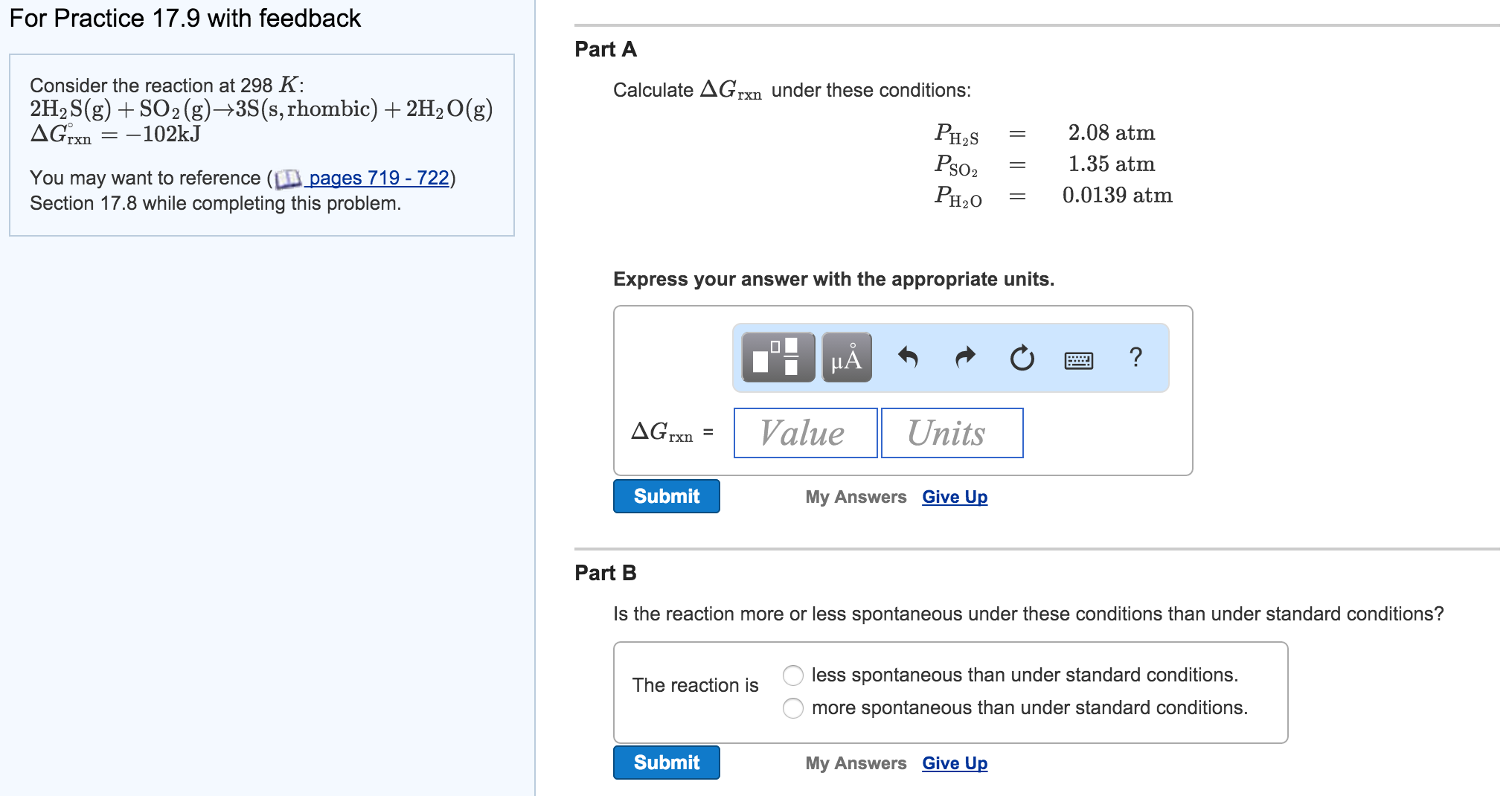
Calculate Delta G For The Reaction At 298 K

Hey there, lovely humans! Ever have one of those days where everything just… flows? You wake up, the coffee's perfect, you find a parking spot right up front, and your favorite song plays on the radio? That’s kind of like what we’re talking about today, but in the world of tiny molecules. We’re going to chat about something called Delta G, or Gibbs Free Energy, and why understanding it is actually way more interesting and useful than you might think. No super-science jargon, promise! Think of it as a peek into the secret life of chemical reactions, happening all around you, all the time.
So, what’s this Delta G all about? Imagine you're standing at the top of a hill, holding a perfectly ripe apple. You're going to let go, right? That apple is going to roll downhill. It's a natural, spontaneous thing. It's not going to suddenly decide to fly back up the hill. Delta G is basically the thermodynamic way of saying whether something, like a chemical reaction, is going to be like that apple rolling downhill – a pretty easy, natural process – or if it’s going to be like trying to push that apple uphill – requiring a whole lot of effort.
In simpler terms, Delta G tells us if a reaction will happen on its own, without us having to give it a big nudge. It's like the universe's energy check. If Delta G is negative, hooray! The reaction is happy to happen. If it's positive, well, it’s going to need some serious encouragement. And if it’s zero, it’s just chilling, balanced like a perfectly stacked Jenga tower.
Must Read
Why Should You Care About This Tiny Molecule Drama?
Okay, you might be thinking, "I’m not a chemist, why do I need to know about invisible molecules doing their thing?" Well, turns out, you are a chemist, and so am I! Every single thing your body does, from digesting that delicious breakfast to your heart beating, is a symphony of chemical reactions. Understanding Delta G helps us understand life itself.
Think about it: your body needs energy to function. Where does that energy come from? From reactions that have a negative Delta G, meaning they happen easily and release energy. It's like your body is constantly finding ways to make those "downhill rolls" happen so you can live, breathe, and even binge-watch your favorite show. Without these spontaneous reactions, you'd be… well, not here!
And it's not just about you! This knowledge is behind so many things we take for granted. Ever wonder how a battery powers your phone? Or how plants turn sunlight into food? Or how that delicious bread rises? All of it involves understanding these energetic "pushes and pulls" of chemical reactions, which Delta G helps us quantify.

Let's Talk About the "Calculate" Part (Without Making Your Brain Hurt!)
So, how do we figure out if our apple is going to roll or needs a push? Scientists use a formula. And don't worry, we're just going to peek at it, not dive headfirst into a calculus textbook. The formula is often shown as:
ΔG = ΔH - TΔS
Let's break down these mysterious letters:

- ΔG (Delta G): This is our main man, the overall "spontaneity" of the reaction. Negative means spontaneous, positive means not. Easy peasy.
- ΔH (Delta H): This is the enthalpy change. Think of it as the heat involved. Is the reaction giving off heat (exothermic, like a cozy fireplace, usually favors spontaneity) or is it sucking up heat from its surroundings (endothermic, like an ice pack, usually makes spontaneity harder)?
- T: This is simply the temperature, measured in Kelvin. Why Kelvin? Because it's an absolute scale, meaning zero Kelvin is the absolute absence of heat. Plus, it’s often a nice round number like 298 K (which is room temperature, by the way – about 25 degrees Celsius).
- ΔS (Delta S): This is the entropy change. This one's a bit more fun! Entropy is basically a measure of disorder or randomness. Think of a perfectly made bed versus a room after a toddler has had a party. The toddler's room has much higher entropy. Reactions that increase disorder (like breaking a big molecule into smaller ones, or dissolving a solid into a liquid) tend to be more spontaneous.
Putting It All Together: The Apple Analogy Revisited
Let’s imagine our apple. Releasing the apple (ΔG negative) is easier if:
- The hill is steep (like a big negative ΔH – a lot of potential energy to release).
- The apple is already a bit wobbly and ready to fall (high entropy, ΔS positive).
Now, what if the hill is flat, but the apple is super sticky to your hand? You’d need to give it a good shove to get it moving. That's like a reaction with a positive ΔH (needs energy input) and maybe a negative ΔS (getting more ordered, which is unlikely for an apple falling, but you get the idea). You'd need to apply force to overcome these unfavorable conditions.
Temperature (T) plays a role too! Imagine you're trying to melt ice. At freezing temperatures, it's a tough sell. But crank up the heat, and that ice melts all on its own! The same applies to reactions. Sometimes, just adding enough heat can make a reaction that was stubborn at room temperature suddenly decide to happen.
Real-Life Examples That Make You Go "Aha!"
Let's think about a few everyday scenarios:

1. Cooking: When you bake a cake, you're initiating a bunch of chemical reactions. Some are driven by heat (ΔH), others by creating new, more stable molecules (which might lower energy). The whole process is orchestrated to end up with a delicious, edible product!
2. Dissolving Sugar in Tea: When you add sugar to your hot tea, it dissolves. This usually increases disorder (ΔS is positive – those sugar molecules are spreading out!). Often, dissolving processes also release a little bit of heat (ΔH can be negative or slightly positive). The combination usually makes dissolving a spontaneous process at room temperature.
3. Your Muscles Working: Your body breaks down glucose (sugar) to produce energy. This is a complex series of reactions, but the overall process is designed to release energy (negative ΔH) and be very efficient. Your body is a master at finding reactions with favorable Delta G values to keep you moving!

4. Rusting of Iron: Have you ever seen an old bicycle left out in the rain? It rusts. This is a spontaneous reaction (negative ΔG) where iron reacts with oxygen to form iron oxide. It’s the universe’s way of saying, "Let's make things a bit more stable, even if it looks messy." The iron is "happier" as rust.
Why 298 K is Special (and Not Just a Random Number)
You’ll often see calculations done at 298 K. This is essentially standard temperature in the chemistry world, which is 25 degrees Celsius or about 77 degrees Fahrenheit. It’s our convenient “room temperature” baseline. By using this standard temperature, scientists can compare the spontaneity of different reactions without the variable of temperature making things complicated. It’s like agreeing to measure everyone’s height with the same tape measure and while they're all standing up straight.
The Big Picture: It’s All About What's "Favored"
Ultimately, Delta G is about what the universe prefers. Is it easier for a reaction to happen and release energy, or become more disordered? If the answer is yes to either of those, the reaction is more likely to be spontaneous.
So, the next time you see something happen naturally – a leaf falling, sugar dissolving, or even your own body working – you can give a little nod to Delta G. It's the invisible force, the energy accountant, making sure the world keeps turning in a way that, for the most part, is energetically "easy." It’s a beautiful, subtle dance of energy and order, and now you’re a little more in on the secret!