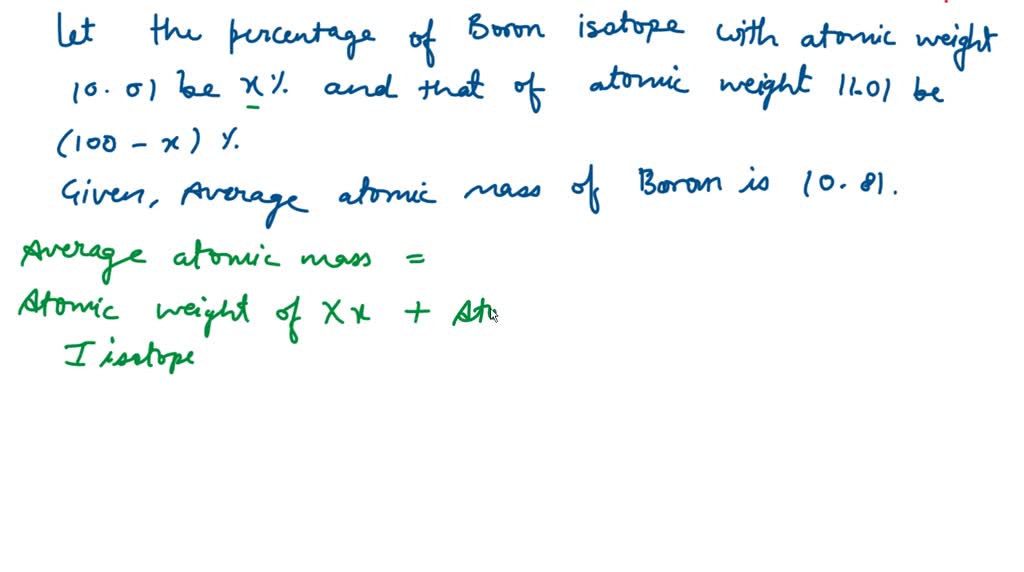Boron Has Two Naturally Occurring Isotopes
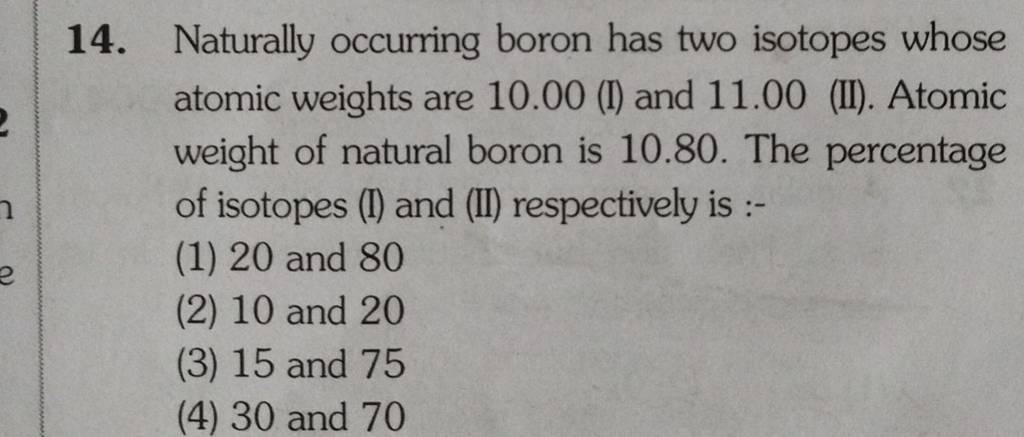
Alright, gather 'round, you magnificent collection of humans who probably think a proton is just a fancy word for a really loud dude! Today, we're diving headfirst into the wonderfully weird world of ... boron. Yeah, I know, thrilling, right? But stick with me, because this humble element, the one you might vaguely remember from your high school chemistry class as being part of something or other, has a secret. And it’s a secret that’s, well, doubly interesting. Boron, my friends, is a bit of a twin!
Now, when I say twin, I don't mean it has a doppelgänger running around the periodic table, stealing its electrons or trying to frame it for stealing the last cookie. No, no. Boron’s twin-ness is much more subtle, much more... atomic. It turns out, when nature was busy crafting all the elements, it got a little creative with boron. Instead of just making one kind, it decided to whip up two versions. And these aren't just any old versions; these are called isotopes.
Think of isotopes like identical twins who, despite looking exactly the same on the outside, have slightly different personalities. Or, in the case of atoms, slightly different weights. That's the key difference between these boron twins. They’ve both got the same number of protons – that’s what makes them boron in the first place, like their shared last name. But one of them, the slightly heavier twin, has an extra neutron hanging out in its nucleus. It's like having an extra passenger on a bus that was already full, but nobody seems to mind.
Must Read
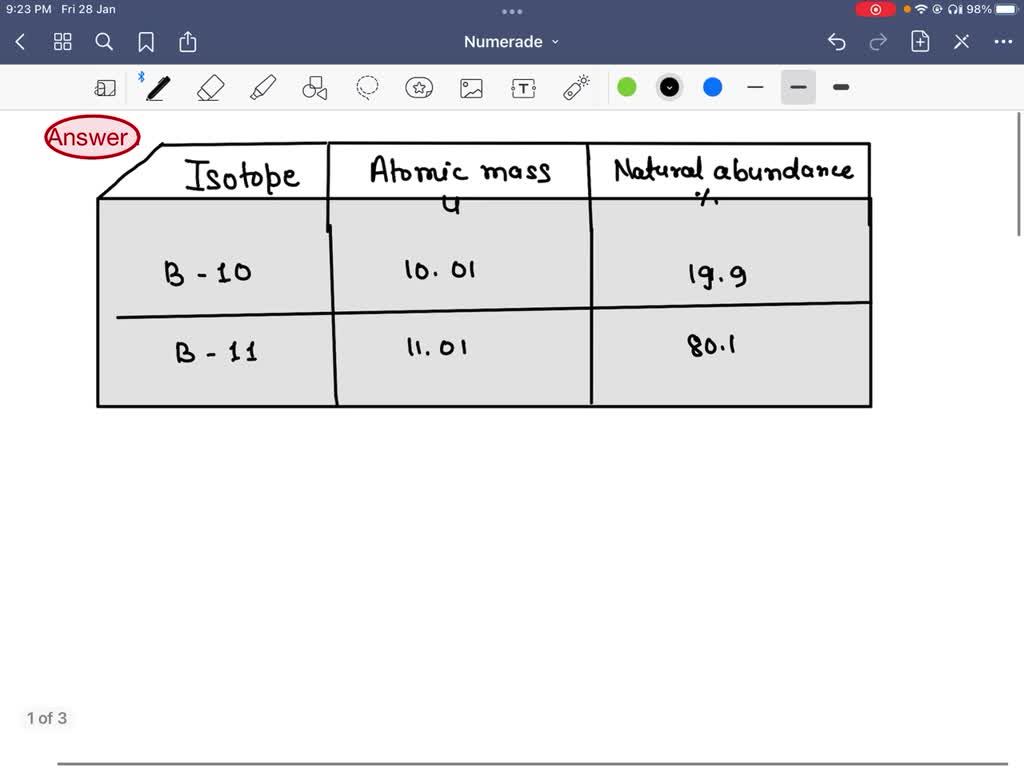
So, we've got Boron-10 and Boron-11. Say it with me: Boron-TEN and Boron-ELEVEN. It’s not exactly a catchy superhero duo, but hey, they get the job done. Boron-10 is the slightly lighter, more agile one, making up about 20% of all the boron you’ll find out there in the wild. And Boron-11? That’s the beefier, more abundant one, strutting its stuff at a hefty 80%. It’s like the popular kid in school who’s just naturally better at everything, or at least, more common.
Why should you care about these two slightly different versions of boron? Well, besides the fact that it’s a fun party trick for your next awkward family gathering (“Did you know boron has isotopes?” – watch the glazed-over eyes commence!), these isotopes have some seriously cool jobs. It’s like they were designed for them. Imagine a world without boron isotopes – you’d be stuck with things like duller fireworks, less effective neutron shields (more on that later, if you dare!), and maybe even slightly less impressive glass. Who wants that?

The Case of the Missing Neutron (or the Extra One!)
Let’s break down this neutron situation a little more. Protons are like the core identity of an atom. They’re the VIPs, the ones that decide, “Yep, you’re a boron!” Neutrons, on the other hand, are the quiet supporters. They just hang out in the nucleus, keeping things stable and adding a bit of heft. Boron, bless its little atomic heart, just couldn’t make up its mind. It’s like trying to decide between a double scoop and a triple scoop – both are good, but one’s got a bit more substance.
So, Boron-10 has 5 protons and 5 neutrons. That’s a nice, neat little package. Boron-11, the popular kid, has 5 protons and 6 neutrons. That extra neutron makes it just a smidge heavier. It’s like the difference between wearing a light sweater and a slightly thicker cardigan. You might not notice it from across the room, but up close, there’s a subtle distinction.

This difference in weight might sound trivial, but in the tiny world of atoms, it’s a big deal. It affects how they interact, how they behave in different environments, and what they’re good at. It’s like how a marathon runner and a sprinter might have similar muscle groups, but their training and capabilities are wildly different due to subtle variations. Boron-10 and Boron-11 are the marathon runner and the sprinter of the boron world.
Boron's Superpowers (Thanks to Its Twins)
Now, for the juicy part: what can these isotopic twins do? Boron, in general, is quite a useful element. It’s found in detergents, glass, ceramics, and even some fertilizers. But its isotopes give it some special talents.
Let’s talk about Boron-10. This guy is a neutron hog. Seriously, it’s like a black hole for neutrons. When a neutron bumps into a Boron-10 atom, the atom can’t resist. It sucks that neutron right in, and then, poof! The atom splits into a couple of other, smaller atoms and releases a burst of energy. This is like a tiny, controlled atomic explosion. Pretty neat, huh?
Because of this neutron-gobbling superpower, Boron-10 is absolutely crucial for nuclear reactors. You know, those big, powerful places that generate electricity? Boron-10 is used as a control rod. Imagine the reactor is a super-energetic toddler. Boron-10 is like the patient parent, calmly stepping in to absorb some of that excess energy (neutrons) and keep things from getting too wild. Without it, those reactors could go haywire. So, next time you flick on a light switch, give a silent nod to Boron-10 for keeping things in check.
And what about Boron-11, the more common twin? While it’s not as much of a neutron fanatic as Boron-10, it’s still incredibly important. Boron-11 is used in many of the everyday applications of boron that we mentioned. It’s part of the glass that makes your windows sparkly, the ceramics in your coffee mug, and even the special ingredients in some advanced alloys. It’s the workhorse, the reliable friend who’s always there, doing its job without much fuss.

Furthermore, the ratio of Boron-10 to Boron-11 in a sample can actually tell scientists something about where that boron came from and what kind of environment it was in. It’s like an atomic fingerprint! So, if you find a piece of boron rock, its isotopic signature could reveal its ancient geological journey. Pretty cool detective work for atoms, wouldn't you say?
A Cosmic Connection
But wait, there's more! The story of boron isotopes isn't just limited to Earth. It stretches all the way back to the Big Bang! Scientists believe that in the chaotic early moments of the universe, both Boron-10 and Boron-11 were forged. Their relative abundances, the fact that there's more Boron-11 than Boron-10, are actually clues that help us understand those very first moments of cosmic creation. It’s like finding an ancient diary that tells us what happened at the dawn of time. Mind-blowing, right?
So, the next time you encounter boron – maybe in a glass of wine (some wines have boron compounds!), or in a high-tech gadget – remember its double life. Remember the two subtly different, yet equally important, twins, Boron-10 and Boron-11. They're a reminder that even in the seemingly simple building blocks of our universe, there's a hidden complexity and a story waiting to be told. And that, my friends, is far more interesting than you might have initially thought about something as seemingly mundane as boron. Cheers to the isotopic twins!