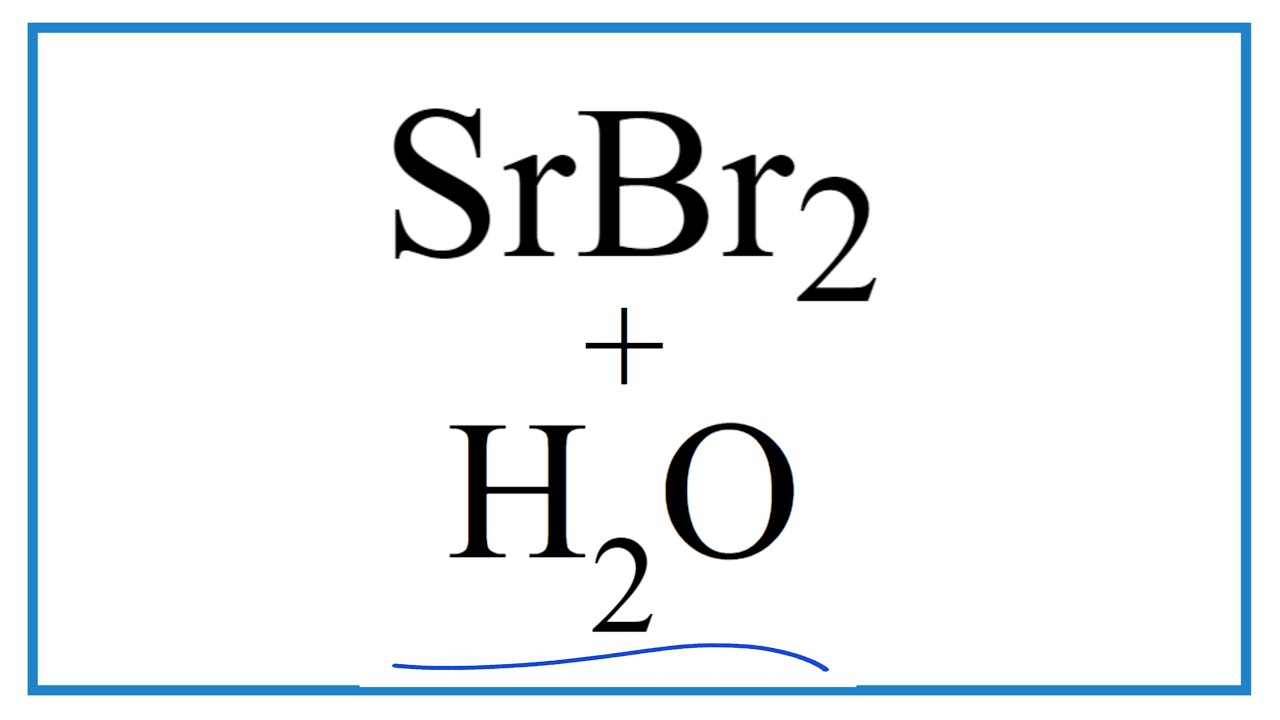Chemical Formula For Strontium And Bromine

Ever wondered what makes fireworks burst into dazzling color or how some medicines work their magic? It all comes down to the fascinating world of chemistry, and today, we're going to peek into a specific corner of it: the chemical formula for strontium and bromine. While it might sound a bit technical, understanding these simple building blocks of the universe can be surprisingly fun and even useful for everyday folks!
So, why is this particular combination worth a moment of your time? Well, strontium is one of the elements that gives fireworks their vibrant red hues – imagine the spectacular displays you've seen, and you're already connecting with this element! Bromine, on the other hand, is a bit more behind-the-scenes but plays a crucial role in many everyday items. For beginners dipping their toes into science, learning about how elements combine is like learning the alphabet of the universe. For families, it’s a fantastic way to spark curiosity in children and turn everyday observations into learning opportunities. Hobbyists, perhaps those interested in model rocketry or even certain types of photography, might find this knowledge directly applicable to their passions.
The chemical formula for the compound formed between strontium and bromine is SrBr₂. Let's break that down simply. Sr is the symbol for strontium, and Br is the symbol for bromine. The little number '2' tells us that for every one atom of strontium, there are two atoms of bromine. Think of it like a tiny recipe: you need one unit of strontium and two units of bromine to make this particular chemical compound. This compound, strontium bromide, has its own unique properties, often appearing as a white crystalline solid.
Must Read
There aren't too many drastic variations to the basic strontium and bromine formula itself, as it typically forms this specific ratio. However, you might encounter situations where other elements are involved, creating more complex compounds that utilize strontium or bromine. For example, in fireworks, strontium might be combined with other chemicals to achieve different shades of red or to enhance the intensity of the glow.
Getting started with understanding this is easier than you think! You don't need a fancy lab. Start by noticing the colors around you. Think about what makes a red sparkler. You can also look up other common chemical formulas for everyday substances like water (H₂O) or salt (NaCl) to see how symbols and numbers work together. Websites and books aimed at young scientists often have simple, engaging explanations of elements and compounds. Don't be afraid to ask questions; curiosity is the best tool in chemistry!
Understanding the basic chemical formulas like SrBr₂ might seem small, but it’s a step towards appreciating the intricate and beautiful science that shapes our world. It’s a reminder that even the most spectacular fireworks or useful inventions are built from these fundamental, tiny particles working in perfect harmony. Enjoy the journey of discovery!