Common Units Of Pressure Smallest To Largest
Hey there! Grab a coffee, get comfy. We're about to dive into something that sounds kinda fancy, but is actually way more everyday than you might think: pressure. Yeah, that thing that makes your ears pop on a plane, or when you squeeze a balloon. It's everywhere, right? And to talk about it, we've got these different units, like little measurement buddies. Ever wonder which ones are the teensy tiny guys and which ones are the big kahunas? Stick around, we're gonna break it down, from the microscopic to the, well, almost cosmic!
So, why even bother with all these different units? It’s like having a whole toolbox, you know? Sometimes you need a tiny screwdriver for a delicate screw, and other times you need a hefty wrench. Pressure is kinda the same. Depending on what you're measuring – the air in your bike tire, the water deep in the ocean, or even the weird stuff inside a star – you need the right tool. And these units? They’re our tools. It can get a little confusing, I know. Like, why do we need so many? But trust me, by the end of this, you’ll be a pressure unit pro. Almost. No promises, but we'll have fun trying!
The Little Guys: Getting Down and Dirty with Small Pressures
Let's start small. Like, really small. Imagine you’re talking about the pressure inside a really, really thin-walled container, or maybe some super-sensitive scientific experiment. You don't need a giant number, do you? That's where our pint-sized pressure pals come in.
Must Read
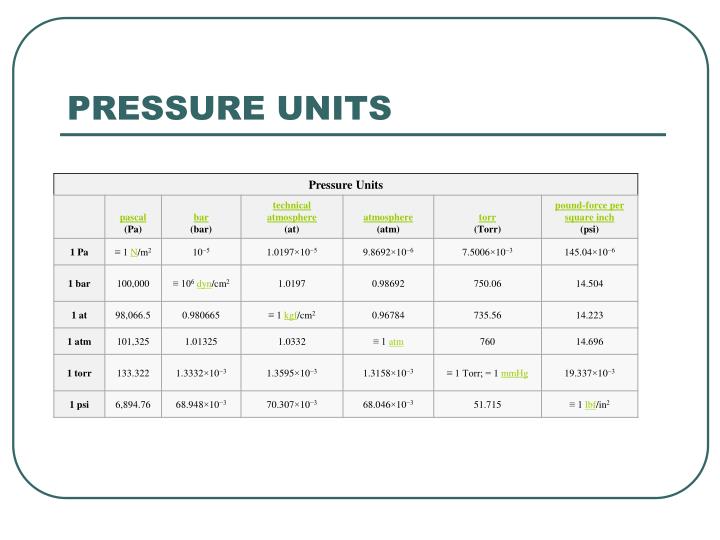
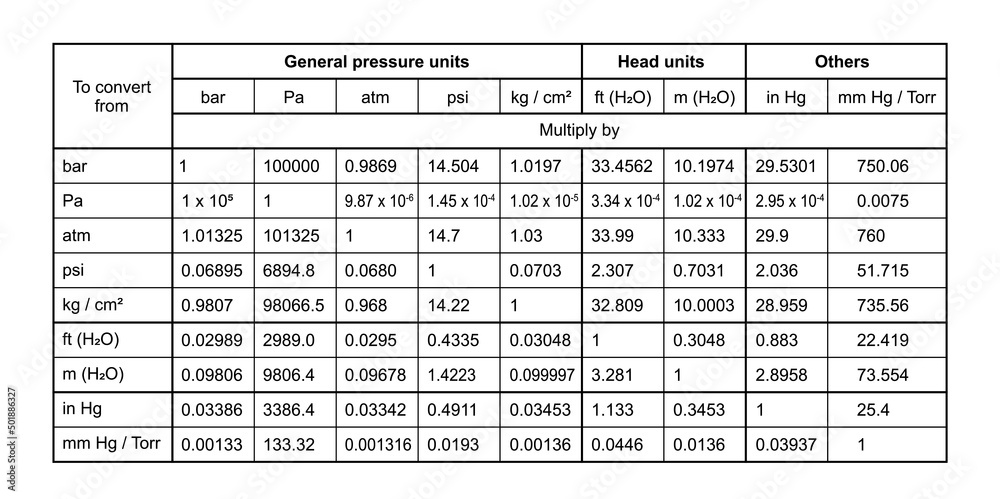
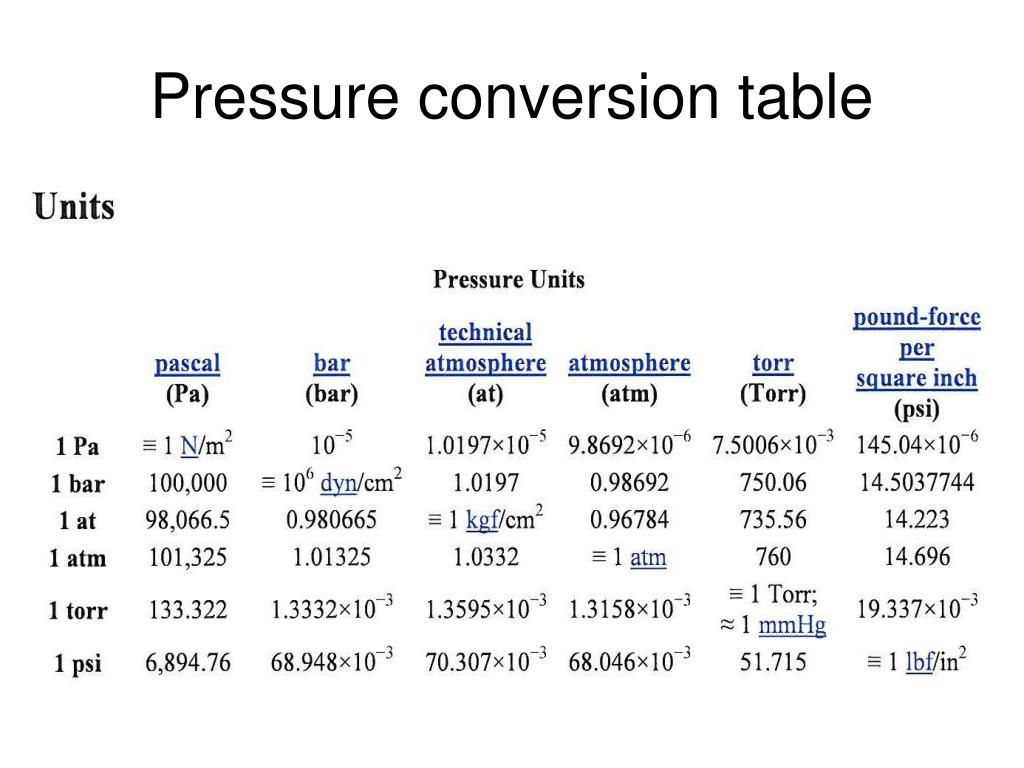
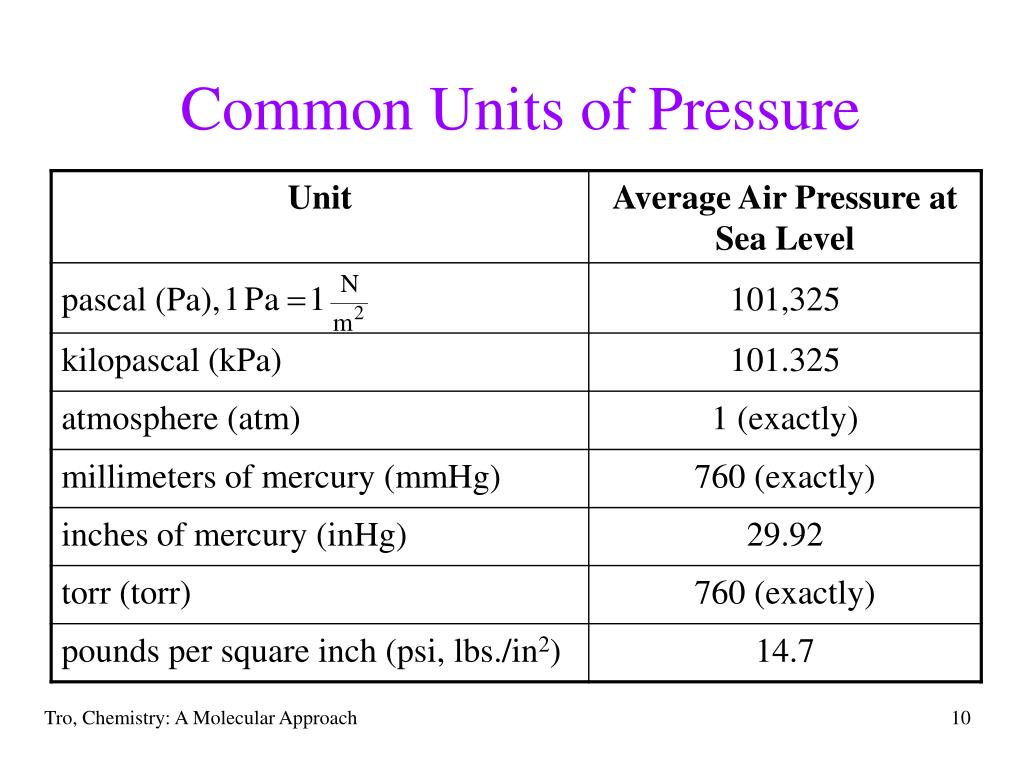
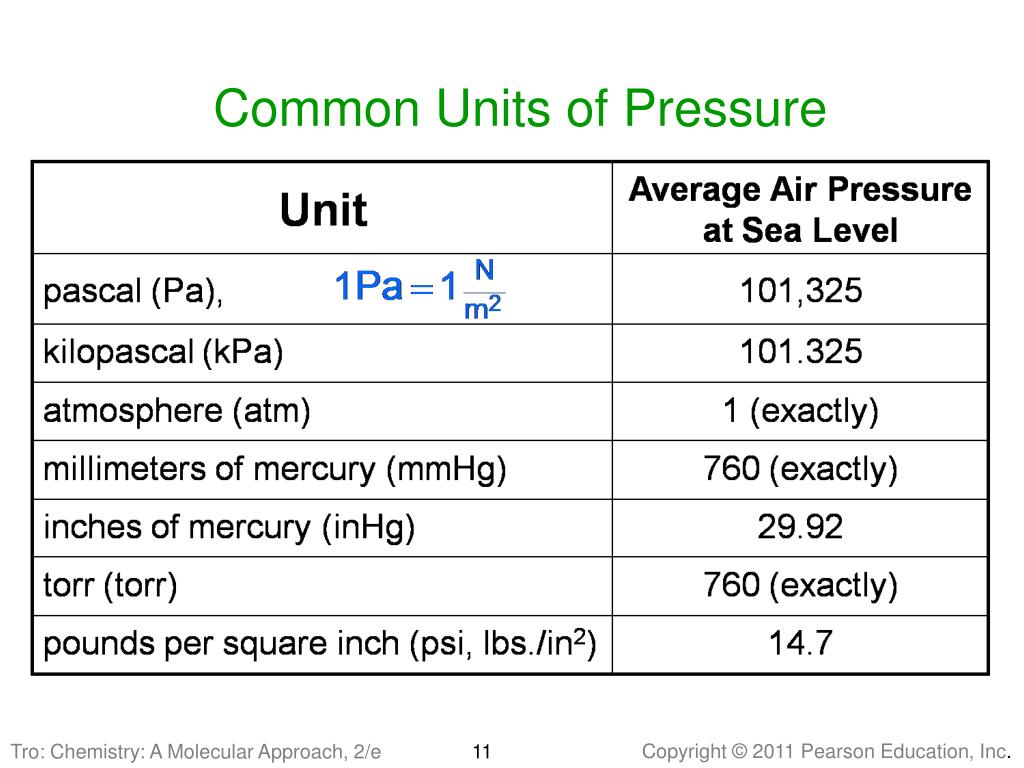
First up, we've got the Pascal (Pa). This is like the foundational unit, the meter of pressure, if you will. Named after a French guy, Blaise Pascal, who apparently liked his physics. One Pascal is, like, the pressure exerted by a tiny little weight – imagine a single sheet of paper sitting on your hand. Not exactly something you'd feel, right? It’s pretty darn small. So, in most everyday situations, you’ll see Pascals multiplied by a thousand, giving you kilopascals (kPa). That’s much more manageable. Like, the atmospheric pressure at sea level is roughly 101,325 Pascals, or about 101.3 kPa. See? That’s already getting a bit more bite to it.
Now, sometimes you’ll hear about Pounds per Square Inch (psi). This one’s more common in the US, where we’re still clinging to our imperial system like a toddler to a teddy bear. A psi is literally one pound of force spread over one square inch. So, if you have a tire that’s inflated to 30 psi, that means there are 30 pounds of pressure pushing outwards on every single square inch of the tire’s inner surface. Pretty neat, huh? It’s relatable, right? You can kinda picture a pound. A Pascal? Not so much. So, psi is often used for things you can feel, like tire pressure or the water pressure in your home plumbing. It’s a solid, mid-range unit.
What’s interesting is how these relate. A psi is actually way bigger than a Pascal. Like, 1 psi is about 6895 Pascals. So, if you have 10 psi in your bike tire, that's already over 68,000 Pascals! The Pascal is truly the minuscule marvel here. You’d need a lot of them to equal even a moderate psi.
Then there's the Torr. This one’s a bit quirky. It's named after Evangelista Torricelli, another Italian dude who invented the barometer. And guess what a Torr is basically equivalent to? The pressure exerted by a millimeter of mercury (mmHg) in a barometer. So, if the atmospheric pressure is 760 mmHg, that's 760 Torr. It’s often used in vacuum measurements and medical applications. Think about blood pressure readings – those are often given in mmHg, which is basically Torr. So, your doctor is essentially telling you your pressure in Torrs! Isn’t that a fun little fact to impress your friends with at parties? (Or, you know, just to know yourself.) It’s a unit that’s closely tied to a specific measurement device, which is kinda cool.
So, to recap our small fry: Pascal (tiny!), psi (relatable, mid-range), and Torr (good for vacuums and blood pressure). They all measure the same thing, but they give us different perspectives, depending on what we’re looking at. It’s all about scale, really. You wouldn’t use a microscope to look at a mountain, right?

Stepping Up: The Medium-Sized Wonders
Okay, so we’ve dipped our toes into the smaller end of the pressure spectrum. Now let’s crank it up a notch. These units are still pretty common, but they deal with a bit more oomph.
Remember how we said atmospheric pressure at sea level is around 101.3 kPa? Well, that's a pretty important pressure! And it’s so fundamental that it gets its own unit, kind of. It’s called the atmosphere (atm). One atmosphere is, by definition, the average atmospheric pressure at sea level. So, when you see "1 atm," you can think of it as a standard reference point for how much pressure the air around us is exerting. It’s like the "normal" setting on your pressure dial. It’s a great way to compare pressures to our everyday experience of the air we breathe.
Now, here’s where it gets a little confusing, but also kinda fun. That mmHg we talked about with the Torr? It’s also a very common unit for measuring atmospheric pressure. So, at sea level, it’s roughly 760 mmHg. And since 1 atm is the standard sea-level pressure, that means 1 atm is equal to 760 mmHg (or 760 Torr). See how they all link up? It’s like a secret code they’re all speaking! So, if you see a weather report saying the pressure is 1020 millibars (we'll get to that!), you can convert it to atmospheres or mmHg if you need to. It’s all about having the right conversion factors. Maths to the rescue!
And speaking of millibars, here’s another one: the millibar (mbar). This unit is super popular in meteorology, you know, for weather forecasts. Why? Because 100 millibars is pretty close to 1 atmosphere. So, standard sea-level pressure is about 1013.25 millibars. This makes it really easy for meteorologists to work with. Instead of saying "the pressure is 101,325 Pascals," they can just say "the pressure is 1013 millibars." Much cleaner, right? It's just a convenient way to express those atmospheric pressures without getting lost in a string of zeros.
So, our medium guys are the atmosphere (atm), which is our handy "normal" air pressure, and the millibar (mbar), which is weather folks’ best friend. And remember, they’re all interconnected with mmHg and Torr. It's like a pressure family reunion where everyone knows each other!

The Big Guns: When Pressure Gets Serious
Alright, we’re moving on up! These next few units are for when things get really pressurized. We’re talking about industrial applications, deep sea diving, or even what’s happening inside a rocket engine. Things get intense!
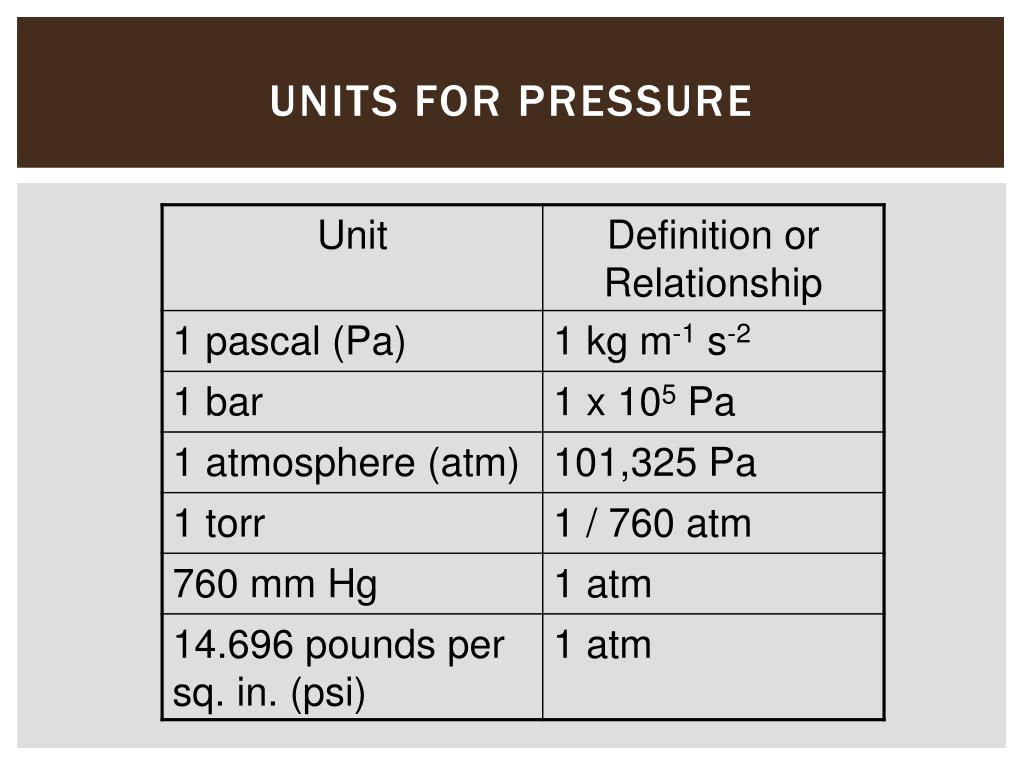
Let’s revisit our friend, the bar. Remember the millibar (mbar)? Well, a bar is simply 1000 millibars. So, if 1 atm is about 1013 mbar, then 1 bar is just a smidge less than 1 atm. It's another unit commonly used in industry and meteorology because it's closer to atmospheric pressure than the Pascal, but still a nice round number for many applications. You might see it used in things like tire pressure gauges for trucks or in hydraulic systems. It’s got a good solid feel to it, you know? Not too tiny, not too huge. Just right for many practical uses.
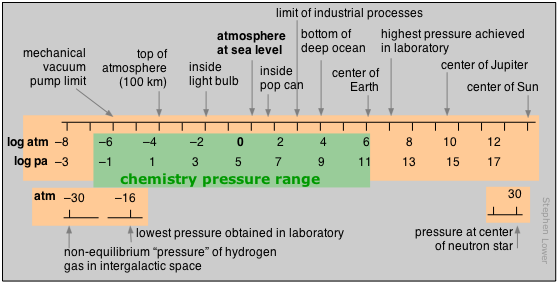
Now, what happens when you go really, really deep underwater? The pressure builds up FAST. That’s where you might hear about kilobars (kbar). One kilobar is 1000 bars. So, that's a lot of pressure. The pressure at the bottom of the Mariana Trench, the deepest part of the ocean? We're talking about pressures in the hundreds of kilobars! Imagine the force. It’s enough to crush pretty much anything that isn't designed to withstand it. So, kbar is for when you're dealing with some seriously extreme conditions. Think geological pressures, deep ocean exploration, that kind of stuff.
And then, for the really, really serious stuff, like what’s happening inside a star or during a supernova explosion, we get into units that are almost too big to comprehend for our everyday brains. While not always a single standardized unit that everyone uses for everything, you might encounter terms related to densities and forces that, when translated to pressure, are astronomical. For instance, the pressure at the core of the Sun is estimated to be in the hundreds of billions of atmospheres. That’s… a lot of atmospheres. You're talking about pressures so immense that matter behaves in ways we can barely imagine.
So, our big kahunas are the bar, which is a nice, solid industrial unit, and the kilobar (kbar), for when you need to talk about crushing, deep-sea pressures. And then there are those mind-boggling pressures found in space, where the numbers just get ludicrously large. It’s a good reminder that our little planet has a pretty gentle pressure system compared to the universe out there!
Putting It All Together: The (Sort of) Orderly Chaos
Okay, so let's try to put these pressure pals in order, from smallest to largest. Remember, these are approximate relationships, and sometimes the context matters more than the strict number. It's like trying to order grapes by size – some are a bit fatter, some a bit thinner, but they're all grapes!
We're going to start with the absolute tiddlers and work our way up. Think of it as a pressure parade!
1. Pascal (Pa): The undisputed champion of tiny. The unit you'll rarely see on its own without a prefix. It's the microscopic whisper of pressure.
2. mmHg (or Torr): A bit bigger than a Pascal, especially when you're talking about atmospheric pressure. Still relatively small, but useful for specific measurements like blood pressure or vacuums. It’s the gentle puff.
3. psi (Pounds per Square Inch): This is where we start to get into more relatable territory. You can feel psi in your tires. It's a decent chunk of pressure, much bigger than a Pascal.
4. Millibar (mbar): This is where weather maps live. It’s a convenient way to express atmospheric pressure without huge numbers. It's a comfortable, everyday pressure for air.
5. Atmosphere (atm): The standard for our everyday air pressure at sea level. A good reference point. It’s like the "medium" setting on your pressure cooker.
6. Bar: A solid industrial unit, just a bit bigger than an atmosphere. Often used for things that require a bit more robustness than typical air pressure.
7. Kilobar (kbar): Now we're talking serious pressure. Deep ocean, geological forces – this is where you'll find kilobars. It's the squeeze that can reshape mountains (over time, of course).
8. Astronomical Pressures: Beyond kilobars, you enter realms where the numbers are so immense they’re hard to grasp. Think nuclear reactions, the cores of stars. These are pressures that defy our everyday experience.
It's important to remember that the "smallest to largest" is based on the magnitude of the unit itself. One Pascal is a tiny amount of pressure. One kilobar is a massive amount. However, you can express the same amount of pressure using different units. For example, 1 atm is roughly 101,325 Pa. So, a "small" unit like the Pascal can be used to represent a large total pressure, just with a big number. It's all about perspective and convenience!
Why so many? Well, different fields just found different units more practical. Imagine if all your recipes were in grams, but your doctor measured your blood pressure in grams too. It would get weird! Each unit has its niche, its sweet spot where it makes the most sense. And knowing them, even just a little, helps us understand the world around us a bit better. From the air in our lungs to the forces that shape planets, pressure is a fundamental concept, and its units are our tools for measuring it. So, next time your ears pop, you'll have a slightly better idea of what's going on. Pretty cool, right?