Calculate The Formula Mass Of Ethanol C2h5oh
.jpg)
Hey there, science curious folks! Ever find yourself staring at a label, maybe on a bottle of hand sanitizer or even contemplating the ingredients in your favorite perfume, and see something like C₂H₅OH? It looks like a secret code, right? Like something you'd need a decoder ring to understand. But guess what? You absolutely don't! And today, we're going to crack that code together and calculate the formula mass of ethanol. Think of it as a fun little brain-tickler that will make you feel like a chemistry whiz, even if you haven't touched a Bunsen burner since high school.
So, what is this magical C₂H₅OH? That, my friends, is the chemical formula for ethanol! It's the stuff that makes some things… well, a little more festive (responsibly, of course!), and it's also a super useful ingredient in tons of everyday products. Knowing its formula mass isn't just about memorizing numbers; it's about understanding the building blocks of the world around us. Pretty cool, huh?
Now, before we dive into the nitty-gritty of calculating, let's talk about what "formula mass" actually means. Imagine you're building with LEGOs. Each LEGO brick is like an atom, and they all have a specific weight. The formula mass is simply the total weight of all the LEGO bricks (atoms) in one complete molecule of ethanol. Easy peasy, right? No complex machinery needed, just good old-fashioned addition!
Must Read
Ready to get your calculator (or even just your brain!) warmed up? Here’s what we need: the atomic masses of each element present in ethanol. Think of these as the weights of our individual LEGO bricks. You can find these handy numbers on the periodic table, which is basically a cheat sheet for all the elements. If you don't have one handy, a quick online search will bring up tons of them – they're everywhere!
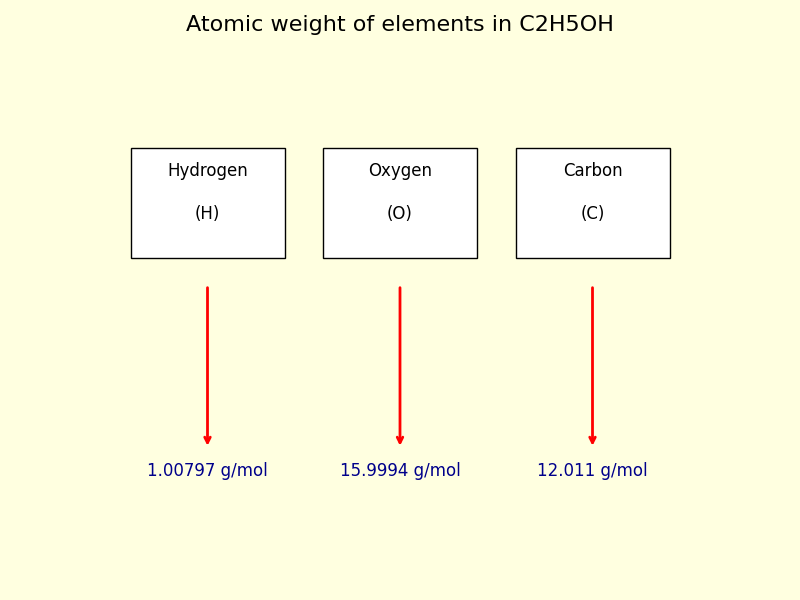
In C₂H₅OH, we have three types of atoms: Carbon (C), Hydrogen (H), and Oxygen (O). Let's grab their approximate atomic masses. We'll use simplified, rounded numbers for this fun exploration, but remember, in super-precise scientific work, they use more decimal places!
For Carbon (C), the atomic mass is approximately 12.01 atomic mass units (amu). For Hydrogen (H), it's about 1.01 amu. And for Oxygen (O), it's around 16.00 amu.
See? Not so scary, is it? Just three little numbers to get us started on our grand formula mass adventure.
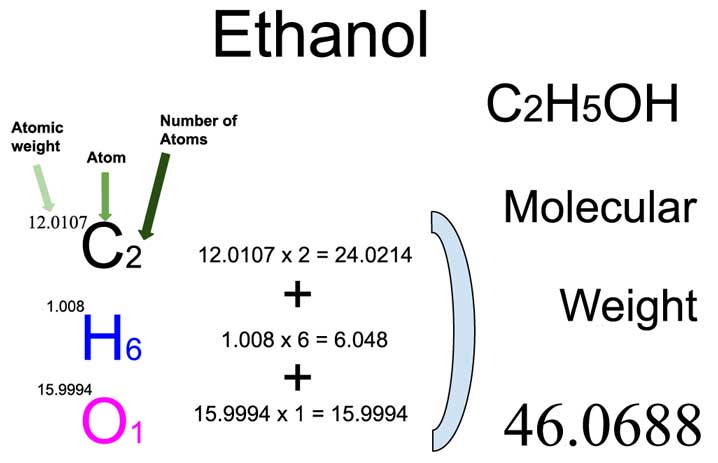
Now, let’s look back at our formula: C₂H₅OH. The little numbers after the element symbols are super important. They tell us how many of each atom are in one molecule of ethanol. So:
- The '2' after the 'C' means we have two carbon atoms.
- The '5' after the 'H' means we have five hydrogen atoms.
- And then there's an 'OH' at the end. This is a little grouping. Think of it like a pre-assembled LEGO component. We have one oxygen atom and one more hydrogen atom in this part. So, in total, that's one oxygen atom and, adding to the previous five, a grand total of six hydrogen atoms! Yes, you read that right – six! Sometimes chemistry likes to keep us on our toes!
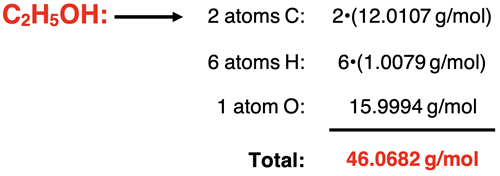
Let’s break it down systematically. This is where the "calculation" part really shines, and it’s surprisingly satisfying! We’re going to multiply the number of each atom by its atomic mass and then add all those results together.
Calculating the Carbon Contribution:
We have 2 carbon atoms, and each weighs about 12.01 amu. So, 2 * 12.01 amu = 24.02 amu.
Isn't it neat how we're essentially weighing out our ingredients? Like baking a cake, but with molecules!
Calculating the Hydrogen Contribution:
Remember, we have a total of 6 hydrogen atoms (5 from the C₂H₅ part and 1 from the OH part). Each hydrogen atom weighs about 1.01 amu. So, 6 * 1.01 amu = 6.06 amu.
See how that 'OH' at the end cleverly adds another hydrogen into the mix? It’s like a little chemical bonus!
Calculating the Oxygen Contribution:
We have 1 oxygen atom, and it weighs about 16.00 amu. So, 1 * 16.00 amu = 16.00 amu.
And there you have it! The individual weights of all the different types of atoms in our ethanol molecule.

Now for the grand finale! We just need to add up all these individual contributions to get the total formula mass of ethanol.
Total Formula Mass = (Carbon Contribution) + (Hydrogen Contribution) + (Oxygen Contribution)
Total Formula Mass = 24.02 amu + 6.06 amu + 16.00 amu
Total Formula Mass = 46.08 amu
Ta-da! You just calculated the formula mass of ethanol! Give yourself a pat on the back. You’ve successfully navigated the fascinating world of chemical formulas and atomic weights. It’s not just numbers on a page; it’s a glimpse into the tiny, fundamental particles that make up everything we see, touch, and even smell!
Why is this fun and inspiring, you ask? Because it unlocks a new way of looking at the world! Suddenly, that label on the hand sanitizer isn't just a list of chemicals; it's a recipe for something useful, built from atoms with specific weights. This knowledge empowers you. It sparks curiosity about how things are made and how they work. It’s the beginning of a lifelong journey of discovery!
And the beauty of it is, this same principle applies to calculating the formula mass of any chemical compound. Water (H₂O)? You can do it! Table salt (NaCl)? Absolutely! Every compound has its own unique atomic "fingerprint" represented by its formula mass.
So, the next time you encounter a chemical formula, don't be intimidated. See it as an invitation to explore. You've already taken a fantastic first step by understanding ethanol. Keep that curiosity alive, and who knows what other amazing scientific secrets you'll unlock!