 's' orbital. This 1s orbital can hold two electrons. So, we fill that up: 1s2. We've used 2 electrons, and we have 7 left.
Next, we move to the second floor (n=2). This floor has a bit more variety. It has another 's' orbital (the 2s orbital), which also fits two electrons. So, we fill that up too: 2s2. Now we've used a total of 2 + 2 = 4 electrons, and we have 5 remaining. Where do these last 5 go?
The second floor also has three 'p' orbitals (the 2p orbitals). These are like three separate rooms, all on the same floor, and each can hold two electrons. So, we have three 2p orbitals available. We need to put our remaining 5 electrons into these 2p orbitals. The rule here is that electrons like to spread out and have their own space first, kind of like how people at a party might grab their own chair before doubling up. So, we put one electron in each of the three 2p orbitals (2px, 2py, 2pz) until they're half-full. That’s 3 electrons used.
We still have 2 electrons left, and our 2p orbitals are all occupied by one electron. Now, these electrons will pair up. So, we add one more electron to each of two of the 2p orbitals. This means our 2p orbitals will end up with 2p5 (3 single electrons + 2 paired electrons). So, the electron configuration for neutral Fluorine is 1s22s22p5. Pretty neat, right?
Now, for the star of our show today: the F- ion. Remember that minus sign? It means our Fluorine atom has gained one electron. So, instead of 9 electrons, we now have 9 + 1 = 10 electrons to place. We’re basically adding one more electron to our neutral Fluorine setup.

Constructing the Orbital Diagram for F-
Let's visualize this. The orbital diagram is like a little map. We use boxes or lines to represent the orbitals, and arrows to represent the electrons. An upward arrow is often used for one electron, and a downward arrow for the other when they're paired up in the same orbital.
Level 1: The 1s Orbital
We start with the lowest energy level, n=1. There's one 1s orbital. It can hold 2 electrons. So, we draw one box and put two arrows in it, one pointing up and one pointing down. This represents 1s2.
[↑↓]
Level 2: The 2s and 2p Orbitals
Next, we move to the second energy level, n=2. This level has a 2s orbital and three 2p orbitals. The 2s orbital is lower in energy than the 2p orbitals on the same level.
First, the 2s orbital. It can also hold 2 electrons. So, we draw another box next to the 1s box and fill it with two arrows: 2s2.
[↑↓] [↑↓]
(Where the first box is 1s and the second is 2s)
Now for the 2p orbitals. We have three of them (let's call them 2px, 2py, 2pz). They are all at the same energy level. We have 5 electrons to put into these three orbitals in our neutral Fluorine. We first spread them out:
[↑ ] [↑ ] [↑ ]
But wait! We have 5 electrons in total for the 2p subshell of neutral Fluorine. So, we add the remaining two electrons, which pair up:
[↑↓] [↑↓] [↑ ]
This is for neutral Fluorine: 2p5.
Okay, here’s where the F- ion comes in! Remember, it gained one electron. Where does this extra electron go? It goes into the lowest available energy spot. In neutral Fluorine, the 2p orbitals are where we have some "space" – specifically, one orbital has only one electron. So, our extra electron will pair up with that lonely electron in one of the 2p orbitals.
So, for the F- ion, the 2p subshell will have a total of 6 electrons (5 from neutral Fluorine + 1 gained electron). And with 6 electrons in three orbitals, each orbital gets exactly two electrons!
Our orbital diagram for F- will look like this:
1s2: One box with two arrows (↑↓)
2s2: One box with two arrows (↑↓)
2p6: Three boxes, each with two arrows (↑↓) (↑↓) (↑↓)
So, the complete orbital diagram for F- is:
[↑↓] [↑↓] [↑↓] [↑↓] [↑↓]
(Where the first box is 1s, the second is 2s, and the next three are the 2p orbitals)
Isn’t that cool? The F- ion has completely filled its 1s, 2s, and 2p orbitals. It's like all the "rooms" are perfectly occupied with two guests each! This makes the F- ion very stable. It's achieved a full outer shell, which is a really happy state for an atom or ion. Think of it as having all your favorite comfortable chairs taken – no more need to look for anything!
This stable configuration is why Fluorine is so keen to grab that extra electron. It's essentially aiming for this peaceful, full arrangement. It’s like a gamer achieving a perfect score or a baker perfecting a recipe – a state of completion and satisfaction.
So, the next time you see that little minus sign next to an element symbol, you'll know it means something special is happening with its electron arrangement, leading to potentially new and interesting properties. Building these diagrams helps us understand not just what atoms and ions are made of, but how they're put together, and why they behave the way they do. It’s a little peek into the amazing, organized chaos of the universe at its smallest scale!

![[DIAGRAM] F Block Orbital Diagram - MYDIAGRAM.ONLINE](https://www.chemtube3d.com/images/4forbitalsnew.png)
