Clay Like Substance From Which Aluminium Is Obtained

Okay, so let's talk about something you probably never thought you'd find exciting: dirt. Yeah, you heard me. Not just any dirt, though. We're diving into the magical, sort of gloopy stuff that gives us the shiny, lightweight metal that's in everything from your soda can to your airplane. Pretty cool, right?
This isn't your average garden variety soil. Nope. This is the source material for aluminum. Think of it as the ultimate treasure buried deep within the Earth. It’s like nature’s secret recipe for making things fly and fizz.
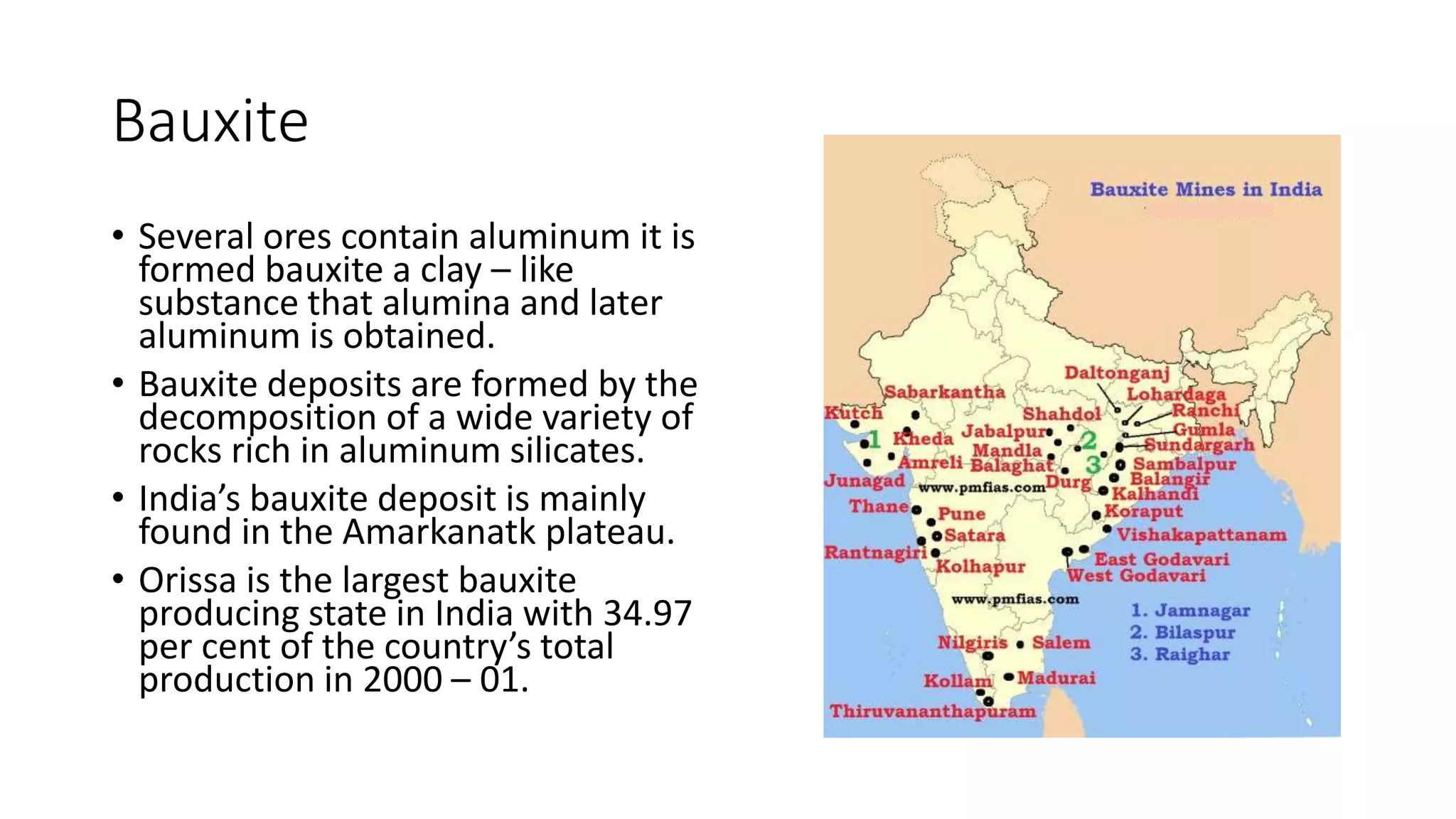
The star of our show is a rock, a mineral, really, called bauxite. Say it with me: baux-ite. It sounds a bit fancy, doesn't it? Like something you'd find in a posh French bakery. But this stuff is seriously tough stuff.
Must Read
Digging Up the Dream Metal
So, how do we get this aluminum goodness out of bauxite? Well, it’s not exactly like scooping it out with a shovel and a sieve. It’s a bit more… intense. Think of it as a very, very patient extraction process. Nature took its sweet time creating bauxite, and it takes us a good chunk of effort to unlock its secrets.
Bauxite isn’t usually found looking like a neat little brick. It’s often mixed in with other earthy bits and pieces. So, the first step is a big ol’ mining operation. They dig it up, usually from open pits. Imagine vast landscapes being carefully excavated, all in the name of finding this precious stuff.
But here’s a fun fact: bauxite itself isn't pure aluminum. Not even close. It’s more like a chunky stew of aluminum compounds, iron oxides, and silica. So, it looks more like reddish-brown clay or gravel than a shiny metal. Totally unassuming, right? Yet, it holds the promise of lightness and strength.
The Alchemical Transformation
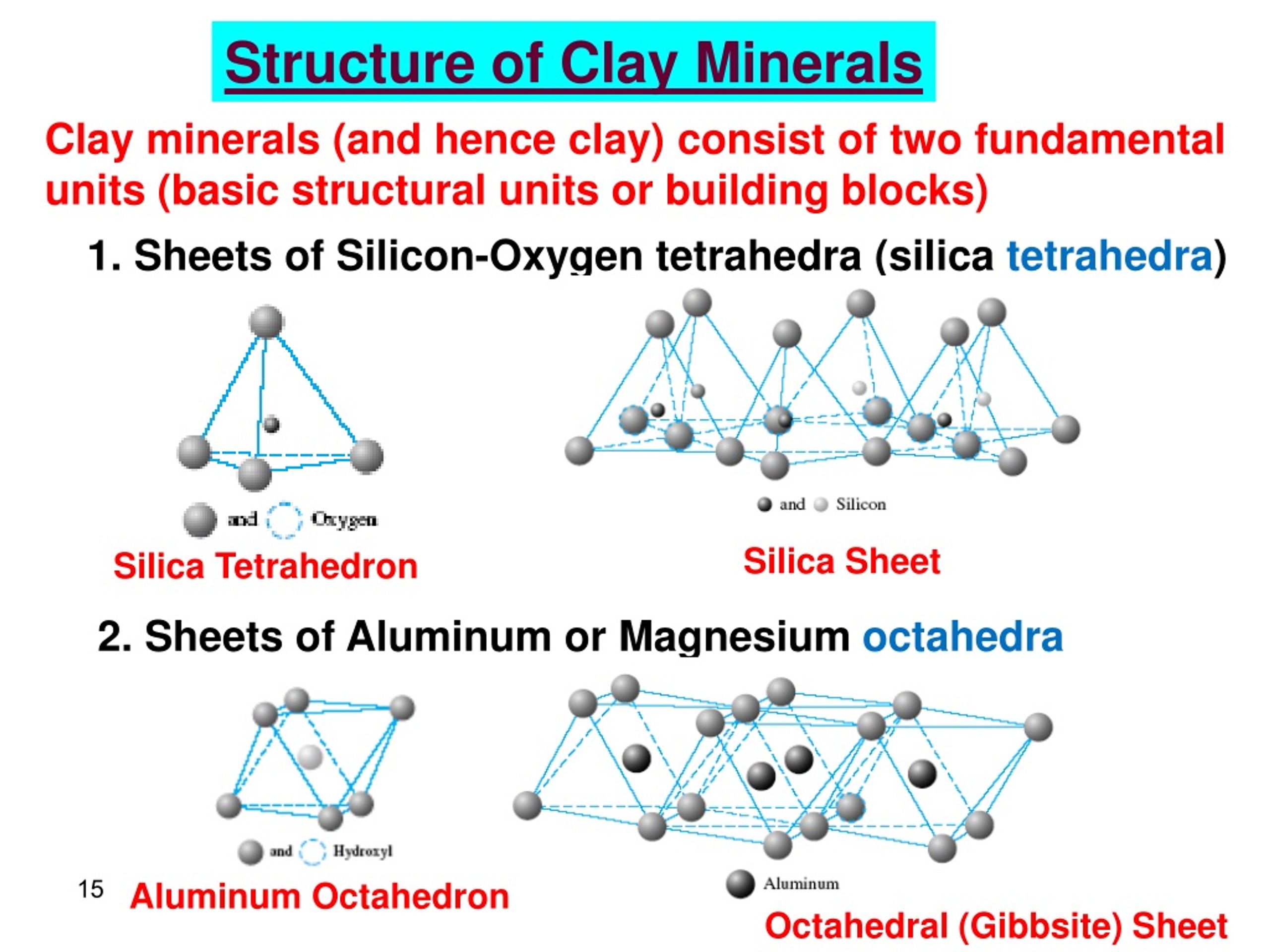
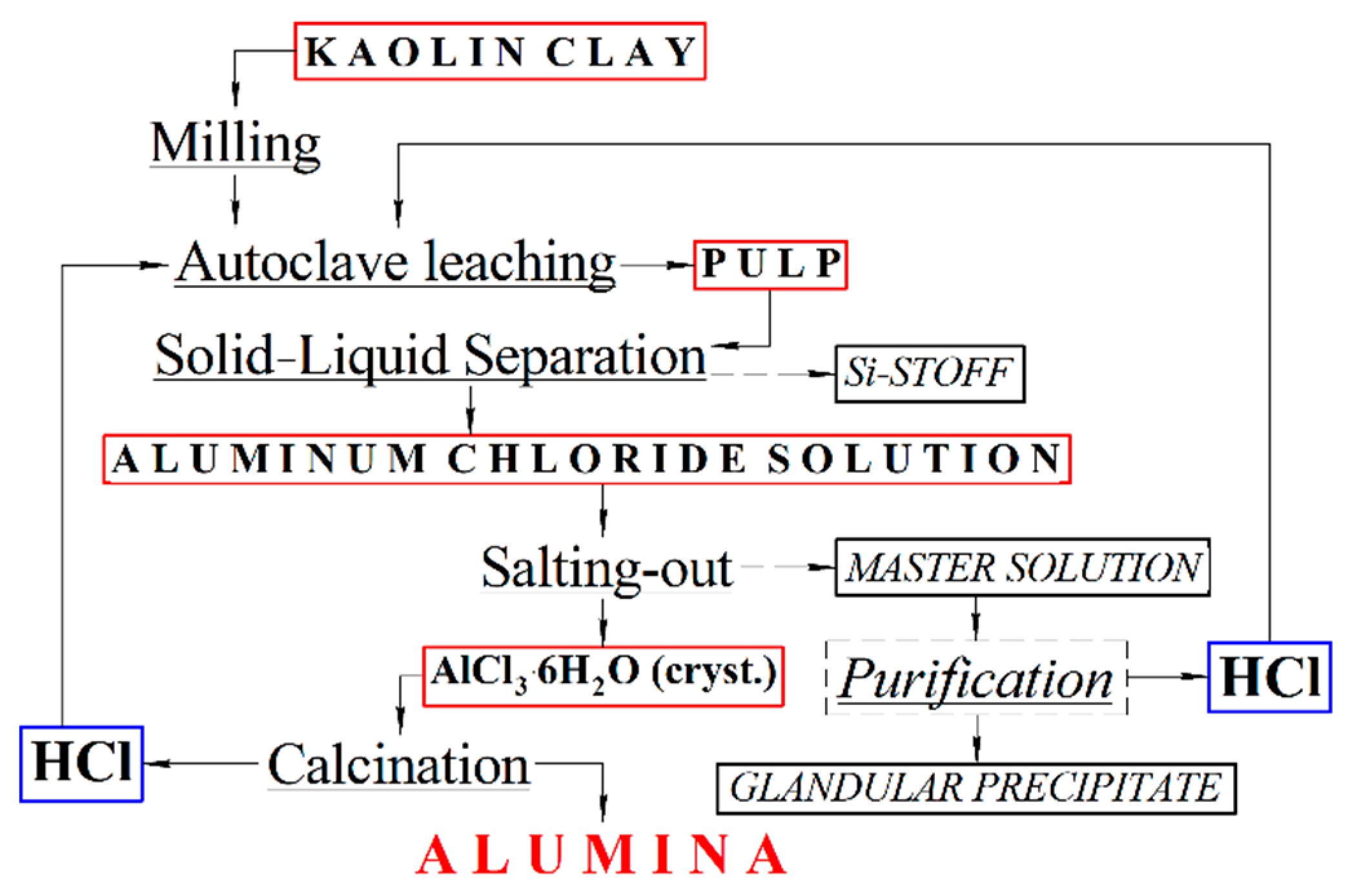
Once they’ve got the bauxite, the real magic begins. It’s not quite like turning lead into gold, but it's pretty darn close in terms of engineering marvel. They use a process called the Bayer process. Fancy name, right? It’s basically a super-powered washing and cooking routine for the bauxite.
First, they grind the bauxite into a fine powder. Then, they heat it up with a strong chemical called caustic soda (sodium hydroxide, for the science nerds). This hot, bubbly bath dissolves the aluminum compounds, leaving behind all the yucky stuff like iron and silica. Think of it like making a super concentrated tea, but instead of tea leaves, it’s bauxite, and instead of water, it’s caustic soda.
After this steamy spa treatment, they filter out the solids. What’s left is a liquid that’s packed with dissolved aluminum. They then cool this liquid down, and something amazing happens: pure aluminum hydroxide crystals start to form. It’s like the aluminum saying, “Okay, I’m ready to be myself now!”
These crystals are then heated up even more, to a scorching temperature. This drives off any remaining water, leaving behind a white powder. This powder is called alumina. And this alumina is what we eventually turn into aluminum.
From Powder to Powerhouse Metal
Now, alumina is almost aluminum, but not quite. It’s still in a chemical compound form. To get the pure, silvery metal, we need another incredible step: the Hall-Héroult process. This is where things get really electrifying, literally.
Imagine a giant bathtub, but instead of water, it’s filled with molten cryolite. Yes, cryolite! It’s another mineral, and it’s crucial because it lowers the melting point of alumina, making the whole process possible at a slightly less supernova temperature. Inside this bath, they dunk huge carbon electrodes.
Then, they pour the alumina into this super-hot, bubbly cryolite mixture. And here’s the mind-blowing part: a massive electric current is passed through the electrodes and the liquid. This electricity literally rips the oxygen away from the aluminum in the alumina. It’s like a powerful electrical karate chop!
The aluminum, now freed from its rocky prison, is heavier and sinks to the bottom of the pot. They can then tap it off, pure and molten. It’s a dazzling, fiery spectacle. Imagine a blacksmith's forge, but on an industrial, huge scale.
Quirky Bauxite Bits
Let’s throw in some more funnies. Did you know that bauxite is found in a lot of tropical and subtropical regions? Think Australia, Brazil, Guinea. It’s like the sun-loving cousin of aluminum.

And the color? It can range from white and gray to reddish-brown and even pink! So, the source of our sleek metal can sometimes look like a giant patch of colorful mud. Nature’s got a sense of humor, for sure.
Another cool thing: the name “bauxite” comes from a small village in southern France called Les Baux-de-Provence. So, the rock is literally named after a place! It’s like if your favorite snack was named after your hometown. “Pass the hometown crisps, please!”
And get this: it’s not just one mineral. Bauxite is actually a mixture of aluminum hydroxide minerals. So, it’s a whole team effort in that rocky stew to create our favorite lightweight metal.
Why Should You Care About Mud?
Okay, so why is this dusty, muddy origin story so darn interesting? Because it highlights the incredible ingenuity of humans and the amazing resources our planet provides.
Aluminum is everywhere! It’s in your car, making it lighter and more fuel-efficient. It’s in your bike, making it speedy and nimble. It's in your window frames, keeping the weather out. It's in your foil, keeping your leftovers fresh. It's even in your toothpaste!
And the best part? Aluminum is infinitely recyclable. Unlike some other materials that lose their quality with each recycling cycle, aluminum can be melted down and reformed into new products over and over again without losing its strength. That soda can you just finished? It could be back on the shelf as a new can in as little as 60 days!
So, next time you see a shiny aluminum object, remember its humble beginnings. Remember the reddish-brown clay, the intense chemical baths, and the electrifying pots. It’s a journey from the ground to your hands, a testament to what we can achieve when we understand and harness the power of nature.
It’s a story of transformation, from something we might overlook as just dirt, to a metal that shapes our modern world. Pretty wild, huh? It makes you look at a simple soda can a little bit differently, doesn't it? It's not just a drink container; it's a little piece of geological history and human innovation, all rolled into one.


