Bisphenol A Solubility In Organic Solvents

Okay, let's talk about something that sounds super fancy but is actually kind of… everywhere. We're diving into the world of Bisphenol A, or as the cool kids (and scientists) call it, BPA. Now, before you scroll away thinking this is going to be a dry chemistry lesson, hold up! We're approaching this from a place of sheer, unadulterated curiosity, fueled by probably too much coffee and a healthy dose of "huh, that's interesting."
So, BPA. You might have heard of it. It’s that thing that used to be in a lot of plastics. Think reusable water bottles, those handy food containers, and even the lining of some tin cans. It’s like the ghost in the machine of our modern convenience life. And like any good ghost, it has its own peculiar habits. One of those habits is its relationship with organic solvents. Sounds a bit like a secret handshake, doesn't it?
Now, what exactly are these "organic solvents"? Imagine them as the ultimate party animals of the chemical world. They're the solvents that love to hang out with other molecules that aren't water. Think of things like ethanol (that's in your hand sanitizer, by the way – useful, right?), acetone (nail polish remover, anyone?), or even good old isopropyl alcohol. These guys are the life of the solubility party. They're not picky. They're like that friend who can make conversation with literally anyone.
Must Read
And then there’s our friend, BPA. BPA is a bit of a social butterfly. It’s not a huge fan of plain old water. Water, bless its simple heart, is like the quiet guest at the party who just sips their water and observes. BPA prefers a more… lively atmosphere. It likes molecules that are a bit more like itself. Molecules that have that certain je ne sais quoi that makes them buddy up.
So, when you introduce BPA to an organic solvent, it's like sending two old pals to a reunion. They just get each other. The BPA molecules happily mingle, dissolve, and become one with the solvent. It’s a beautiful friendship, really. The solvent, with its own kind of molecular charm, just opens its arms (or whatever chemical equivalent of arms it has) and welcomes BPA in. It’s like finding your tribe, but on a microscopic level.

Think about it this way: If water is a cozy, quiet cafe, BPA is the person who’d rather be at a loud, bustling pub. It needs that extra buzz, that extra oomph to really spread out and feel comfortable. And the organic solvents? They’re the ultimate pub. They’ve got the music, the chatter, the whole vibe that BPA just adores.
This is where things get a little bit… interesting. Because BPA likes to dissolve in these solvents, it means it can move around. It’s not stuck. If BPA is in a plastic container and that container comes into contact with an organic solvent – say, you accidentally spill some hand sanitizer on a plastic toy, or you're cleaning some plastic with a particular cleaner – that BPA might decide it’s time to pack its bags and go for a little adventure in the solvent. It's like a tiny, chemical escape artist.

And this is where my unpopular opinion comes in. While everyone else is busy stressing about BPA leaching into their water bottles (which, yes, is a valid concern!), I can't help but be a little bit fascinated by the how. It's not about saying "don't worry about BPA." It's more like, "wow, chemistry is wild, isn't it?" The fact that a molecule can be so happy to dissolve in one thing but not another is just mind-bogglingly cool. It’s like a tiny, chemical personality test happening all around us.
Imagine a scientist, probably wearing a lab coat (because that's what scientists do, right?), carefully measuring out different organic solvents. They’re not just doing it to be difficult. They’re trying to understand how BPA behaves. They’re like detectives, figuring out the escape routes, the preferred hangouts, the secret life of BPA. And their tool? The humble, yet powerful, organic solvent.
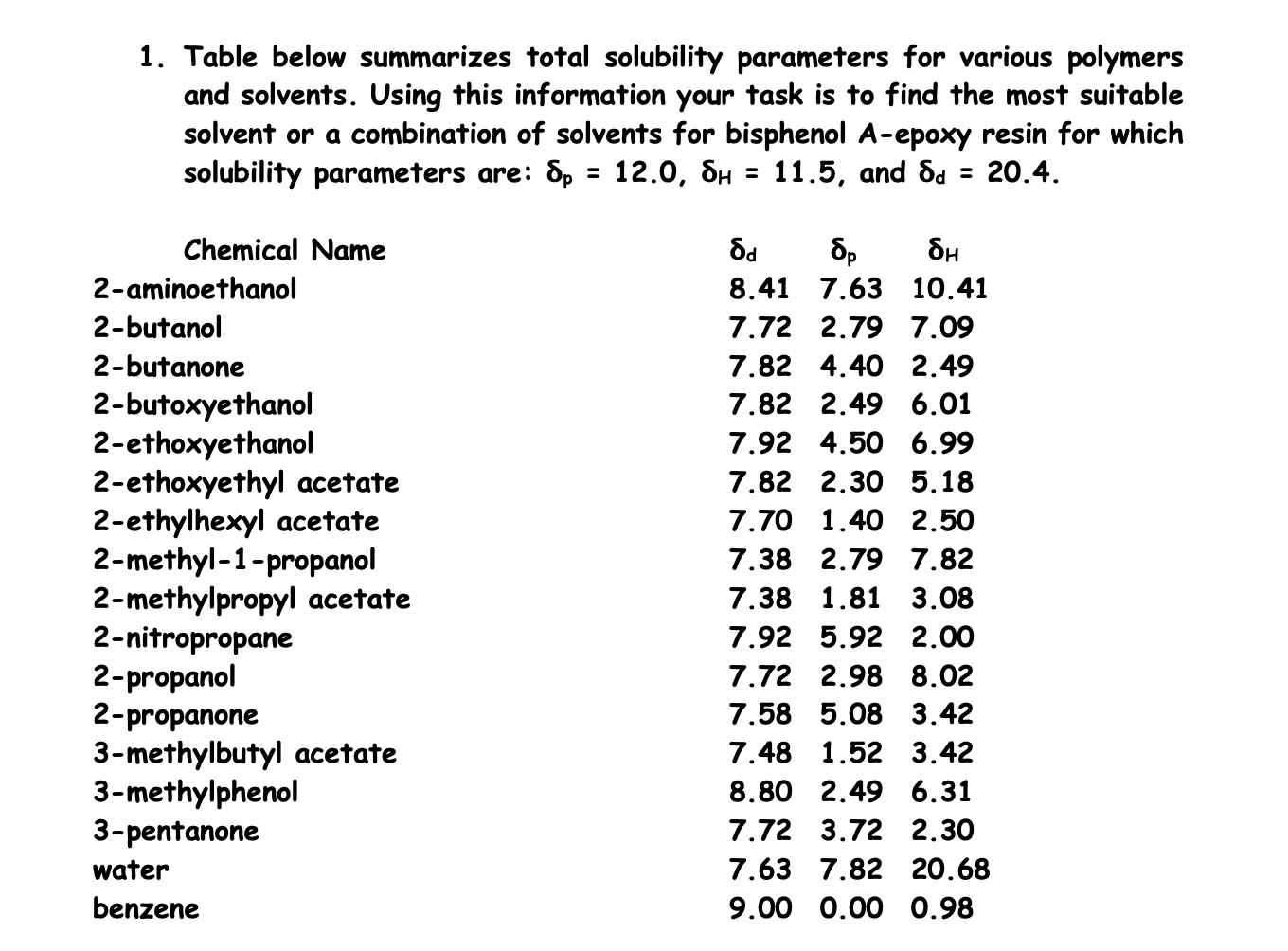
It's a testament to the incredible diversity of the molecular world. Water, the universal solvent for so many things, is just not BPA's cup of tea. It needs something a little more… organic. Something with that special molecular structure that says, "Come on in, the water's… well, it's not water, but you'll love it here!"
So next time you see a bottle of rubbing alcohol or even a nail polish remover, spare a thought for our friend BPA. It’s probably out there, somewhere, having a grand old time dissolving in its favorite organic solvents. It's a small, chemical drama playing out, and honestly, I find it oddly charming.
It’s a reminder that even the most mundane things in our lives have a hidden complexity, a whole universe of interactions happening on a scale we can’t even see. And the solubility of BPA in organic solvents is just one tiny, but surprisingly entertaining, chapter in that story. It’s not just about chemistry; it’s about the fascinating, sometimes quirky, ways molecules choose to mingle. And who doesn't love a good mingling story?