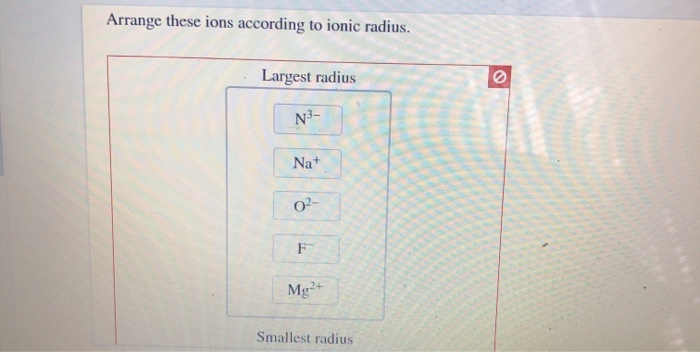
Arrange These Ions According To Ionic Radius

Hey there, science fans and curious minds! Ever wondered if you could play a little game with tiny charged particles, like a super-miniature game of "bigger or smaller"? Well, get ready, because we're diving into something pretty neat called "Arrange These Ions According to Ionic Radius."
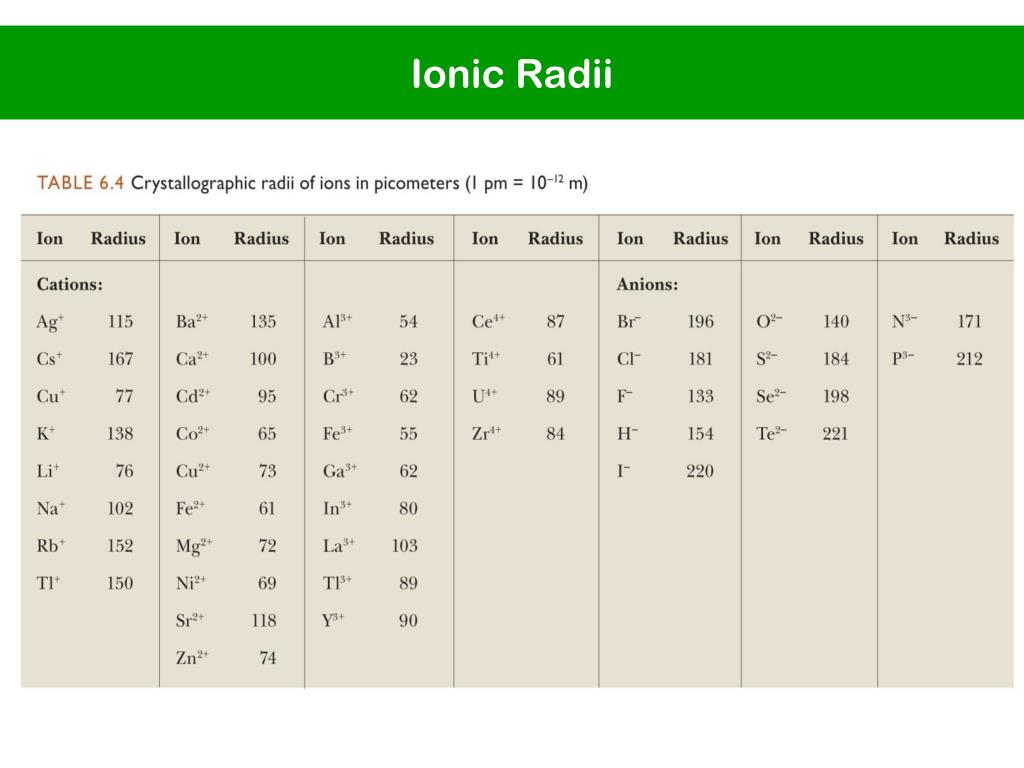
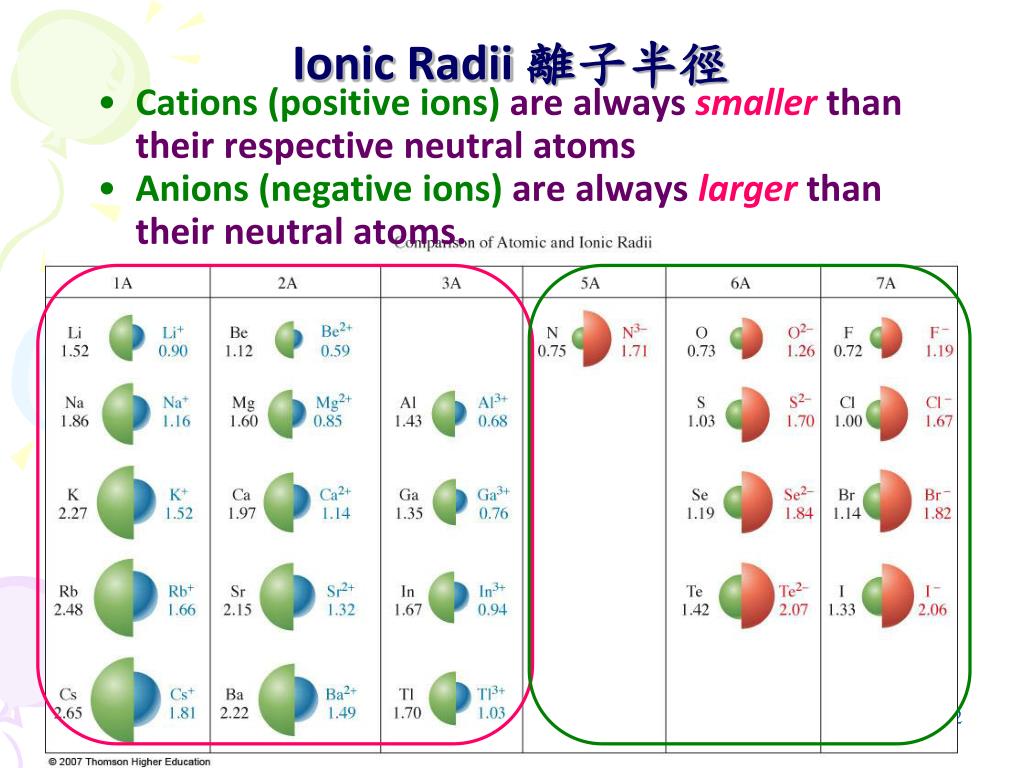
Now, you might be thinking, "Ionic radius? What's that?" Don't sweat it! Think of it like this: ions are atoms that have a little bit of a superhero power – they've either gained or lost an electron, giving them a charge. And ionic radius? That's just the fancy way of saying how big or small these charged-up atoms are. It's like measuring their invisible bubbles!
So, what's the big deal about arranging them? This is where the fun really kicks in. Imagine you're a detective, but instead of solving crimes, you're solving the puzzle of these ions. You're given a list of them, and your mission, should you choose to accept it, is to put them in order from smallest to largest, or vice-versa. It sounds simple, right? But oh boy, there's a whole lot of cool science packed into that little task.
Must Read
Why is this so entertaining? Because it's like a mental workout disguised as a game. You have to look at each ion and think, "Hmm, this one lost an electron, so it probably shrunk. This one gained an electron, so it probably got a bit puffier." It's a delightful dance of attraction and repulsion, of electron clouds zipping around. You start to see patterns emerge, like magic tricks happening right before your eyes. It's all about understanding how these little guys behave in the grand atomic ballroom.
What makes it special? Well, it’s more than just memorizing facts. It’s about understanding. When you can successfully arrange these ions, you’re not just getting a high score; you’re getting a glimpse into the fundamental forces that hold the universe together. You're developing an intuition for how atoms interact, and that's pretty darn powerful. It's like learning to speak a secret language, the language of chemistry. And once you start to understand it, you see it everywhere!

Think about it: every time you see a piece of metal, a pinch of salt, or even a colorful gemstone, you're looking at the results of these ionic interactions. The way these ions arrange themselves, their sizes, their charges – it all contributes to the properties of the materials we encounter every single day. So, this seemingly simple task is actually a doorway to understanding the building blocks of everything around us.
And the best part? It’s totally accessible. You don’t need a fancy lab coat or a supercomputer to get started. You just need your brain and a little bit of curiosity. You can find these challenges online, in textbooks, or even create your own! It’s a fantastic way to engage with science without feeling overwhelmed. It’s like a friendly challenge from Mother Nature herself, inviting you to explore her intricate designs.

Imagine you’re presented with a group like Lithium ion (Li⁺), Sodium ion (Na⁺), and Potassium ion (K⁺). You see they all have a plus charge, meaning they've lost an electron. But wait, they're different elements! How do you figure out their sizes? This is where the fun detective work begins. You might recall that as you go down a column in the periodic table, atoms generally get bigger. So, does that apply to their ions too? Spoiler alert: it often does!
Or consider a scenario with ions like Chloride ion (Cl⁻) and Sulfide ion (S²⁻). Both have gained electrons, making them negatively charged. But one has a minus one charge, and the other has a minus two. How does that extra electron affect their size? This is the kind of delightful puzzle that makes “Arrange These Ions According to Ionic Radius” so captivating. You’re not just passively absorbing information; you’re actively participating in the discovery process.

It’s a bit like a puzzle where the pieces are invisible, and the rules are based on the fundamental laws of physics. You get a thrill when you correctly place a larger ion next to a smaller one, knowing you've understood the underlying principles. It’s a satisfying feeling, like finally figuring out a tricky riddle.
So, if you're looking for a way to spice up your learning, to engage your brain in a fun and rewarding way, I highly recommend giving "Arrange These Ions According to Ionic Radius" a try. It’s a fantastic introduction to the world of chemistry, a playful challenge that reveals the secrets of the tiny world of atoms. You might just find yourself hooked, eager to explore more of the amazing science hidden in plain sight!
Who knows what other atomic mysteries you'll uncover? It’s a journey that’s both educational and incredibly entertaining. So, go ahead, embrace your inner science detective and see if you can crack the code of ionic radius. You might be surprised at how much fun you have!

.jpg)

