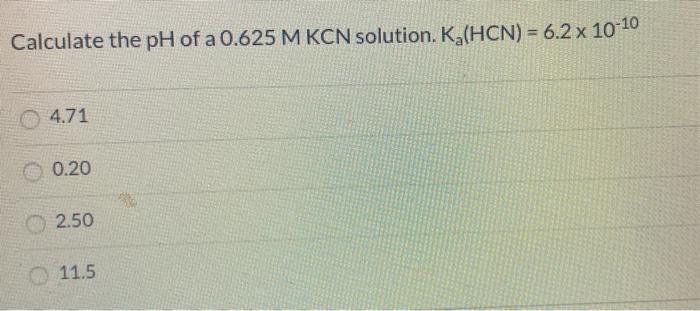
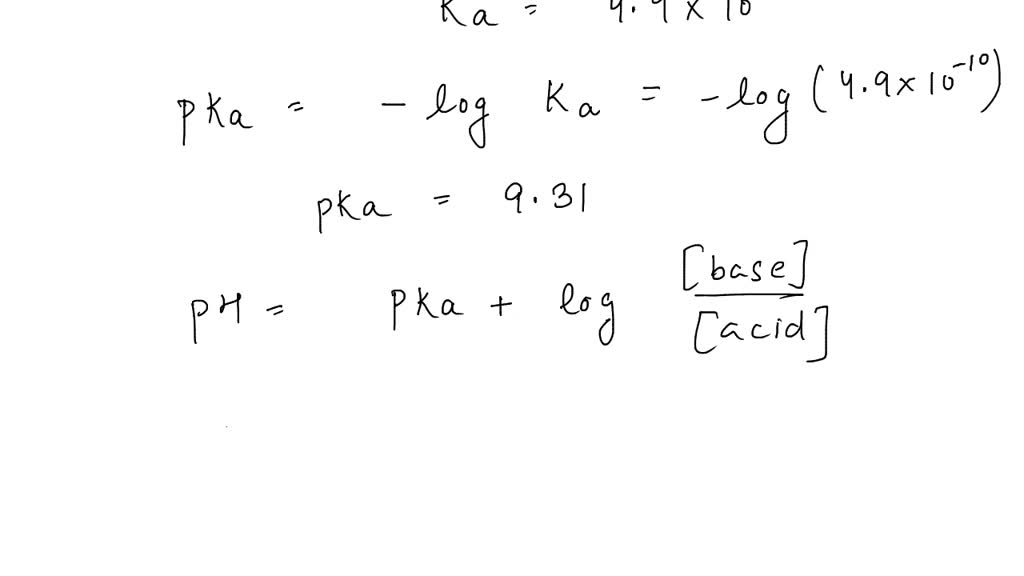Calculate The Ph Of A 0.100 M Kcn Solution

Ever wondered about the secret life of solutions? It might sound a bit scientific, but understanding things like pH is actually quite fascinating and incredibly useful in our everyday lives! Today, we're diving into a specific one: calculating the pH of a 0.100 M KCN solution. Sounds technical, right? But stick with me, and we'll make it as easy as pie, or rather, as easy as figuring out if your morning coffee is just right!
So, why is this whole pH calculation thing worth your time? For beginners in science, it's a fantastic way to get your feet wet with basic chemistry concepts without feeling overwhelmed. Think of it like learning your ABCs before writing a novel! For families, understanding pH can lead to fun experiments at home. Imagine testing the pH of different household liquids – it’s a mini science lab right in your kitchen! And for hobbyists, whether you're into brewing, gardening, or even making your own soaps, knowing the pH of your ingredients and final products can make a world of difference to the outcome. A well-balanced soap or a thriving plant often depends on the right pH.
Let's break down our specific challenge: calculating the pH of a 0.100 M KCN solution. KCN, or potassium cyanide, is a salt. When it dissolves in water, it doesn't just sit there. The cyanide ion (CN⁻) is the key player here. It's the conjugate base of a weak acid (HCN). Because it’s a conjugate base of a weak acid, it has a tendency to react with water, a process called hydrolysis. This reaction will produce hydroxide ions (OH⁻), making the solution slightly basic. And that's what we need to figure out – how basic!
Must Read
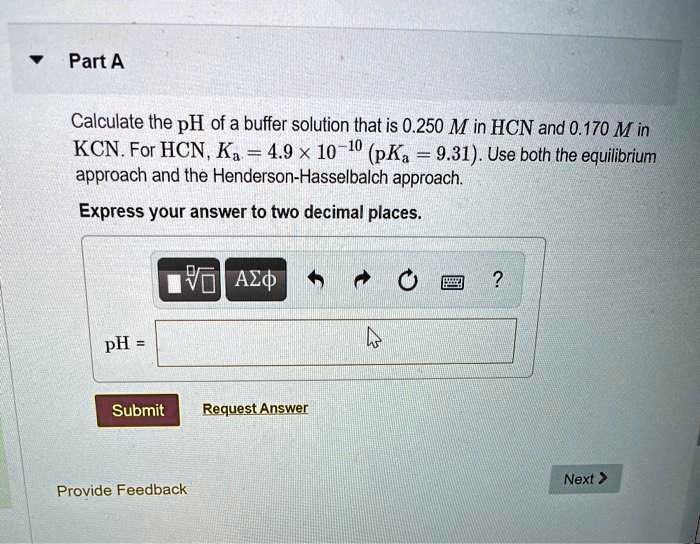
To get started, you’ll need a couple of things. First, a calculator! Second, you’ll need the base dissociation constant (Kb) for the cyanide ion. This is a value you can usually find in a chemistry textbook or by a quick online search. It tells us how strongly the cyanide ion will react with water. Once you have that, the calculation involves a few steps, typically setting up an equilibrium expression and solving for the concentration of hydroxide ions. From there, it’s a straightforward path to finding the pOH and then, finally, the pH. Don't worry if the terms sound daunting; the actual math is manageable!
Think of variations like calculating the pH of other salt solutions. What if you had a salt formed from a strong acid and a strong base? Those usually have a neutral pH. Or a salt from a weak acid and a weak base? Those can be a bit more complex, sometimes requiring both Ka and Kb values! The principles, however, remain the same: understanding how the ions behave in water.
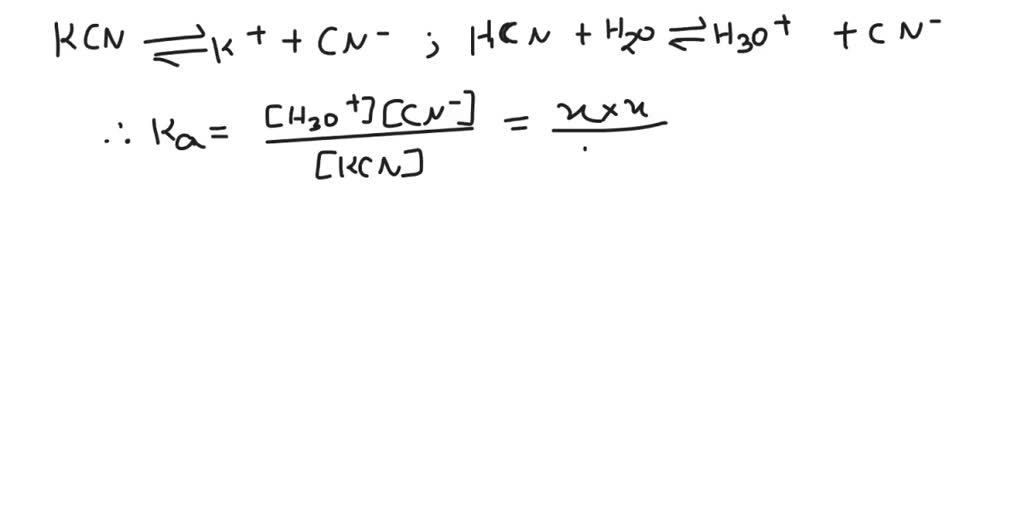
For a practical tip to begin, try this: Before tackling KCN, practice calculating the pH of a solution of a common salt like sodium acetate. It's also formed from a weak acid and strong base, so the hydrolysis concept is similar, but the numbers might be a little friendlier to start with. You can find the Kb for acetate easily!
So, while calculating the pH of a KCN solution might sound like a daunting chemistry problem, it’s actually a wonderful opportunity to explore how substances interact. It’s a puzzle with tangible results, and the satisfaction of solving it is a great reward. It’s a small window into a much larger, and often surprisingly accessible, world of chemistry!