Aromatic Vs Antiaromatic Vs Nonaromatic Quiz
Hey there! So, you’ve been diving into the wild, wonderful world of organic chemistry, huh? Awesome! It’s like a whole secret language, right? And let me tell you, some of the coolest words are the ones that sound super fancy, like… aromatic, antiaromatic, and nonaromatic. Sounds like a perfume convention and a chemical war zone had a baby, doesn’t it?
Honestly, when I first saw these terms, I probably made a face like I’d just bitten into a lemon. What even are these things? Are they related? Do they have opinions on cats? (Spoiler alert: probably not on cats. That’s where my expertise lies, not theirs.)
But don't worry! We're going to break it down, easy peasy. Think of it like this: we’re going to have a little quiz. A mental coffee break, if you will. No actual pens or papers needed, just your brilliant brain. Ready to put on your chemical detective hat? Let's do this!
Must Read
The Big Three: What's the Buzz?
So, these three terms, aromatic, antiaromatic, and nonaromatic, basically describe how certain molecules behave. It's all about their stability and their electron setup. Think of electrons as little party animals, and they have very specific requirements for a good time. Aromatic compounds? They're the life of the party, super chill, and always invited back. Antiaromatic? Oh boy, they're the ones who cause drama, stir up trouble, and are definitely not invited to the next gathering. Nonaromatic are just… there. They’re the guests who are polite but don't really add much to the conversation. A bit bland, if we're being honest.
It all comes down to a few key rules, like a secret handshake or a password. You gotta know the password to get into the cool club (aromatic). Mess up the handshake, and you might end up in the awkward "antiaromatic" situation. And if you just can't get it right, well, you're nonaromatic. No biggie, just not the main event.
Rule #1: The Ring Thing!
First off, for a molecule to even consider being aromatic or antiaromatic, it has to be a ring. Yep, a cycle. If it's just a straight line of atoms, it's automatically relegated to the "nonaromatic" category. It's like trying to have a round-robin tournament with a single player. Doesn't quite work, does it?
So, our first mental question is: Is it a ring? If the answer is NO, then BAM! Nonaromatic. Case closed. You can go grab another coffee. But if the answer is YES, then we gotta keep digging. This is where it gets juicy!
Rule #2: Planar Power!
Okay, so it’s a ring. Great start! Now, is it flat? Like, perfectly flat? This is called being planar. Imagine trying to stack a bunch of perfectly flat frisbees. They fit nicely, right? Now imagine trying to stack wobbly dinner plates. Chaos! Aromatic and antiaromatic compounds are like those perfectly stacked frisbees. They need to be able to lie down flat, all in the same plane.
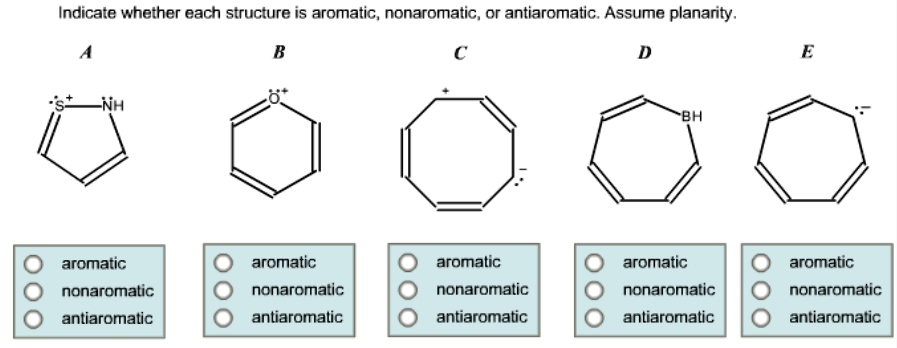
Why is flatness so important? It’s all about the electrons, my friend. When a molecule is flat, its p orbitals (those are like little electron highways) can line up nicely. This allows for something super special called delocalization. More on that in a sec!
If a ring isn't planar, it's a bit of a fail for aromaticity. It’s like trying to have a dance party in a room with a ridiculously low ceiling. Things just don't flow. So, if it's a ring but it's all bent out of shape, it's usually going to be nonaromatic. It's not causing trouble, but it's not winning any popularity contests either.
Rule #3: The Electron Count (This is the Real Showstopper!)
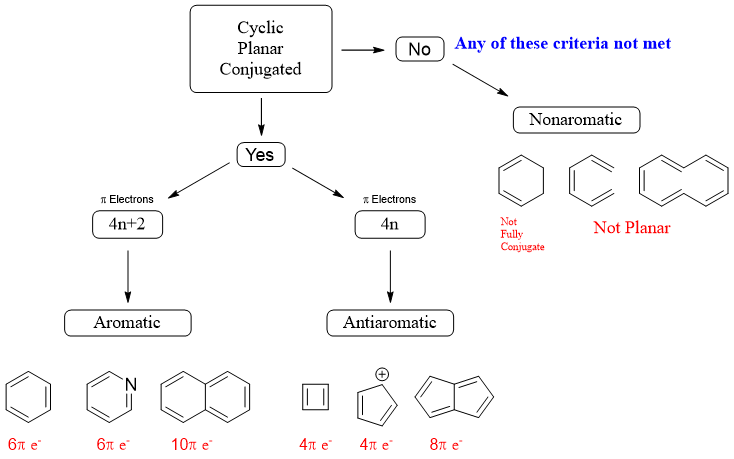
Alright, this is where the magic (or the madness) really happens. For our ring to be either aromatic or antiaromatic, it needs to have a specific number of delocalized pi electrons. This is where Hückel's Rule comes in, and honestly, it’s like a secret code you need to crack.
The magic numbers are 2, 6, 10, 14, and so on. Basically, you're looking for a number of pi electrons that follows the pattern 4n + 2, where 'n' is a whole number (0, 1, 2, 3...).
So, if you have a ring that’s planar and has 2 pi electrons, it's aromatic! Woohoo! If it has 6 pi electrons and is planar, aromatic again! If it has 10 pi electrons and is planar, you guessed it – aromatic! These guys are super stable, like a well-loved, cozy armchair. They're happy just the way they are.
Now, what if the ring is planar, but the pi electron count is… well, not 4n + 2? What if it’s 4, 8, 12, 16, etc.? This is where the dreaded 4n rule comes in. If it follows this 4n pattern of pi electrons and is planar, then congratulations! You’ve found an antiaromatic compound. These guys are the opposite of stable. They're like a ticking time bomb, constantly trying to rearrange themselves to get away from this unstable state. They're the chemical equivalent of stubbing your toe – it just feels wrong.
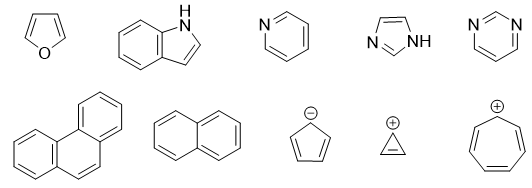
What About Lone Pairs and Empty Orbitals?
Okay, this is where it can get a little tricky, like trying to sort laundry in the dark. Sometimes, atoms in the ring have lone pairs of electrons (those are pairs of electrons hanging out with no partner) or empty p orbitals. These can participate in the pi system, meaning they can contribute to the delocalization and count towards your pi electron total. It's like a guest who brings a friend to the party – they both count towards the headcount!
So, when you're counting those pi electrons, don't forget to check if any lone pairs or empty orbitals are in the right position to get involved in the ring. If they can, they become part of the pi electron cloud. Sneaky, right?
Let's Play "Is It Aromatic, Antiaromatic, or Nonaromatic?"
Alright, enough theory! Let's put this knowledge to the test with some imaginary molecules. Grab your mental magnifying glass!
Scenario 1: Benzene! (The Poster Child)
We all know benzene, right? That classic six-membered ring with alternating double and single bonds. It’s practically the mascot for aromaticity.
- Is it a ring? YES.
- Is it planar? YES. (It's super flat!)
- How many pi electrons? It has three double bonds, and each double bond has 2 pi electrons. So, 3 x 2 = 6 pi electrons.
Does 6 fit the 4n + 2 rule? Let 'n' = 1. 4(1) + 2 = 6. YES! So, benzene is undeniably, gloriously AROMATIC! It's super stable and loves hanging out like this.
Scenario 2: Cyclobutadiene! (The Troublemaker)
Now, let's look at cyclobutadiene. It's a four-membered ring with alternating double bonds. Looks kinda like a square with a party going on inside.
- Is it a ring? YES.
- Is it planar? YES. (It’s pretty flat.)
- How many pi electrons? It has two double bonds, so 2 x 2 = 4 pi electrons.
Does 4 fit the 4n + 2 rule? Nope. Does it fit the 4n rule? Let 'n' = 1. 4(1) = 4. YES! So, cyclobutadiene is ANTIAROMATIC! Uh oh. This little guy is unstable. It hates being planar with those 4 pi electrons. It’ll often distort itself to avoid this fate, becoming nonaromatic, but if forced into planarity, it's a chemical mess. Poor thing.
Scenario 3: Cyclohexane! (The Chill One)
Cyclohexane. This is the saturated version of benzene. It’s a six-membered ring, but it’s got all single bonds. It’s not a ring of double bonds; it’s more like a ring of… well, single bonds.
- Is it a ring? YES.
- Is it planar? NO. This one is famously not planar. It prefers to exist in a "chair" or "boat" conformation to relieve ring strain. Think of it like a hula hoop that’s been bent into a more comfortable shape.
- How many pi electrons? Since there are no double bonds, there are 0 pi electrons.
Because it's not planar, even though it has a ring and 0 pi electrons (which technically fits 4n+2 with n=-1/2, but we only use positive integers for n!), it's automatically NONAROMATIC. It's not stable in the aromatic sense, and it's not unstable in the antiaromatic sense. It's just… there. Chilling. Doing its own thing.
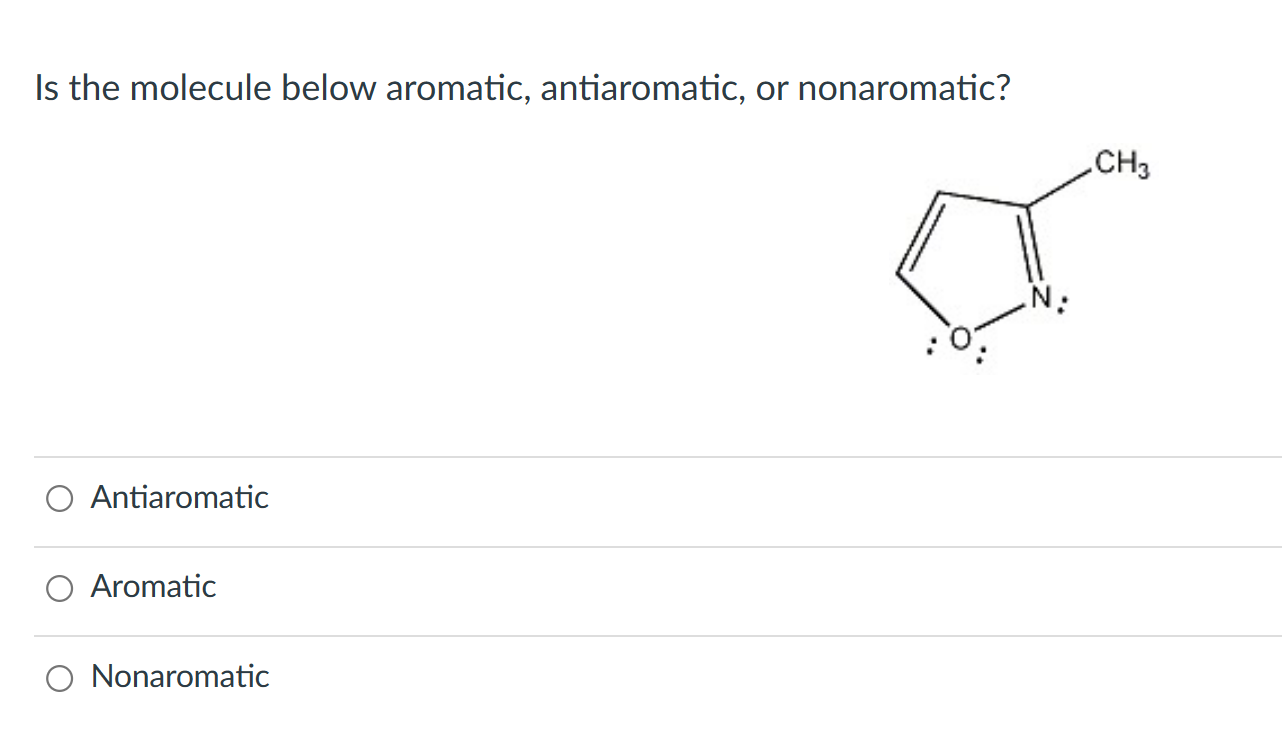
Scenario 4: Pyrrole! (The One with a Secret)
Pyrrole is a five-membered ring containing one nitrogen atom. The nitrogen has a lone pair of electrons.
- Is it a ring? YES.
- Is it planar? YES. (It likes to be flat.)
- How many pi electrons? There’s one double bond (2 pi electrons). BUT, the nitrogen’s lone pair of electrons are in a p orbital that is perfectly aligned to participate in the ring! So, we add those 2 electrons from the lone pair. Total pi electrons = 2 (from the double bond) + 2 (from the nitrogen’s lone pair) = 4 pi electrons.
Does 4 fit the 4n + 2 rule? Nope. Does it fit the 4n rule? Let 'n' = 1. 4(1) = 4. YES! So, pyrrole is ANTIAROMATIC? Hold on! BUT... pyrrole is actually quite stable. What’s going on? This is where the rules get a little fuzzy, and chemists have debates. In many contexts, pyrrole is considered AROMATIC. Why? Because its stability overrides the strict '4n' rule in this case. It's like a rule-breaker who gets away with it because they're just that good. The lone pair can participate, but it doesn't have to. The molecule prefers the aromatic 6 pi electron system (from the ring and one double bond, if the lone pair didn't participate) over the antiaromatic 4 pi electron system. So, it chooses aromaticity.
This is a great example of how things aren’t always black and white in chemistry. Sometimes, stability wins!

Scenario 5: Pyridine! (The "Normal" Heterocycle)
Pyridine is another six-membered ring, but it has a nitrogen atom instead of one of the carbons.
- Is it a ring? YES.
- Is it planar? YES.
- How many pi electrons? It has three double bonds, so 3 x 2 = 6 pi electrons. What about the nitrogen’s lone pair? That lone pair is in an sp2 orbital, in the plane of the ring, not a p orbital. So, it doesn’t participate in the pi system.
Does 6 fit the 4n + 2 rule? Let 'n' = 1. 4(1) + 2 = 6. YES! So, pyridine is AROMATIC. See? It’s all about what those electrons are doing!
The Takeaway Message (Don't Stress Too Much!)
Look, these concepts can seem a bit daunting at first. It’s like learning a new dance. You’re going to step on some toes, maybe trip a little. But the more you practice, the more natural it becomes.
The key things to remember are: ring, planar, and the 4n+2 pi electron rule. If a molecule ticks all those boxes, it's probably aromatic. If it's a ring, planar, but has 4n pi electrons, it's likely antiaromatic (and unstable!). If it's a ring but not planar, or has no pi electrons, it's nonaromatic. Easy, right? (Okay, maybe not that easy, but you're getting there!)
Don't be afraid to draw it out, count those electrons, and think about the shape. And remember, sometimes there are exceptions that make chemistry so darn interesting. You're doing great just by diving in and asking these questions. So, next time you see one of these terms, you won't run screaming for the hills (or the nearest aromatic perfume). You'll be like, "Ah, I see what you're doing there, molecule! Let's see if you follow the rules."
Keep practicing, keep questioning, and most importantly, keep enjoying the amazing world of chemistry. You've got this!