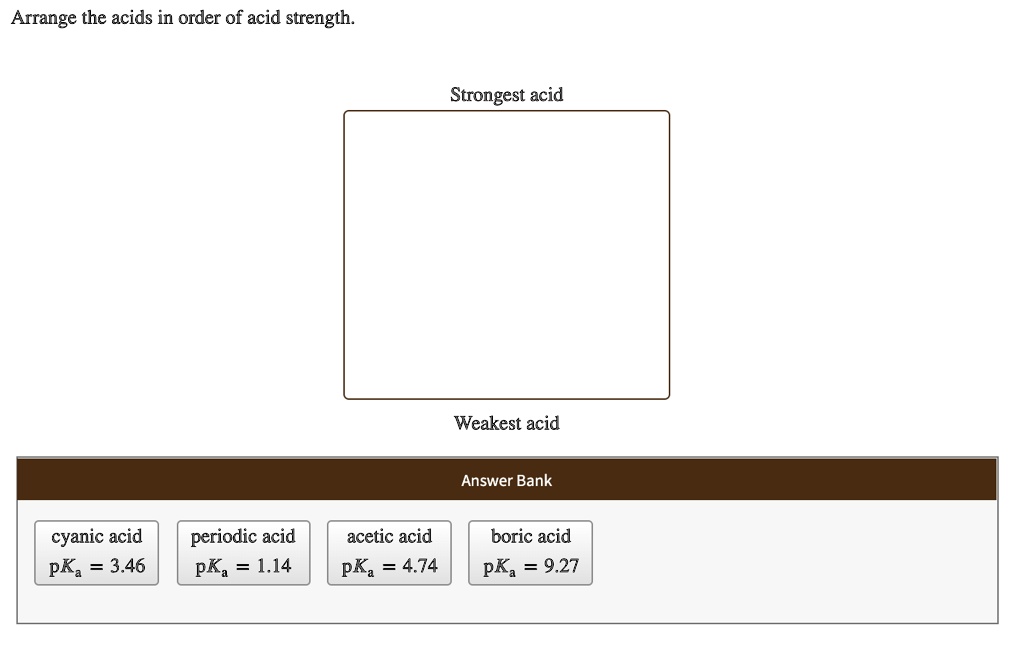
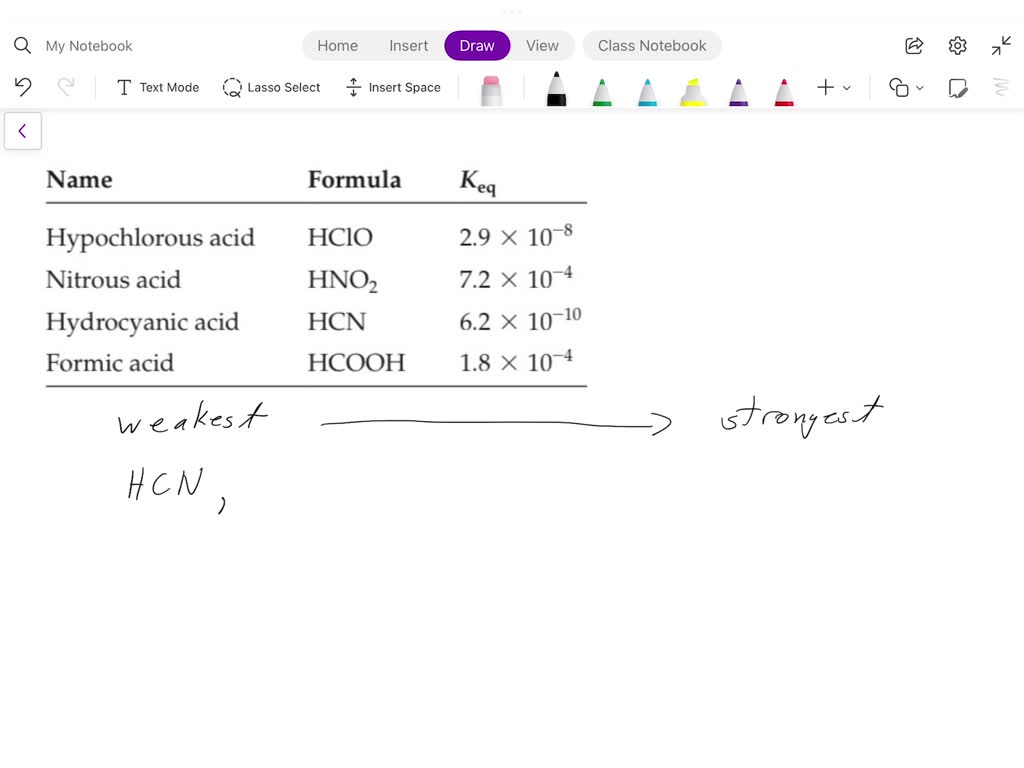
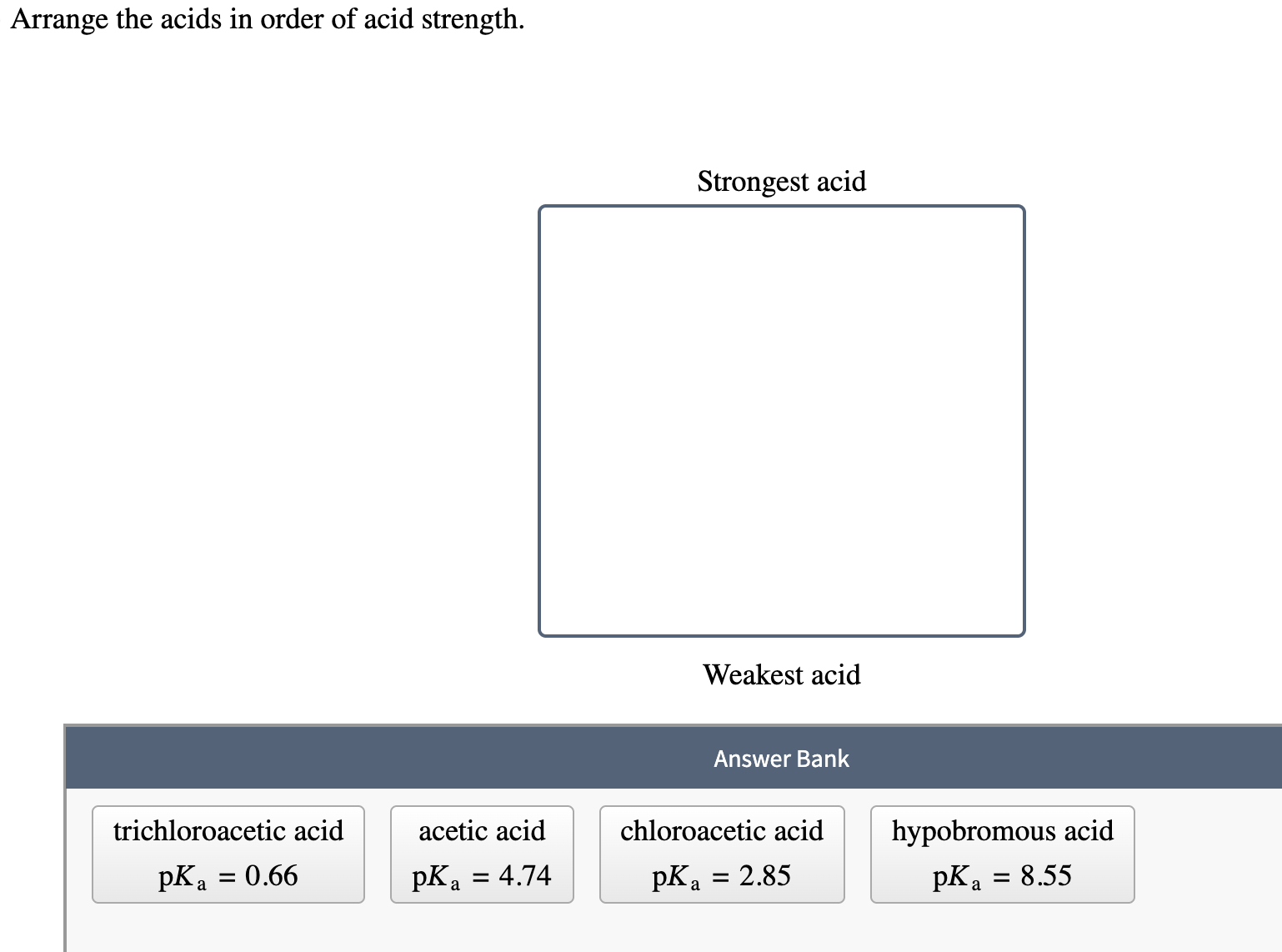
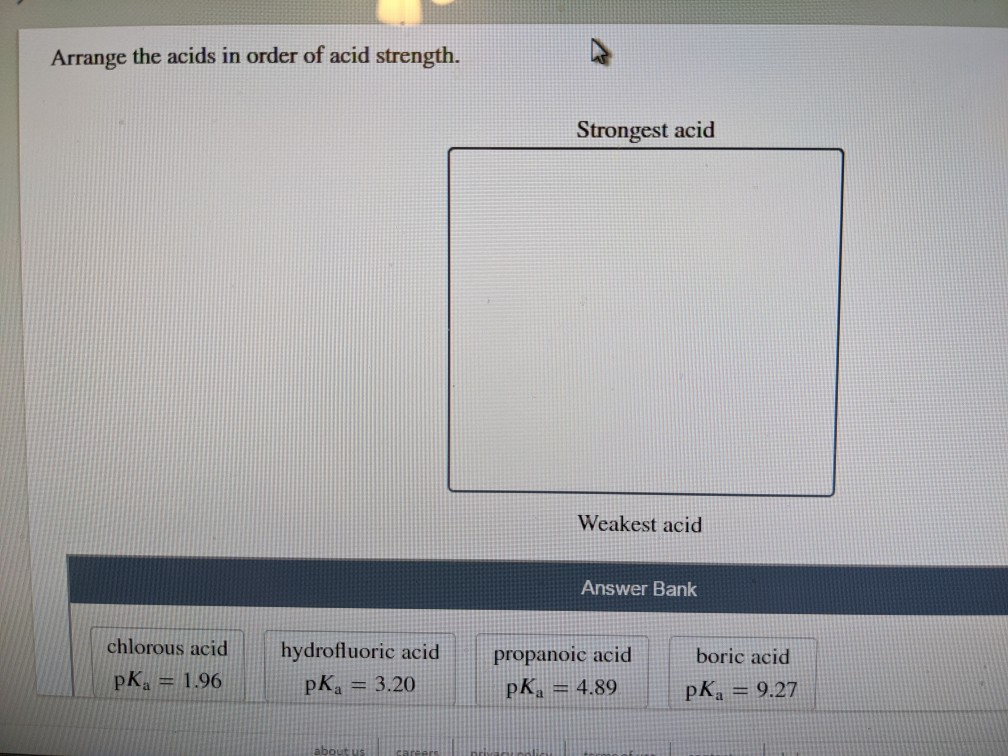
Arrange The Acids In Order Of Acid Strength.

Hey there, science explorers! Ever found yourself staring at a list of acids and wondering, "Which one is the real acid powerhouse?" It's kind of like looking at a lineup of superheroes, right? You've got your everyday folks, and then you've got your planet-saving titans. Well, acids are a lot like that, and today, we're going to chat about how to sort them out based on their acid strength. No need to get all stressed about it; we're keeping it super chill and curious.
So, what even is an acid, in the first place? Think about it this way: acids are those substances that are really good at donating something. In the chemical world, what they love to donate is a proton, which is basically a positively charged hydrogen atom. When an acid is hanging out in water, it’s like a generous friend who’s always ready to share. It splits off one of its hydrogen atoms (with its positive charge) and leaves behind something called a conjugate base.
Now, the strength of an acid isn't about how corrosive it is (though some are definitely more ouchy than others!). It’s really about how easily and how completely it gives away that proton. A strong acid is like a super-eager beaver. It's practically jumping at the chance to dump its proton into the water. It does this almost 100% of the time. A weak acid, on the other hand, is a bit more hesitant. It might donate a proton, but it’s also happy to keep it. So, only a small percentage of its molecules will actually let go.
Must Read
Imagine you have a bunch of balloons at a party. A strong acid is like someone who pops balloons the instant they get them. Pop, pop, pop! All the balloons are gone, fast! A weak acid is more like someone who might poke a balloon a little, maybe it deflates a bit, but most of them stay mostly intact. You still have some popped balloons, but not nearly as many as the super-eager balloon popper.
So, how do we actually arrange these acids from strongest to weakest? It all comes down to this concept of dissociation. Dissociation is just the fancy word for when the acid splits apart in water. The more it dissociates, the stronger it is. We often use this little symbol, Ka, to measure this. Think of Ka as the acid's "proton-donating score." A higher Ka means a stronger acid. If the Ka is really, really big (like, astronomically large!), we usually just call it a strong acid because it dissociates so completely.
On the flip side, we have the pKa. This is just the negative logarithm of Ka. Don't let the math scare you! It's just another way to talk about acid strength. The lower the pKa, the stronger the acid. So, if you see a pKa of, say, -2, that's a mighty strong acid. If you see a pKa of 10, that's a much weaker guy.

Let's get to the fun part: some examples! We often talk about these common acids, and putting them in order can be pretty eye-opening.
The Usual Suspects: Common Acids
Here are a few acids you might have heard of, and we're going to put them in order from strongest to weakest. Get ready for the lineup!
Hydrochloric Acid (HCl)
Ah, hydrochloric acid. This is one of the classics, a real heavy hitter. When HCl meets water, it’s like a toddler at a candy store – it just goes wild and dissociates almost completely. Its Ka is huge, and its pKa is very, very small (think around -6 to -7). It's practically a superhero acid, right up there with the best of them.
Sulfuric Acid (H2SO4)
Sulfuric acid is another beast! It's a diprotic acid, meaning it has two protons it can potentially donate. The first donation is super strong, similar to HCl (pKa around -3). The second donation is weaker, but still pretty significant. So, as a whole, it's definitely in the strong acid category. Think of it as a superhero with a secondary superpower that's a bit less flashy but still very effective.
Nitric Acid (HNO3)
Nitric acid is right there with HCl and H2SO4 in terms of being a super strong acid. It also dissociates almost entirely in water. Its pKa is around -1.4. It’s another one of those acids that you definitely don't want to mess with casually. It’s always ready to donate that proton and get the party started.
Phosphoric Acid (H3PO4)
Now we're starting to see a shift. Phosphoric acid is a triprotic acid (three protons to give), but unlike sulfuric acid, all its proton donations are significantly weaker. The first proton has a pKa of about 2.15. This makes it a weak acid. It's still an acid, but it's not as enthusiastic about giving away its protons as the ones we just talked about. It's more like a polite guest at the party who might offer you a drink, but they're not going to force it on you.
Acetic Acid (CH3COOH)
Ever heard of vinegar? Well, acetic acid is the main ingredient that gives vinegar its characteristic sour taste and smell! This is a classic example of a weak acid. Its pKa is around 4.76. This means that in a solution of acetic acid, most of the molecules are still holding onto their protons. It's much less likely to cause a dramatic reaction compared to the super strong acids. It's like the gentle breeze versus a hurricane – both are weather, but one is way more intense.
Carbonic Acid (H2CO3)
Carbonic acid is super interesting because it's what gives fizzy drinks their fizz! It forms when carbon dioxide dissolves in water. It's a weak acid, with a pKa of about 6.35 for its first proton. This is why those bubbles eventually disappear – the carbonic acid is constantly trying to reach an equilibrium, and it’s not a strong enough acid to keep them around forever. It’s like the temporary excitement of a pop-up shop versus a permanent fixture.

Water (H2O)
Now, this is where things get a little meta and super cool. Water itself can act as an acid (and a base!). When water donates a proton, it forms a hydroxide ion (OH-). This is an incredibly weak process. The pKa for water is around 14. This means water is a very, very weak acid. It's the benchmark for what we consider practically neutral. It's like the ultimate laid-back friend who rarely gets involved in anything dramatic unless absolutely necessary.
Putting It All Together: The Order!
So, if we were to arrange these guys from the most acidic (strongest) to the least acidic (weakest), based on their tendency to donate protons, here's a simplified rundown:
Strongest Acids:
- Hydrochloric Acid (HCl)
- Sulfuric Acid (H2SO4 - first proton)
- Nitric Acid (HNO3)
Weak Acids:

- Phosphoric Acid (H3PO4 - first proton)
- Acetic Acid (CH3COOH)
- Carbonic Acid (H2CO3 - first proton)
Extremely Weak Acid:
- Water (H2O)
Isn't that neat? It's like a ladder of proton-giving prowess! Understanding this order helps us predict how these substances will behave in reactions. It's not just about memorizing facts; it's about understanding the underlying "personality" of these molecules.
Why does this even matter, you ask? Well, knowing acid strength is crucial in so many fields. In the kitchen, it helps us understand why lemon juice (citric acid, another weak acid) makes things taste tart but doesn't eat through your plate. In medicine, it’s vital for understanding how drugs work in our bodies. And in industry, it's essential for everything from making fertilizers to cleaning products. It's a fundamental concept that unlocks a whole world of chemical understanding.
So next time you see an acid, take a moment to wonder about its strength. Is it a proton-donating champion or more of a reluctant giver? It's all part of the amazing, ongoing adventure of exploring the world of chemistry!