Construct The Orbital Diagram For Arsenic
Ever found yourself gazing at the night sky, a little bit mesmerized by the sheer vastness of it all? That feeling of wonder is exactly what we're tapping into today, but instead of planets and galaxies, we're zooming in on something a little closer to home: arsenic. Now, before you go picturing nefarious potions, let’s chill. Arsenic, element number 33 on the periodic table, is actually a fascinating character in the atomic world, and understanding its orbital diagram is like getting a backstage pass to its atomic personality. Think of it as getting to know your favorite indie band by understanding their lyrical themes and musical influences. It's less about the hard science and more about the vibe.
So, what exactly is an orbital diagram? Imagine your atom is a tiny, bustling city. The nucleus is the central town hall, buzzing with protons and neutrons. The electrons? They’re the citizens, zipping around in their own designated neighborhoods or 'orbitals'. These orbitals aren't just random hangouts; they're specific regions of space where an electron is most likely to be found. Each orbital has its own unique shape and energy level. It’s like having different types of housing: some are cozy studios (s orbitals), others are more spread out bungalows (p orbitals), and some are even sprawling mansions (d and f orbitals). We’re going to chart out the atomic real estate for arsenic, and trust me, it’s more engaging than binge-watching a procedural drama.
Unpacking Arsenic's Atomic Address
To start building our orbital masterpiece for arsenic, we first need to know its atomic number. A quick peek at our trusty periodic table (that stylish infographic of all elements!) tells us arsenic has an atomic number of 33. This means a neutral arsenic atom has 33 electrons. Our mission, should we choose to accept it, is to figure out where all these electrons will settle down. It’s like assigning apartments to 33 new residents in our atomic city. We've got rules to follow, of course, but they're pretty straightforward once you get the hang of them.
Must Read
These rules are governed by fundamental principles of quantum mechanics, which, while sounding intimidating, are essentially just nature’s way of keeping things organized. The Aufbau principle is our primary guide here. It’s like the ultimate real estate agent, telling us to fill the lowest energy orbitals first. Electrons are inherently lazy in the best possible way; they prefer to be in the most stable, lowest-energy spots available. So, we start filling from the bottom up, like building a sandcastle, one scoop of sand at a time.
The Orbital Hierarchy: A Symphony of Energy Levels
Let's break down the types of orbitals we’ll encounter. We have:
- s orbitals: These are spherical, like perfectly round disco balls. Each energy level has one s orbital.
- p orbitals: These are dumbbell-shaped, with three of them oriented along the x, y, and z axes. Think of them as three interconnected pathways.
- d orbitals: These are more complex, with five of them, and they often look like cloverleaves. They’re the swankier apartments in our atomic city.
- f orbitals: Even more intricate, with seven of them, and usually found in the inner transition metals. We won’t need these for arsenic, but it’s good to know they exist – the ultra-luxury penthouses of the atomic world!
The energy levels are numbered 1, 2, 3, and so on, with higher numbers indicating higher energy. Within each energy level, the s orbital is the lowest in energy, followed by p, then d, and then f. It’s a predictable pecking order, ensuring the most efficient packing of electrons.
Building Arsenic's Orbital Diagram: Step by Step
Alright, let’s get our hands dirty (metaphorically, of course – we’re dealing with atoms here!). We need to fill 33 electrons. We’ll use a shorthand called electron configuration first, which is like a condensed version of the orbital diagram. Then, we’ll translate that into the visual representation.
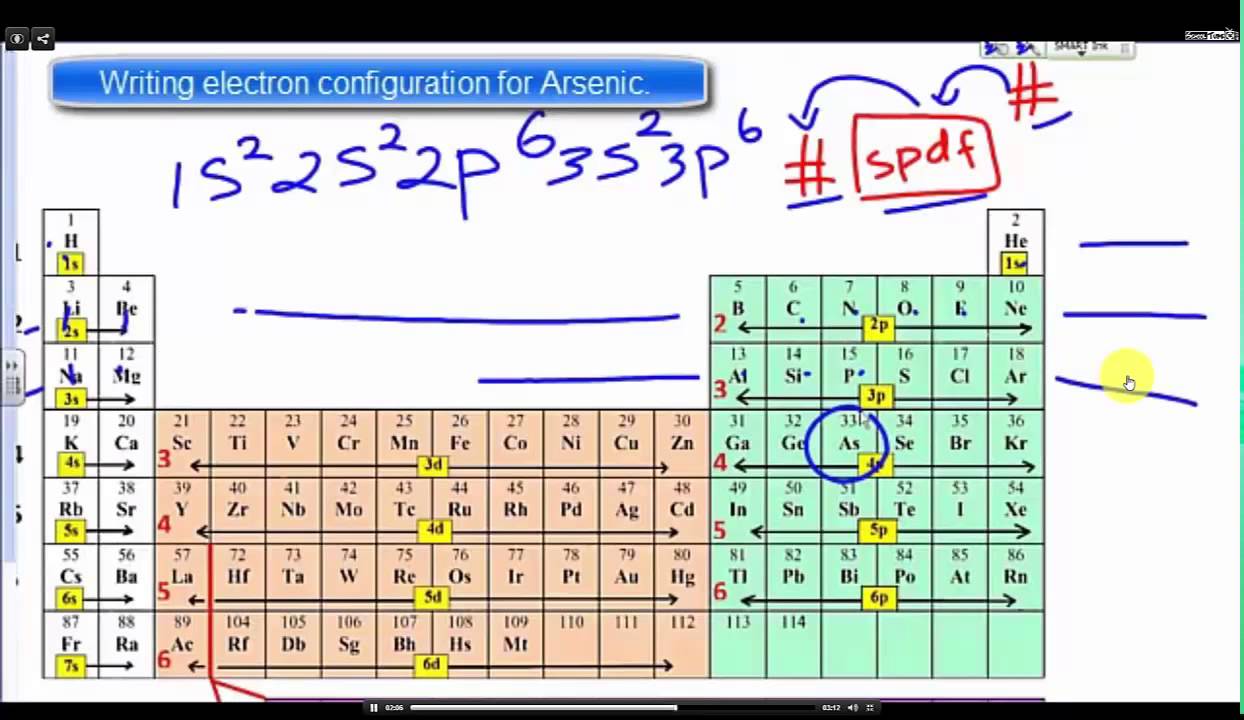
The order of filling orbitals, dictated by their increasing energy, looks like this: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, and so on. This sequence might seem a bit quirky because the 4s orbital fills before the 3d orbital, even though it's in a higher energy level. This is where things get a little more nuanced, like a plot twist in your favorite novel. The 4s orbital is actually slightly lower in energy than the 3d orbitals due to subtle quantum effects.

Now, how many electrons can each type of orbital hold?
- An s orbital can hold a maximum of 2 electrons.
- A p subshell (which has 3 p orbitals) can hold a maximum of 6 electrons (2 electrons per orbital x 3 orbitals).
- A d subshell (with 5 d orbitals) can hold a maximum of 10 electrons.
- An f subshell (with 7 f orbitals) can hold a maximum of 14 electrons.
Let’s start filling! We have 33 electrons to place:
- 1s: We start with the lowest energy level, 1s. It can hold 2 electrons. Our count is now 2.
- 2s: Next up is 2s. It also holds 2 electrons. Total: 4.
- 2p: The 2p subshell has 3 orbitals and can hold up to 6 electrons. Total: 10.
- 3s: Back to the 3s orbital, holding 2 electrons. Total: 12.
- 3p: The 3p subshell takes another 6 electrons. Total: 18.
- 4s: Now we move to the 4s orbital, which holds 2 electrons. Total: 20.
- 3d: Here comes the 3d subshell. It can hold up to 10 electrons. Total: 30.
- 4p: We have 3 more electrons to place (33 total - 30 placed). The 4p subshell is the next available in terms of energy. It can hold up to 6 electrons, so we'll place our remaining 3 electrons here. Total: 33.
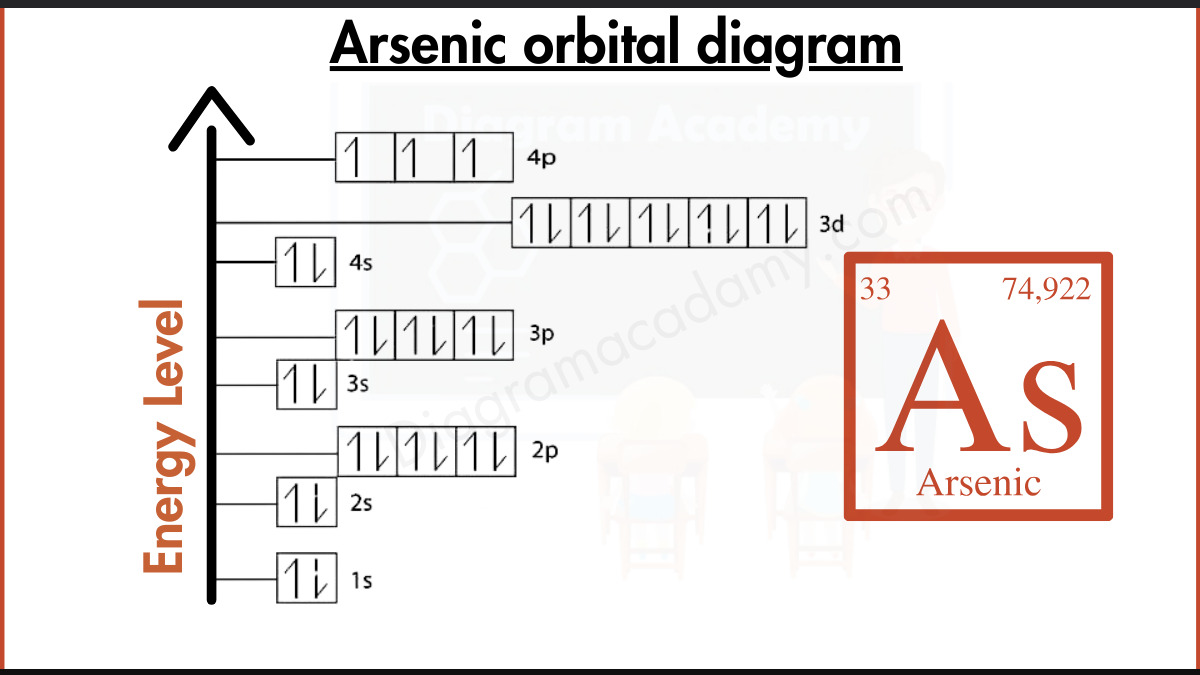
So, the electron configuration for arsenic is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p³. This is our atomic address, neat and tidy.
Visualizing the Atomic Neighborhoods: The Orbital Diagram
Now, let's translate this into a visual orbital diagram. We represent each orbital as a box or a line, and each electron as an arrow. The direction of the arrow indicates the electron’s spin (one up, one down). We also need to remember Hund's rule, which states that within a subshell (like the three p orbitals), electrons will occupy each orbital singly before pairing up, and they will do so with parallel spins. Think of it as students preferring to sit in separate desks before having to share.
Here’s how it looks:

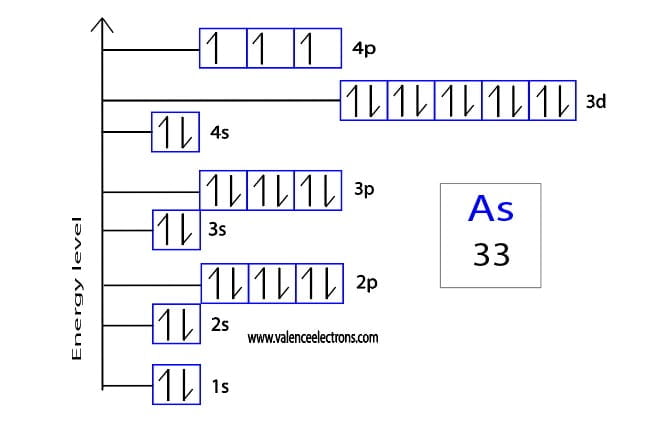
1s: [ ↑↓ ] (One box, two electrons, opposite spins)
2s: [ ↑↓ ] (One box, two electrons)
2p: [ ↑↓ ] [ ↑↓ ] [ ↑↓ ] (Three boxes, each filled with two electrons)
3s: [ ↑↓ ] (One box, two electrons)
3p: [ ↑↓ ] [ ↑↓ ] [ ↑↓ ] (Three boxes, filled)
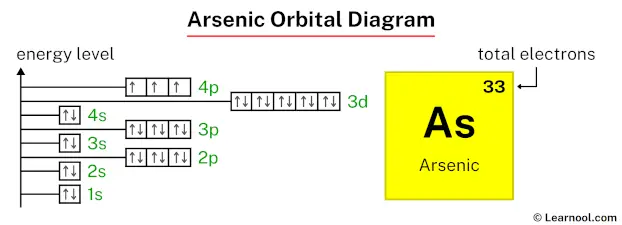
4s: [ ↑↓ ] (One box, two electrons)
3d: [ ↑↓ ] [ ↑↓ ] [ ↑↓ ] [ ↑↓ ] [ ↑↓ ] (Five boxes, all filled with two electrons)
4p: [ ↑ ] [ ↑ ] [ ↑ ] (Three boxes, each with one unpaired electron, all with parallel spins)
And there you have it! Arsenic's orbital diagram. It’s a snapshot of its electron arrangement, a visual representation of its atomic personality. Notice how the 4p subshell has those three unpaired electrons. This tells us a lot about arsenic's chemical behavior – it’s ready to bond and share those electrons to achieve a more stable configuration. It’s like that friend who’s always looking for a new adventure or a collaborative project.
Arsenic: More Than Just a Bad Reputation
It's easy to associate arsenic with its infamous reputation as a poison, thanks to historical tales and classic literature (Agatha Christie would have a field day!). However, arsenic is a metalloid, meaning it has properties of both metals and nonmetals. In its elemental form, it’s a brittle, gray solid. But it’s its compounds that are often where the action – and the danger – lies.

Interestingly, arsenic plays a vital role in some biological processes in trace amounts. For example, it's a component in some enzymes in certain organisms. It’s all about the dose, isn't it? Like caffeine – a morning cup can be invigorating, but a whole pot might lead to a very different experience. The orbital diagram helps us understand why arsenic behaves the way it does chemically. Those 4p electrons, being in the outermost shell and partially filled, are the ones involved in chemical reactions. They're the "hands" of the atom, reaching out to interact with others.
Did you know that arsenic compounds are used in some treatments for certain types of leukemia? Or that gallium arsenide (GaAs) is a crucial semiconductor material used in electronics like LEDs and solar cells? It's a testament to how elements we might fear can also be incredibly useful when understood and handled properly. It's the duality of nature, the yin and yang of the atomic world.
The Beauty of Atomic Organization
Constructing an orbital diagram might seem like a purely academic exercise, but it’s a beautiful illustration of the fundamental order that governs the universe. It’s a peek behind the curtain, showing us that even at the smallest scale, there are rules, patterns, and predictable behaviors. It’s the ultimate system for atomic organization, far more intricate and elegant than any filing system or organizational app we might use in our daily lives.
Think about it: the Aufbau principle, Hund's rule, the Pauli exclusion principle (which states no two electrons can have the same set of quantum numbers, hence the opposite spins in an orbital) – these are the laws of physics that dictate how every atom in existence is structured. From the air we breathe to the device you’re reading this on, it all boils down to these fundamental principles of electron placement.
A Reflection on Our Own "Orbitals"
As we wrap up our journey through arsenic's atomic landscape, let’s take a moment to connect this to our own lives. We, too, have our own "orbitals" – our daily routines, our social circles, our personal spaces. We tend to fill our time and energy in predictable ways, seeking out what's comfortable and stable, much like electrons filling lower energy levels. We have our favorite spots, our go-to activities, and our trusted companions.
Sometimes, though, just like arsenic's partially filled 4p orbitals are ready for interaction, we find ourselves needing to reach out, to connect, to form new bonds. Stepping outside our comfort zones, exploring new interests, or engaging with new people can be like filling those higher energy orbitals – it might feel a little uncertain at first, but it’s often where the most growth and exciting developments happen. Understanding arsenic's orbital diagram, its atomic structure, is a reminder that even the most complex systems are built on a foundation of order and predictable principles. And perhaps, in understanding this atomic order, we can find a little more clarity and intention in navigating our own human "electron configurations."