Classify Each Solvent As Protic Or Aprotic
Alright, pull up a chair, grab a latte, and let's talk solvents. Now, I know what you're thinking, "Solvents? Is this a chemistry lecture disguised as a caffeine fix?" And to that, I say, "Pretty much, but way more fun, and with fewer lab coats." Think of it like this: you're at a party, and everyone's got a personality. Solvents are no different. They can be the life of the party, or the wallflower who secretly knows all the gossip. And today, we're going to learn how to tell them apart based on their social skills – specifically, whether they're protic or aprotic.
So, what's the big deal? Why do we care if a solvent is protic or aprotic? Well, imagine you're trying to get a shy introverted molecule to mingle with a super outgoing one. The right solvent is like the perfect wingman. It can either help them break the ice (protic) or create a super chill, non-judgmental vibe where they can just be themselves (aprotic). Get it wrong, and you might have a molecular meltdown, or worse, a reaction that goes absolutely nowhere. It's the difference between a sparkling social event and a silent elevator ride.
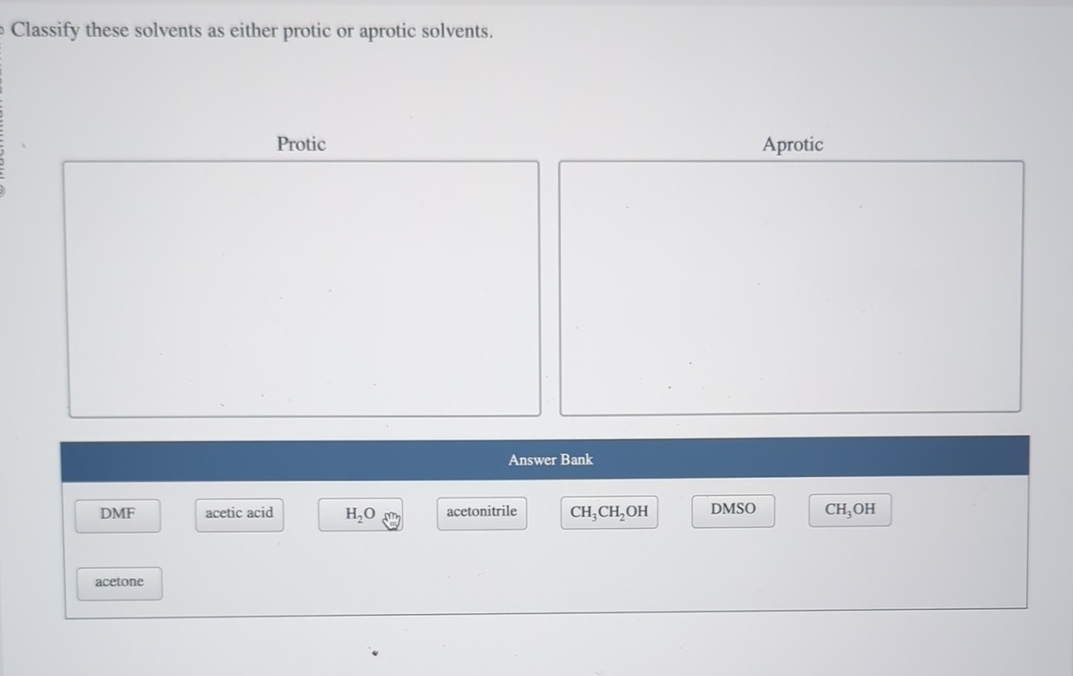
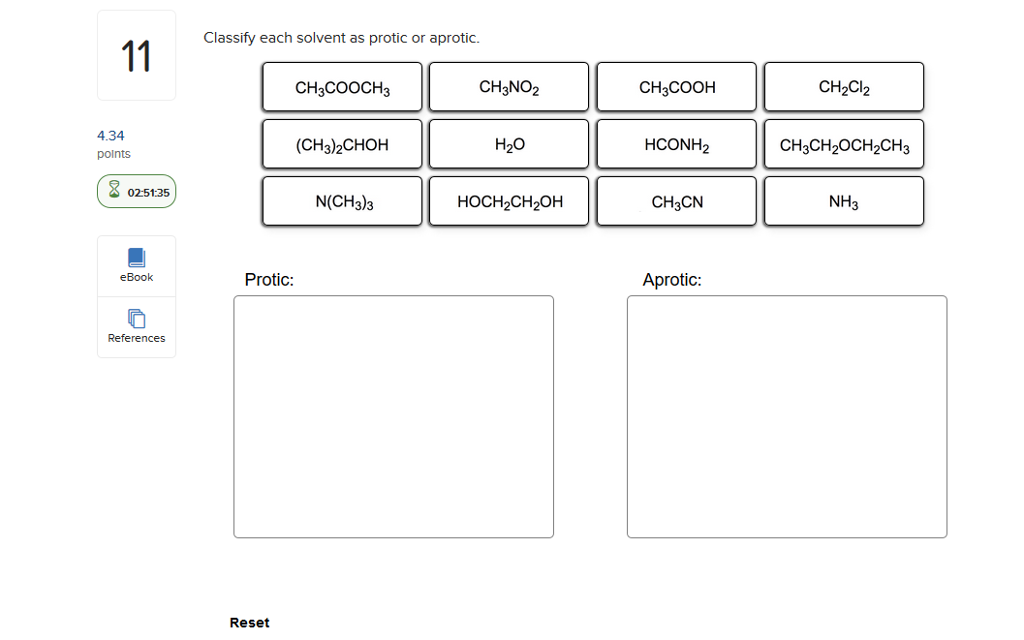
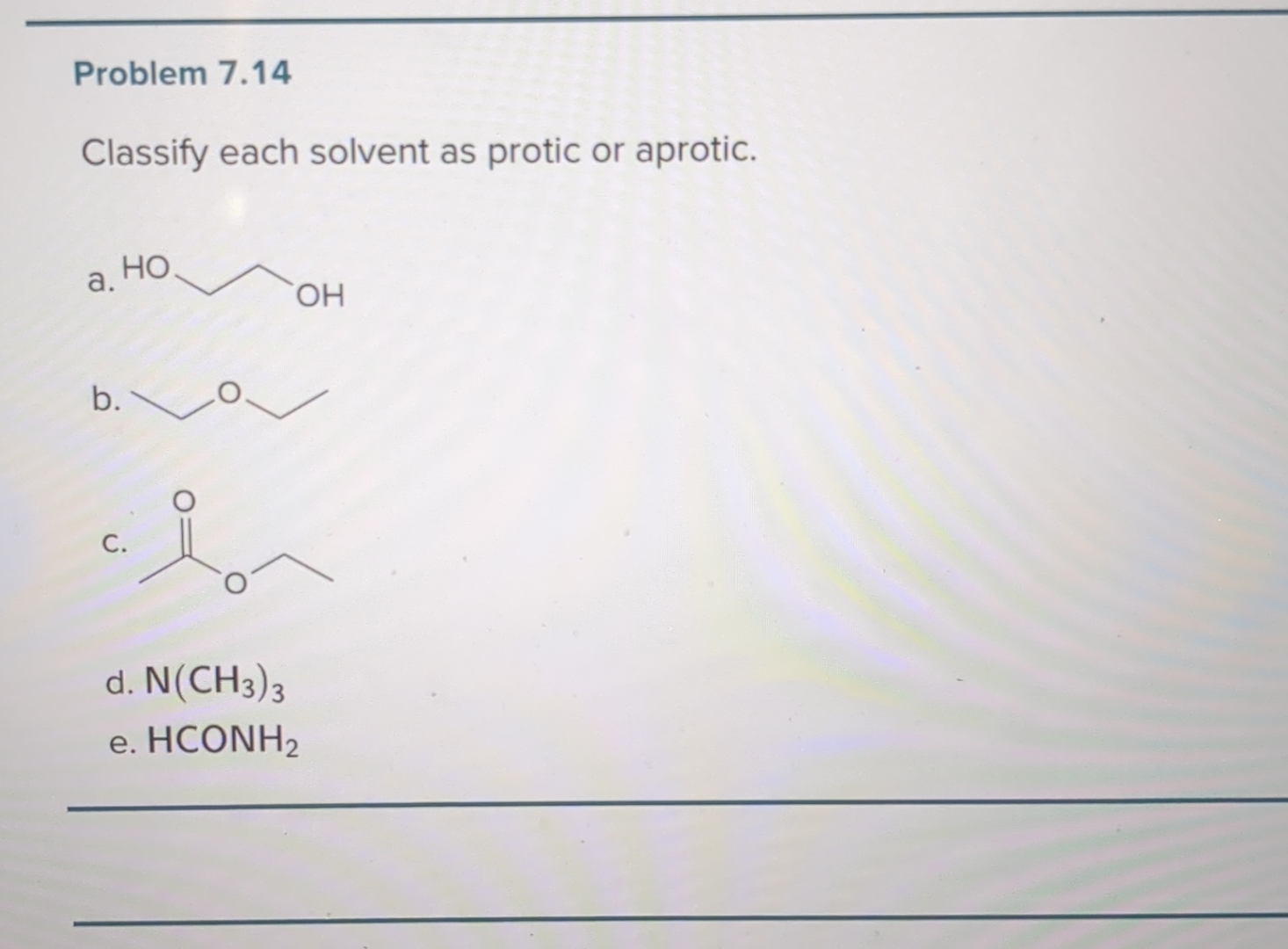
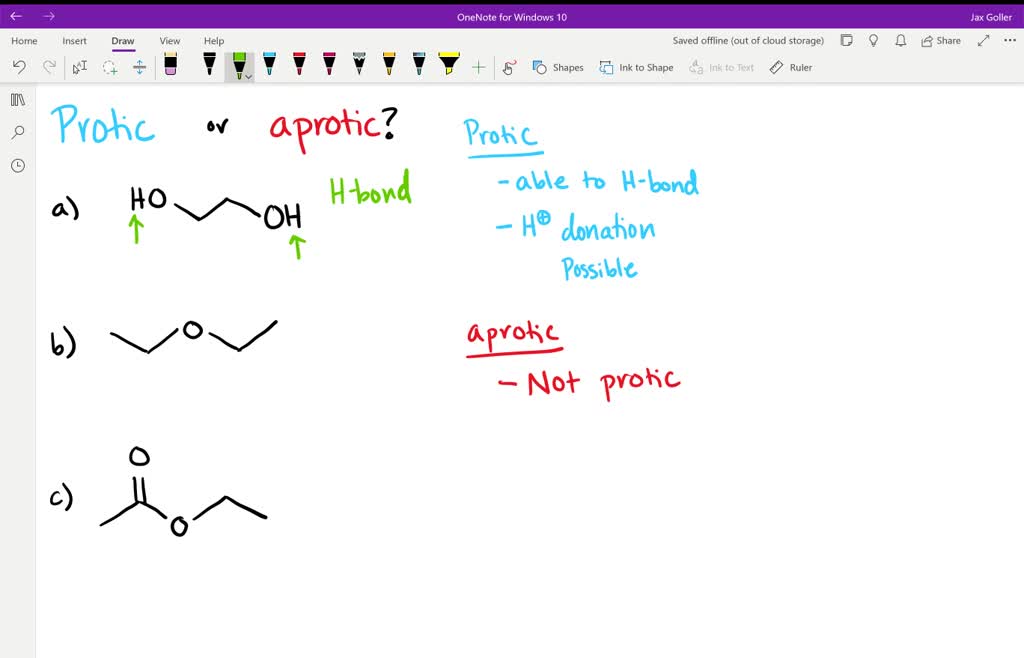
Let's start with the protic solvents. These are your social butterflies, the ones who aren't afraid to get involved. Their defining feature, the absolute giveaway, is that they have a hydrogen atom attached to an oxygen or nitrogen atom. Think of it as them having a little hydrogen friend they're always holding hands with, ready to share. This "hand-holding" hydrogen is quite acidic, meaning it's eager to jump ship and bond with something else. It's like they're constantly saying, "Hey, need a proton? I've got one to spare!"
Must Read
The most famous protic solvent, the Beyoncé of the bunch, is water. Yep, H₂O. That humble liquid we drink, bathe in, and occasionally use to put out fires? That's a protic powerhouse. It's got two hydrogen atoms literally begging to be loaned out. This makes water fantastic at dissolving ionic compounds, like salts. It's like water gives each ion a little H-hug, saying, "Don't worry, I'll keep you company!" It's also why water is such a great solvent for life itself. Imagine your DNA trying to exist without water constantly helping things out. It'd be like trying to throw a rave in a vacuum.
Then you have the alcohols, like methanol (CH₃OH) and ethanol (CH₃CH₂OH). These guys are basically water with a little fancy hydrocarbon skirt. They’ve got that all-important -OH group, which means they're also protic. Ethanol, of course, is the reason we have happy parties and questionable decisions on a Friday night. It’s a protic solvent with a built-in party animal. Who knew chemistry could be so fun?
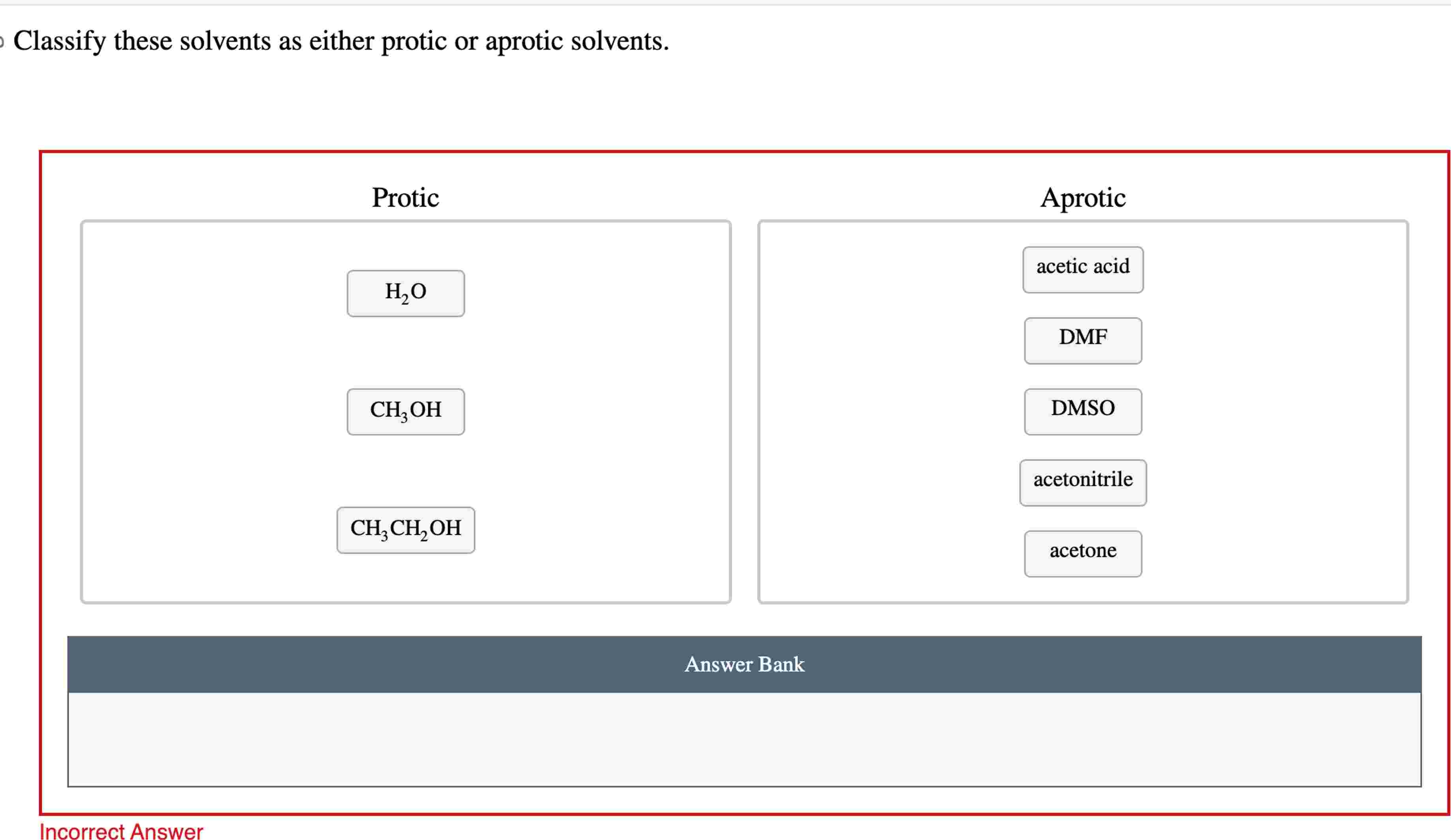
Other protic contenders include acetic acid (CH₃COOH) – that's vinegar, folks! – and even ammonia (NH₃), though its hydrogen-sharing is a bit less enthusiastic than water or alcohols. Acetic acid is like the slightly bossy older sibling, always ready to lend a proton and tell you what to do. Ammonia is more like the quiet cousin who’s surprisingly good at sharing when you really need it.
Now, let's switch gears and talk about the aprotic solvents. These are your cool, detached types. They're not about sharing their hydrogen friends. In fact, they have no hydrogen atoms directly bonded to oxygen or nitrogen. They’re like, "Hydrogen? What hydrogen? I'm all about myself, and I’m perfectly happy being solitary." This doesn't mean they're unhelpful, oh no. They're just helpful in a different way. They're like the chill roommate who lets everyone do their thing without getting involved.

The most famous aprotic solvent, the James Bond of the solvent world, is probably dimethyl sulfoxide (DMSO). This stuff is amazing. It’s like a super-solvent that can dissolve things other solvents just shrug at. But here’s the kicker: DMSO has a sulfur atom with a double bond to oxygen and then two methyl groups. No acidic hydrogen to offer. It’s all about its other electron-rich parts. It’s the solvent that says, "I’m not here to judge your reactants, I’m just here to provide a comfortable space for them to interact."
Another common aprotic friend is acetonitrile (CH₃CN). This one's a bit of a workhorse. It's polar, meaning it has some charge separation, which helps it dissolve things, but it’s not going to donate any protons. It’s like the efficient organizer of the solvent party – keeps things tidy without getting emotionally invested.
And then there's tetrahydrofuran (THF). This one's a cyclic ether, a bit like a tiny donut of atoms. It’s also aprotic and a great solvent for a lot of organic reactions. It’s like the cozy couch at the party – comfortable and accommodating, but not actively participating in any dramatic conversations.
Why is this whole protic/aprotic thing so important, you ask? Well, it drastically affects how reactions happen. Protic solvents, with their easily given-up protons, are great for reactions that involve dissociation – breaking apart. They can stabilize ions that form when bonds break. Think of it like a friendly mediator helping two sides separate peacefully. They're particularly good for dissolving ionic compounds and stabilizing charged intermediates.
Aprotic solvents, on the other hand, are fantastic for reactions where you want to keep your reactants nice and intact, but still dissolved and able to bump into each other. They’re often used in reactions involving strong bases or nucleophiles, where you don't want the solvent getting in the way and reacting itself. It's like throwing a silent disco for molecules – they can groove and interact without any unsolicited vocalizations from the surrounding environment.
Consider the classic SN1 vs. SN2 reactions. Protic solvents tend to favor SN1, where a carbocation intermediate is formed, and the solvent can help stabilize it. Aprotic solvents, especially polar aprotic ones like DMSO or DMF (dimethylformamide), often favor SN2, where the nucleophile attacks directly and there's no stable intermediate to worry about. It's the difference between a slow, deliberate process with lots of hand-holding and a quick, direct pounce.
So, how do you spot them in the wild? Look for the tell-tale O-H or N-H bonds. If you see that, you’ve got a protic friend. No such luck? Then you’re probably dealing with an aprotic type. It’s like a quick personality test for your chemical companions.
In conclusion, solvents, like people, have distinct personalities that influence how they interact with the world (and our chemical reactions). Understanding whether a solvent is protic (generous with its protons, like water and alcohols) or aprotic (self-contained, like DMSO and acetonitrile) is your key to predicting and controlling chemical behavior. It's not just about dissolving things; it's about setting the stage, managing the mood, and ensuring the right molecules have the right kind of party. So next time you’re dealing with a chemical reaction, remember: choose your solvent wisely. They might just be the most important guests at your molecular soirée!