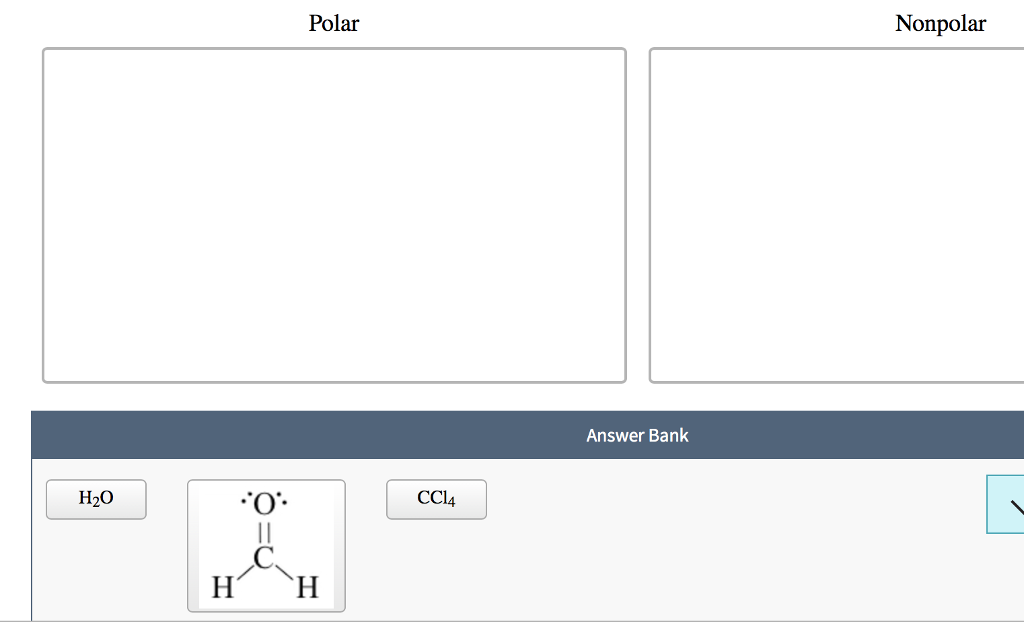
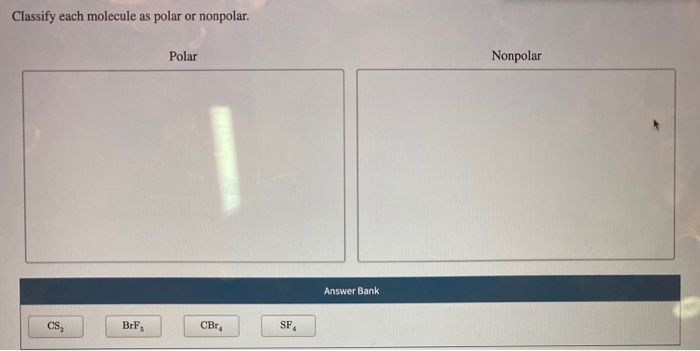
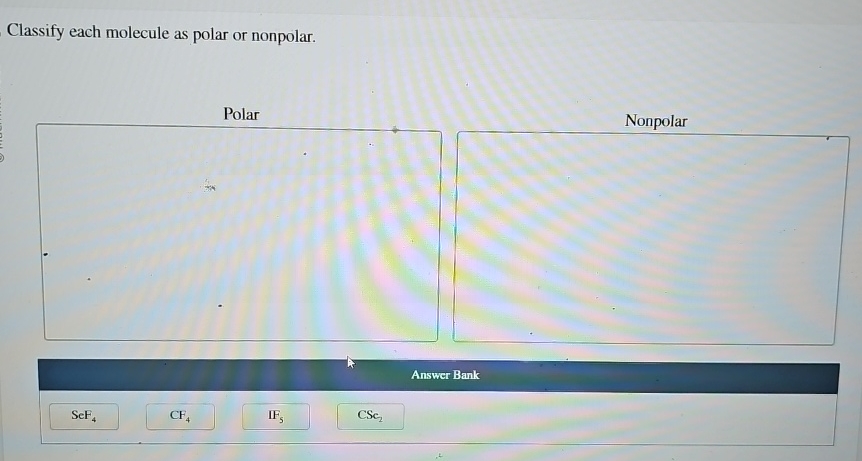
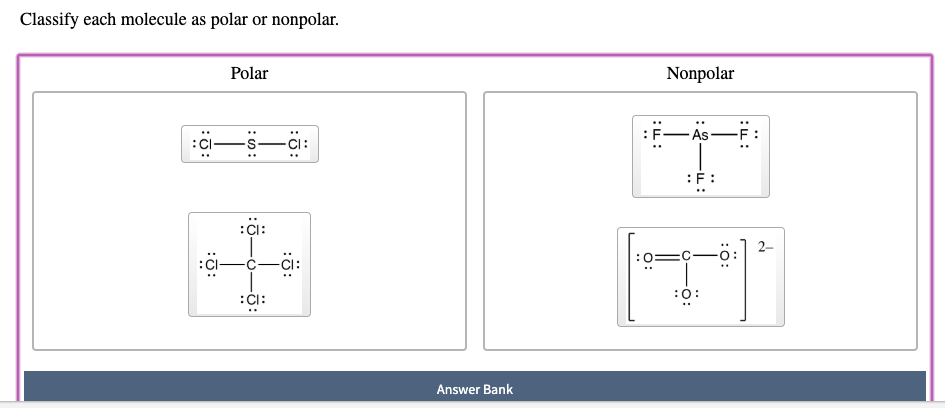
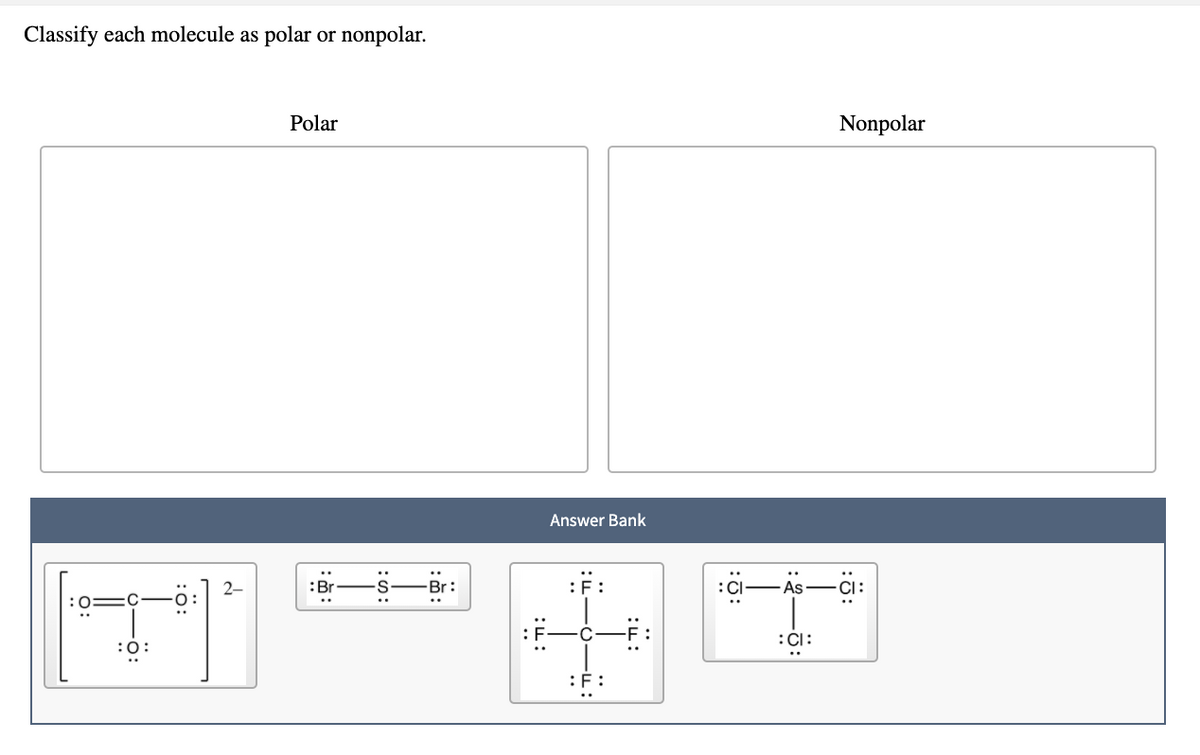
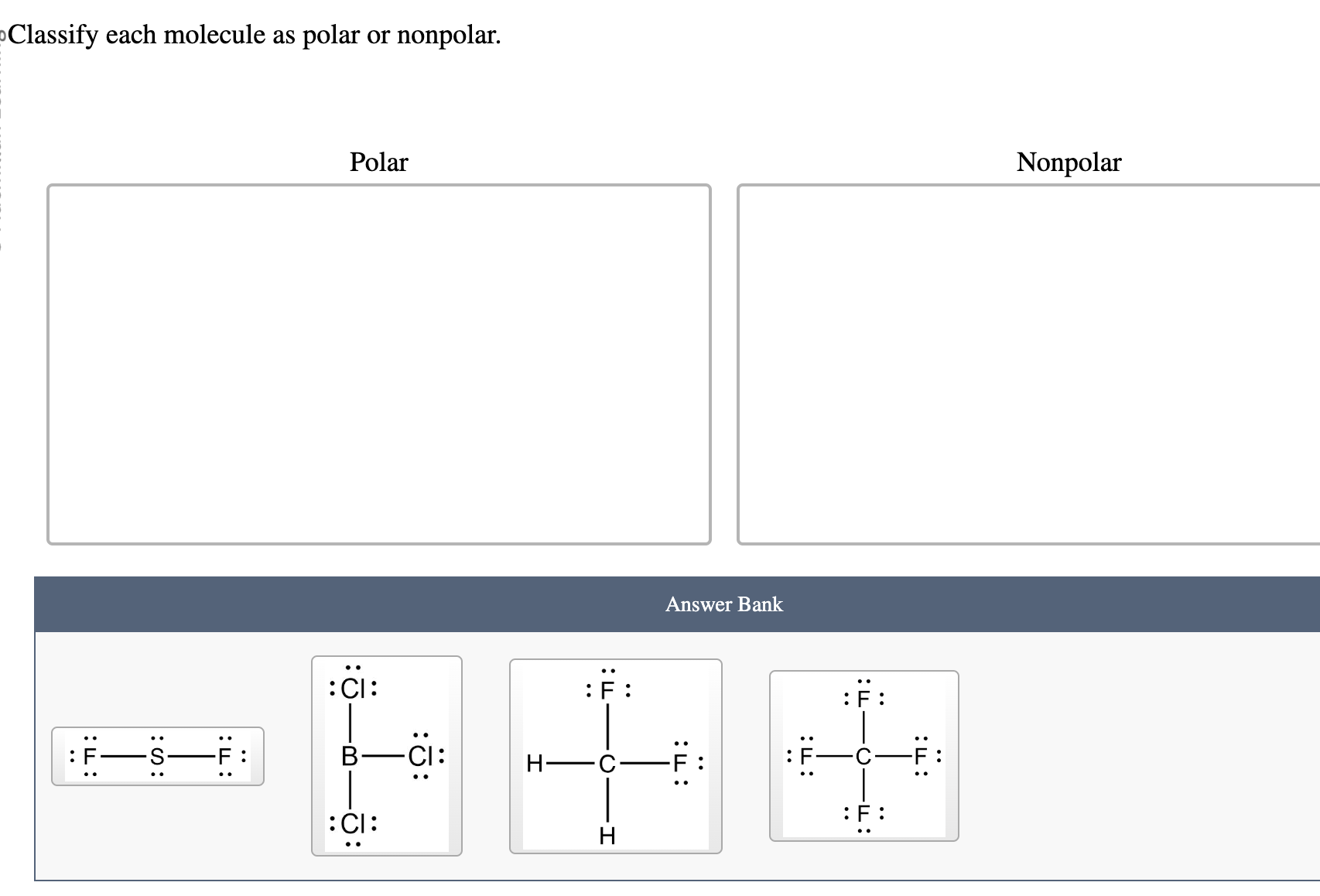
Classify Each Molecule As Polar Or Nonpolar

Hey there, science curious folks! Let's talk about molecules. You know, those tiny little building blocks of everything around us? Sometimes, when we're trying to figure them out, things can get a little… dramatic. It's like they have personalities, these molecules do. And one of the most fundamental ways we try to understand them is by asking: are they polar or nonpolar?
Now, I know what some of you might be thinking. "Polar? Nonpolar? Sounds like something from a really dry textbook." And you're not entirely wrong. But I'm here to tell you that classifying these little guys can be surprisingly entertaining, if you just embrace the silliness of it all. Think of it as assigning them social roles. Are they the life of the party, or the quiet observer in the corner?
My unpopular opinion? This whole polar/nonpolar thing is basically like deciding if a molecule is the friend who always wants to hang out and share everything, or the one who's perfectly happy doing their own thing, even if it means being a little bit aloof.
Let's start with the polar molecules. These are your social butterflies. They've got a little bit of an uneven distribution of "stuff" going on. Imagine a molecule where one end is slightly positive and the other end is slightly negative. It's like they have a favorite side! This unevenness means they're really good at interacting with other polar molecules. They’re the ones who will happily share their secrets (electrons) and get all tangled up with their friends.
Take water, for instance. The classic polar molecule! H₂O, as the chemists call it. Water is like the ultimate extrovert. It loves to dissolve things. It's the reason why sugar disappears in your tea or why salt vanishes in your soup. Water molecules are all about connection, forming these intricate networks of hydrogen bonds. They’re the glue that holds so much of our world together, and they do it by being enthusiastically polar.
Then you have molecules like ammonia (NH₃). Ammonia is also a bit of a show-off in the polarity department. It's got that uneven charge distribution, making it eager to mingle. It’s the friend who’s always ready for a deep conversation, or maybe a good gossip session. Polar molecules, in general, are like that friend who’s always approachable and eager to form bonds.
On the flip side, we have the nonpolar molecules. These are the stoic ones. The independent types. They’ve got their "stuff" (electrons) spread out pretty evenly. There’s no real favorite side. It’s like they're saying, "I’m good, thanks. I don’t need all that drama." They tend to hang out with other nonpolar molecules, forming their own little cliques.

Think about methane (CH₄). This is your quintessential nonpolar molecule. It's like the quiet roommate who pays their rent on time and never causes trouble. Methane just exists, minding its own business. It doesn't have a strong pull towards anything, making it less likely to dissolve in polar substances like water. It’s the molecule that would politely decline an invitation to a big, dramatic party.
Another common nonpolar molecule is carbon dioxide (CO₂). Now, CO₂ might seem a bit contradictory because it's made up of atoms that could create polarity. But the way they're arranged, it’s like a perfectly balanced tug-of-war. The forces cancel each other out. So, despite its components, the molecule itself is decidedly nonpolar. It's the molecule that’s surprisingly chill for its composition.
![[ANSWERED] Classify each molecule as polar or nonpolar Br 2 Polar 5 2 4](https://media.kunduz.com/media/sug-question-candidate/20220427215443053681-3769211.jpg?h=512)
And let’s not forget things like oils and fats. These are generally nonpolar. That’s why oil and water don’t mix! The polar water molecules are too busy socializing with each other to pay attention to the nonpolar oil molecules. The oil molecules are like, "Whatever, we’ll just hang out over here, thanks." They're perfectly happy in their own nonpolar company.
So, why does this even matter? Well, this simple classification has huge implications. It affects how things dissolve, how they react, and even how they behave in our bodies. It's the foundation for understanding everything from how medicines work to why some foods cook the way they do.
It's easy to get bogged down in the technicalities, but I like to think of it as a way of understanding the fundamental nature of these tiny particles. Are they clingy and social, or are they self-sufficient and independent? Polar molecules are the ones that hug tight, while nonpolar molecules prefer a more casual wave from across the room.
Next time you hear someone talking about polar and nonpolar molecules, just picture them as having distinct personalities. It makes the whole thing a lot more relatable, don't you think? And hey, if you've ever felt like a nonpolar molecule in a polar world, or vice versa, you're not alone. The molecules get it.