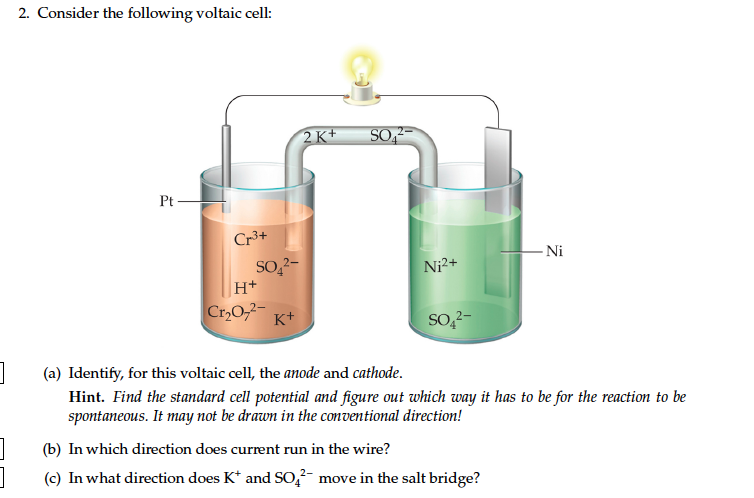
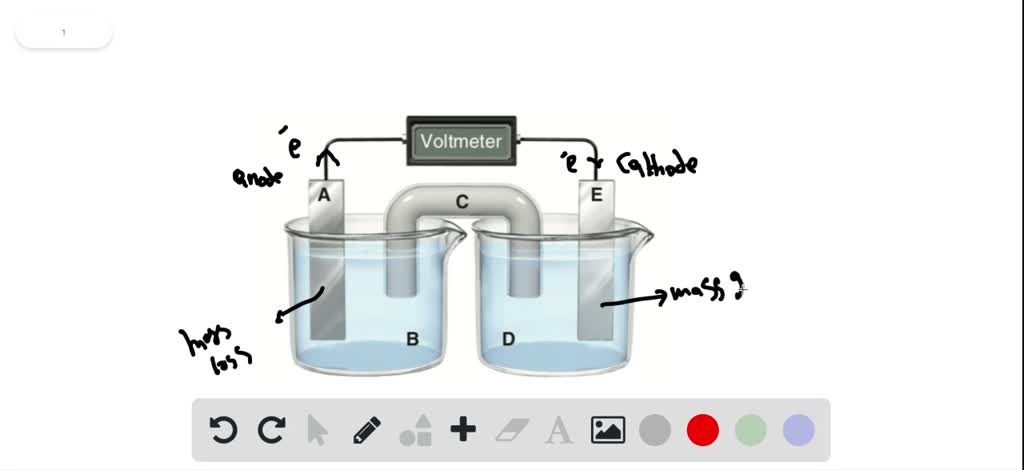
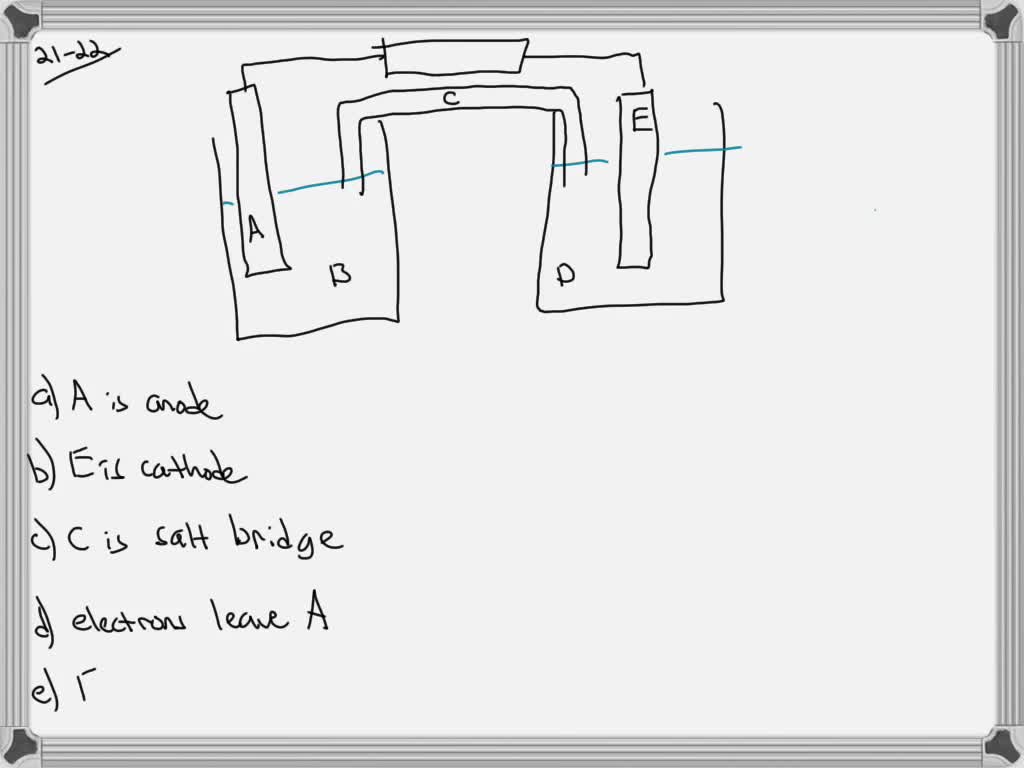
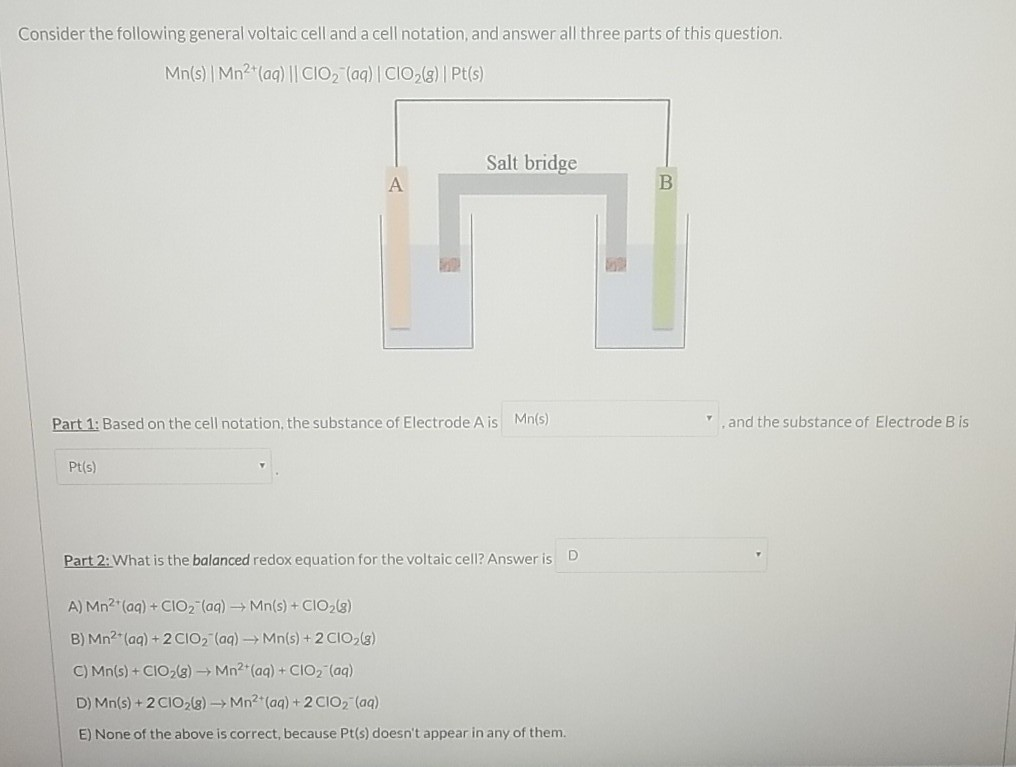
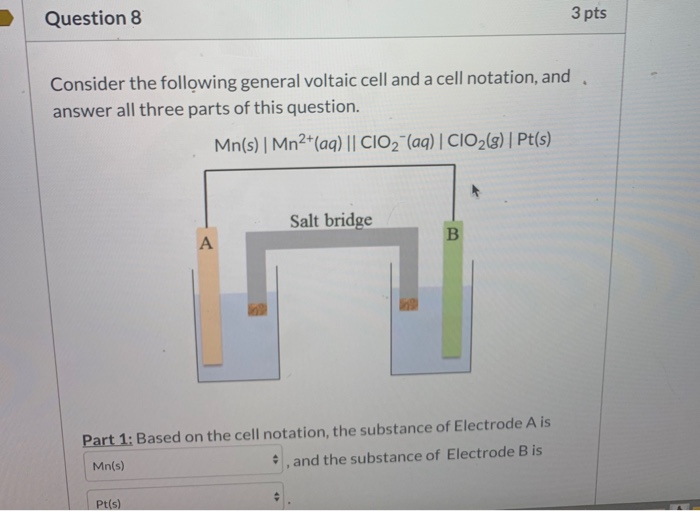
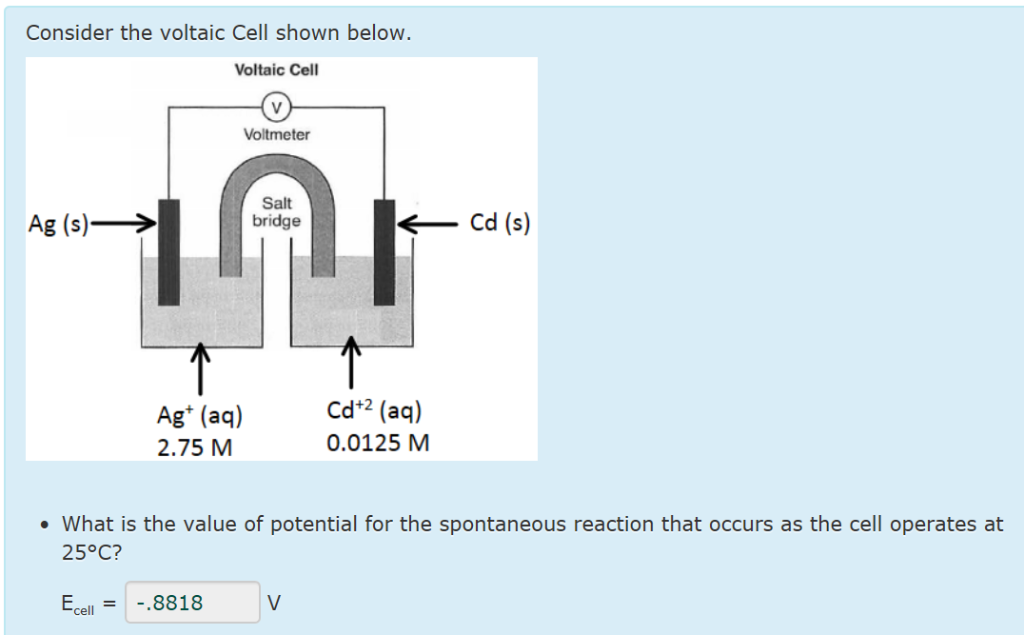
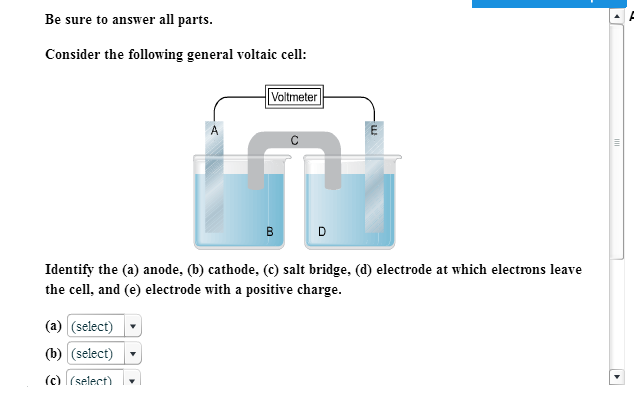
Consider The Following General Voltaic Cell
Ever found yourself wondering about those little sparks that power our gadgets or the humming batteries that keep our world running? Well, you're about to dive into the fascinating world of a general voltaic cell, and trust me, it's a lot more approachable and intriguing than it sounds! Think of it as the fundamental building block of electricity generation from chemical reactions – kind of like a mini, controlled chemical fireworks show that gives us power. Understanding this concept isn't just for scientists in lab coats; it's a peek behind the curtain of everyday technology and a surprisingly fun way to explore the magic of chemistry.
So, what's the big deal with a voltaic cell? Its primary purpose is to convert chemical energy into electrical energy. Imagine a chemical reaction that, instead of just producing heat or a gas, willingly gives up some of its energy in the form of electrons that can then travel through a wire. That's precisely what a voltaic cell does! The benefits are immense. Without voltaic cells, your smartphone would be a pretty paperweight, your car wouldn't start, and even the simplest LED flashlight wouldn't shine. They are the unsung heroes of portable power, offering a way to create electricity wherever and whenever we need it, without needing to be plugged into a wall.
You encounter voltaic cells constantly, even if you don't realize it. The most common example, of course, is a battery. Whether it's the AA batteries in your remote control, the lithium-ion battery in your laptop, or the car battery under the hood, these are all sophisticated versions of the general voltaic cell principle. In education, they're a cornerstone of chemistry and physics lessons, allowing students to build simple cells from common household items and witness the principles of electrochemistry firsthand. It’s a fantastic way to make abstract concepts tangible and exciting. Beyond that, think about galvanized steel – the zinc coating on it acts as a sacrificial anode, protecting the iron underneath from rusting through a similar electrochemical process.
Must Read
Ready to get a little hands-on and curious? Exploring voltaic cells doesn't require a fancy laboratory. A classic and simple way to see this in action is to create a lemon battery. All you need is a lemon, a copper coin (like a penny), and a galvanized nail (which is coated in zinc). Stick the coin and the nail into the lemon, making sure they don't touch inside. Then, you can use a sensitive voltmeter or even a very low-power LED to see if you're generating a tiny bit of electricity. It’s a testament to how even simple materials can hold the potential for power! Another easy exploration is to look at different types of batteries around your house and marvel at the variety of chemical combinations designed to power our modern lives.